Coronavirus disease 2019 (COVID-19) symptoms may persist beyond the acute phase of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, typically about 28 days following the initial diagnosis. Post-acute sequelae of coronavirus disease 2019 (PASC), often referred to as ‘long COVID,’ is often used to describe the presence of these long-term symptoms following the initial recovery from COVID-19. PASC can cause significant morbidity, despite apparent SARS-CoV-2 clearance from the body.
Several mechanisms for long COVID development have been proposed, such as SARS-CoV-2 persistence or immune dysregulation with altered humoral responses. However, further research is needed to determine the exact mechanisms responsible for this condition.
Study: Impact of cross-coronavirus immunity in post-acute sequelae of COVID-19. Image Credit: joshimerbin / Shutterstock.com

 *Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
In a recent study published on the medRxiv* preprint server, researchers perform comprehensive antibody (Ab) profiling against SARS-CoV-2, several endemic pathogens, and vaccine antigens among rheumatic disease patients with or without PASC. This information was used to determine whether SARS-CoV-2 or other pathogen-targeted humoral responses evolve uniquely in long COVID.
About the study
In the present study, researchers explored humoral immune response alterations to SARS-CoV-2, common CoVs, herpesviruses, and several vaccine antigens to investigate whether pathogen-targeted Abs could provide information relevant to the pathogenesis of long COVID. Moreover, they focused on a single long COVID endotype, wherein they applied systems serology to rheumatic disease patients who developed mild to moderate SARS-CoV-2 infection, 50% of whom developed long COVID.
From March 1, 2020, onwards, all systemic autoimmune rheumatic disease (SARD) participants with polymerase chain reaction (PCR)- or antigen-confirmed COVID-19 were identified by the Massachusetts General Brigham (MGB) healthcare system. In addition, patient information was supplemented by rheumatologist referrals of SARS-CoV-2-infected patients.
The study comprised individuals who did not require hospital admission for COVID-19 and excluded individuals with a history of fibromyalgia, mechanical backache, osteoarthritis, gout, or pseudogout without SARD.
Antigen-specific isotype titers and Fc receptor (FcR)-binding were measured by multiplexed Luminex assays. Median fluorescence intensity (MFI) scores and avidity index values were also calculated.
Ab-dependent neutrophil phagocytosis (ADNP) and seropositive assays were also performed. To study the enrichment of a particular inflammatory-driven long COVID endotype, humoral responses of COVID-19-recovered patients with SARD were assessed.
The isotype, subclass, and Fc-R-binding profiles against SARS-CoV-2 nucleocapsid (N), spike (S), S subunit 2 (S2), and S receptor-binding domain (RBD) were assessed among the two groups of rheumatic patients. The team investigated whether alterations in humoral responses to commonly administered vaccines and endemic pathogenic organisms could provide insights into humoral differences among SARD patients with or without long COVID.
The team evaluated Ab isotype and Fc-gamma receptor (FcγR)-binding titers across multiple antigens, including routine vaccine-incorporated antigens such as rubella, tetanus, mumps, and measles, herpesviruses including cytomegalovirus (CMV), herpes simplex virus 1 (HSV1), varicella zoster virus (VZV), and Epstein Barr virus (EBV), as well as other CoVs using the S, S1, and S2 proteins of OC43, HKU1, and SARS-CoV-1.
Humoral responses against other endemic pathogens such as the respiratory syncytial virus (RSV), Staphylococcus aureus, influenza virus, and control pathogenic organisms such as Ebola were assessed. Partial least squares discriminant analysis (PLS-DA) and elastic net regularization were performed to visualize the humoral differences between the two groups.
Study findings
In the analysis, 17 long COVID and SARD patients and 26 non-long COVID patients were profiled, 95% vaccinated against COVID-19. Moreover, 79% of the study participants were women.
Rheumatoid arthritis was the most commonly reported rheumatic disease, whereas tumor necrosis factor (TNF) inhibitors were the most widely used therapeutic agents.
Reduced anti-SARS-CoV-2 S and S2 immunoglobulin M (IgM) and IgG2 titers, as well as greater OC43- and CMV-targeted inflammatory Ab titers, were observed among long COVID patients. Conversely, non-long COVID individuals were likelier to exhibit more significant isotype/subclass titers but not FcγR binding titers. Anti-CMV responses were primarily driven by CMV-seroprevalence imbalances.
