In a recent study posted to the medRxiv* preprint server, investigators in Singapore showed that severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) breakthrough infection elicited broadly specific virus-selective nasal resident CD8 and CD4 T cells.
 Study: SARS-CoV-2 infection in vaccinated individuals induces virus-specific nasal resident CD8 and CD4 T cells of broad specificity. Image Credit: Corona Borealis Studio / Shutterstock
Study: SARS-CoV-2 infection in vaccinated individuals induces virus-specific nasal resident CD8 and CD4 T cells of broad specificity. Image Credit: Corona Borealis Studio / Shutterstock

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Background
The upper respiratory tract in humans is the entry point and the initial replication site of SARS-CoV-2. Due to their elevated angiotensin-converting enzyme 2 (ACE2) receptor expression, nasal ciliated cells are rapidly infected and sustain most early viral production in vivo. Thus, T cells in the upper airway may provide an essential layer of immunity against CoV disease 2019 (COVID-19) by quickly recognizing SARS-CoV-2-infected cells and rapidly clearing the virus. However, it is unclear if COVID-19 vaccination or SARS-CoV-2 infection provokes nasal resident T cells selective for different SARS-CoV-2 proteins.
About the study
The objective of this study was to determine the presence of nasal resident SARS-CoV-2-specific T cells and their durability and functioning in donors vaccinated against COVID-19 with or without a SARS-CoV-2 breakthrough infection. The researchers assessed the SARS-CoV-2 specificity, function, and phenotype of the T cells from the nasal secretions of 35 SARS-CoV-2 vaccinees, 20 of whom developed COVID-19 after vaccination, and 15 subjects did not.
All subjects received two or three doses of the SARS-CoV-2 messenger ribonucleic acid (mRNA) vaccination, and 20 volunteers had a breakthrough COVID-19 incidence detected by the SARS-CoV-2 rapid lateral flow test. Nasal samples were taken eight to 149 days following the last COVID-19 vaccination and seven to 61 days post negative COVID-19 rapid lateral flow test.
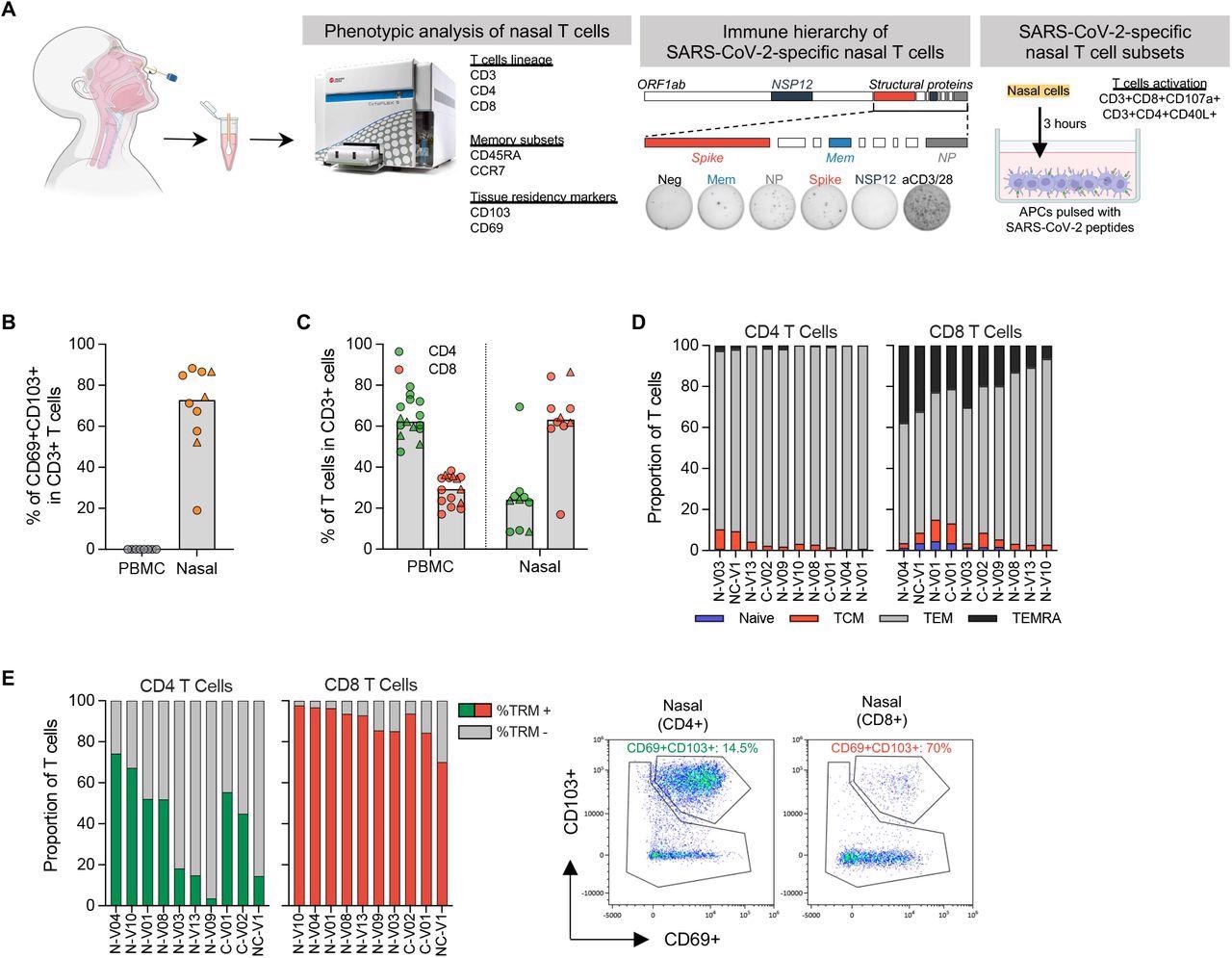
Phenotypic analysis of T cells in nasal secretion. (a) Schematic of experimental design; (b) Frequency of tissue resident T cells present in PBMC (n=8) and nasal secretion (n=10). Convalescent vaccinees are indicated by a triangle symbol; (c) Frequency of CD4 and CD8 T cells present in PBMC (n=14) or nasal cells (n=10). Convalescent vaccinees are indicated by a triangle symbol; (d) Proportion of naïve (CCR7+CD45RA+), central (CCR7+CD45RA - TCM), effector (CCR7-CD45RA - TEM) and terminally differentiated (CCR7-CD45RA+; TEMRA) memory CD4+ and CD8+ nasal T cells; (e) Expression of tissue resident markers in CD8+ and CD4+ nasal T cells (n=10) and corresponding representative plots.