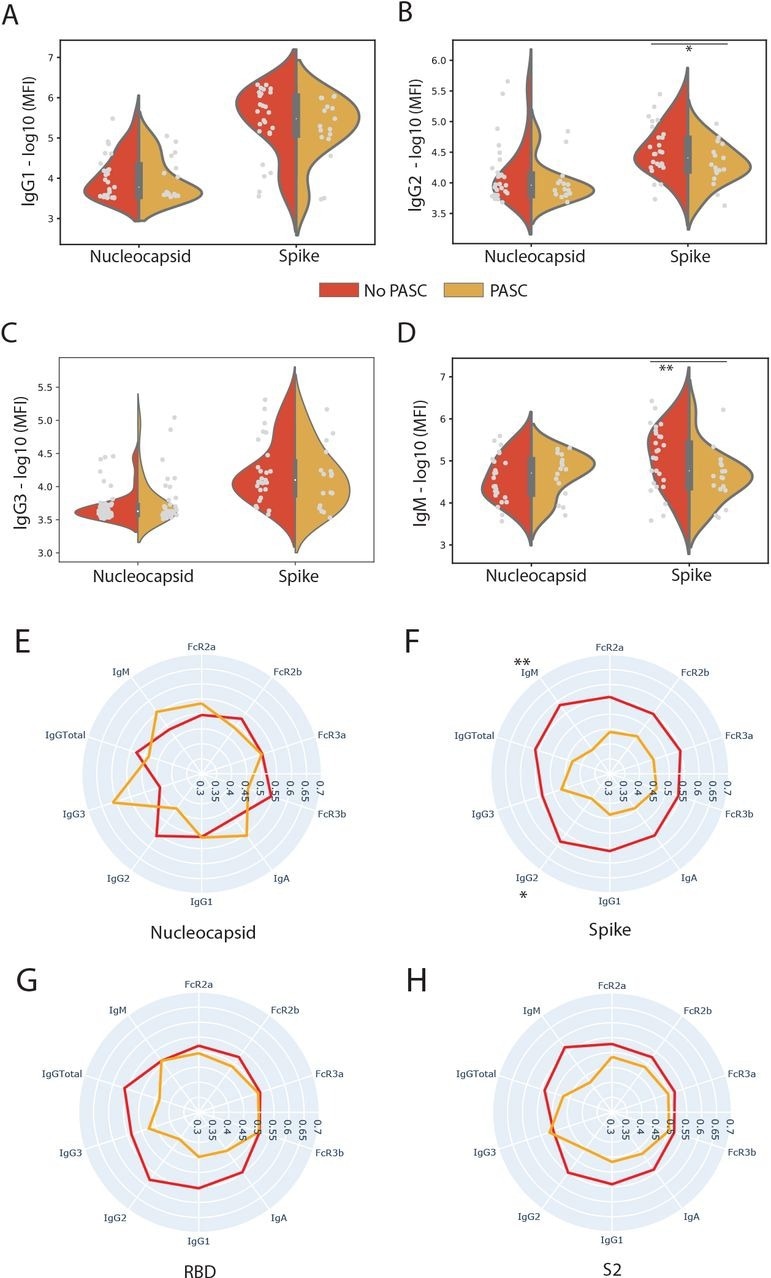
SARS-CoV-2 Spike-directed responses are lower in individuals who experienced. (A, B, C, D) The violin plots show the IgG1, IgG2, IgA1, and IgM titer against SARS-CoV-2 Nucleocapsid (N) and Spike in individuals who experienced PASC (yellow) and those who did not (red). A two-sided Mann-Whitney U test determined significance. (E, F, G, H) The radar plots show the mean percentile rank of antibody titers and Fc receptor (FcR)-binding against Nucleocapsid, Spike (Full Spike, RBD, and S2 domain) and for individuals who experienced PASC (Yellow) and those who did not (Red). Significance was determined by a two-sided Mann-Whitney U test. * p = 0.05, p < ** 0.05
Anti-OC43 responses were mainly driven by high avidity anti-OC43 IgM titers. Class-switched FcγR-binding OC43 responses correlated inversely with the quality and quantity of anti-SARS-CoV-2 humoral responses.
Notably, OC43 S FcγR-binding Abs were significantly elevated among long COVID patients, whereas IgG3 and IgM titers were not elevated. This indicates an expansion of the highly inflammatory IgG1 responses instead of novel OC43 Ab evolution among long COVID patients. Taken together, these findings suggest a potential role of previous common CoV back-boosting as a driver of incomplete SARS-CoV-2 Ab production among SARD patients who developed long COVID.
Anti-S2 responses showed a trend toward greater IgG1, IgG2, IgA, and IgM responses, as well as specific opsonophagocytic FcRs, including FcγR2a and FcγR 2b, among non-long COVID patients. In addition, elevated levels of anti-RSV IgM/IgG3, anti-influenza haemagglutinin (HA) FcγR2a and FcγR2b binding titers, anti-SARS-CoV S1 IgA, and anti-tetanus IgG Ab/IgM Ab were selectively enriched in non-long COVID individuals.
Contrastingly, anti-OC43 S FcγR3a and FcγR3b and anti-CMV gB FcγR2a/3b/IgM binding titers were enriched among long COVID patients. Thus, long COVID appears related to significantly altered polyclonal-type humoral responses to vaccines and common pathogens.
Conclusions
Overall, the study findings highlight the potential role of previous common CoV imprinting or ‘original antigenic sin’ in the incomplete maturation of the SARS-CoV-2-specific humoral immunity as a marker and potential mechanism in the persistence of long COVID symptoms.

 *Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.