A flocked swab was placed into the inferior turbinates of each subject and spun 10 to 20 times to obtain nasal lining fluid. Following the subsequent processing, the isolated nasal cells were quantified using flow cytometric assessment and used in succeeding investigations. Furthermore, peripheral blood was obtained, and Ficoll-Paque density gradient centrifugation was used to separate peripheral blood mononuclear cells (PBMC) from all procured blood samples. Isolated PBMCs were analyzed immediately or cryopreserved and kept in liquid nitrogen until employed in the experiments.
The complete protein sequences of SARS-CoV-2 nucleocapsid protein (NP), non-structural protein 12 (NSP12), spike (S), and membrane (mem) proteins were synthesized as 15-mer peptides that coincided with 10 amino acids. Additionally, freshly obtained nasal cells were activated with peptide collections in an interferon (IFN)-γ enzyme-linked immunospot (ELISPOT) experiment. Furthermore, the team conducted cytokine secretion analysis and phenotyping of nasal T cells and SARS-CoV-2-specific T cells.
Results
Study results showed that breakthrough SARS-CoV-2 infection of COVID-19 vaccinees generated tissue-resident CD8 T cells in the upper airways that were selective for specific SARS-CoV-2 proteins. For at least three months following infection, these SARS-CoV-2 multi-specific tissue-resident CD8 T cells exist at detectable levels using assays evaluating T cell functioning, such as T cell degranulation, direct cytokine assay, and IFN-γ generation by ELISPOT.
On the other hand, similar tests did not identify any T cells selective for SARS-CoV-2 S or any other proteins in the upper airway of COVID-19 vaccinees without infection status, albeit the detection of S-specific T cells in their peripheral blood. Moreover, the current data did not explain if SARS-CoV-2-specific T cells seen in vaccinees' nasal cavity following infection were primed in nasal-linked lymphoid tissue or more structured lymphoid tissues in the upper airway, like the lingual, pharyngeal, tonsils, and palatine.
The lack of SARS-CoV-2-specific T cells in vaccinated only subjects' nasal secretions indicates that SARS-CoV-2 infection, not COVID-19 parenchymal mRNA S vaccination, attracts and sustains a large number of SARS-CoV-2 tissue-resident memory T cells in the nasal cavity. Moreover, analysis of SARS-CoV-2-specific T cells in the nasal cavity in individuals with breakthrough COVID-19 revealed that elevated CD4 and CD8 T cells specific for distinct SARS-CoV-2 antigens were not limited to circulating peripheral blood.
Conclusions
The study findings showed that nasal-resident IFN-γ secreting SARS-CoV-2-specific CD4 and CD8 T cells were only found in COVID-19 vaccinees who developed SARS-CoV-2 breakthrough infection. Furthermore, the induction of CD4 and CD8 T cells specific for distinct SARS-CoV-2 proteins, such as NSP-12 and NP, that remained in the nasal cavity about three months after COVID-19 was not suppressed by vaccine priming of S-specific T cells.
Overall, the present work emphasized the role of viral nasal challenge in the development of antiviral immunity specific to SARS-CoV-2 at the infection site and the immunological characteristics of COVID-19 hybrid immunity. Furthermore, the authors stated that additional longitudinal studies were required to determine whether or not SARS-CoV-2-targeting nasal resident T cells can survive for years like circulating memory T cells after SARS-CoV-1 infection.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
SARS-CoV-2 infection in vaccinated individuals induces virus-specific nasal resident CD8 and CD4 T cells of broad specificity; Joey Ming Er Lim, Anthony Tan, Nina Le Bert, Shou Kit Hang, Jenny Low, Antonio Bertoletti. medRxiv preprint 2022, DOI: https://doi.org/10.1101/2022.05.18.22275292, https://www.medrxiv.org/content/10.1101/2022.05.18.22275292v1
- Peer reviewed and published scientific report.
Lim, Joey Ming Er, Anthony Tanoto Tan, Nina Le Bert, Shou Kit Hang, Jenny Guek Hong Low, and Antonio Bertoletti. 2022. “SARS-CoV-2 Breakthrough Infection in Vaccinees Induces Virus-Specific Nasal-Resident CD8+ and CD4+ T Cells of Broad Specificity.” Journal of Experimental Medicine 219 (10). https://doi.org/10.1084/jem.20220780. https://rupress.org/jem/article/219/10/e20220780/213399/SARS-CoV-2-breakthrough-infection-in-vaccinees.