In addition to humans, SARS-CoV-2 can infect various domestic, wild, and captive animals, including cats, dogs, deer, ferrets, gorillas, lions, mink, otters, tigers, and others. The virus has evolved rapidly, and several genetic variants have emerged. SARS-CoV-2 variants acquired mutations in the spike protein’s receptor-binding domain (RBD) that reportedly facilitate infectivity in rats/mice.
This raises concerns about the risk of reverse zoonosis in rodents. Two surveillance studies independently suggested possible exposure to rats from sewage in Belgium and Hong Kong, but viral genetic material was not detected in Norway rats (Rattus norvegicus). It remains uncertain whether rats are susceptible to more recent SARS-CoV-2 variants (Delta and Omicron).
 Study: SARS-CoV-2 Exposure in Norway Rats (Rattus norvegicus) from New York City. Image Credit: Holger Kirk / Shutterstock
Study: SARS-CoV-2 Exposure in Norway Rats (Rattus norvegicus) from New York City. Image Credit: Holger Kirk / Shutterstock
The study and findings
In the present study, researchers evaluated rat exposure to SARS-CoV-2 in NYC. They conducted surveillance between September 13 and November 21, 2021, when SARS-CoV-2 Delta was predominant in the city. Seventy-nine rats were captured/sampled from three sites in Brooklyn, NYC.
Nine and four rat serum samples were positive for immunoglobulin G (IgG) and IgM, respectively, against both spike and RBD. In addition, these positive samples were evaluated for neutralizing antibodies (nAbs) in a microneutralization assay against B.1 lineage, Alpha, and Delta variants, but all specimens tested negative.
Only four respiratory tissue specimens were positive for viral RNA on a reverse-transcription quantitative polymerase chain reaction (RT-qPCR) test. Cross-reactivity with six murine beta-CoV strains was not observed. Only two rats were positive for SARS-CoV-2 RNA and antibodies. Whole-genome sequencing of the four PCR-positive samples identified partial viral sequences with up to 21.3% genome coverage.
Phylogenetic analyses and molecular characterization suggested the sequences were associated with SARS-CoV-2 lineage B. Next, the team investigated whether SARS-CoV-2 variants could infect rats. Six-week-old rats were intranasally challenged with SARS-CoV-2 Alpha, Delta, or Omicron. Lung tissues and turbinates were harvested at 2 and 4 days post-infection (dpi).
High viral RNA levels and infectious virus titers were detected in both tissues and turbinates, despite the notable absence of clinical signs and body weight loss. Specifically, Delta-infected rats had the highest RNA copy numbers and infectious virus titers at 2 dpi. Viral antigen expression was evident in the lungs at either point in all infected animals, and the highest expression was observed in Delta-infected animals.
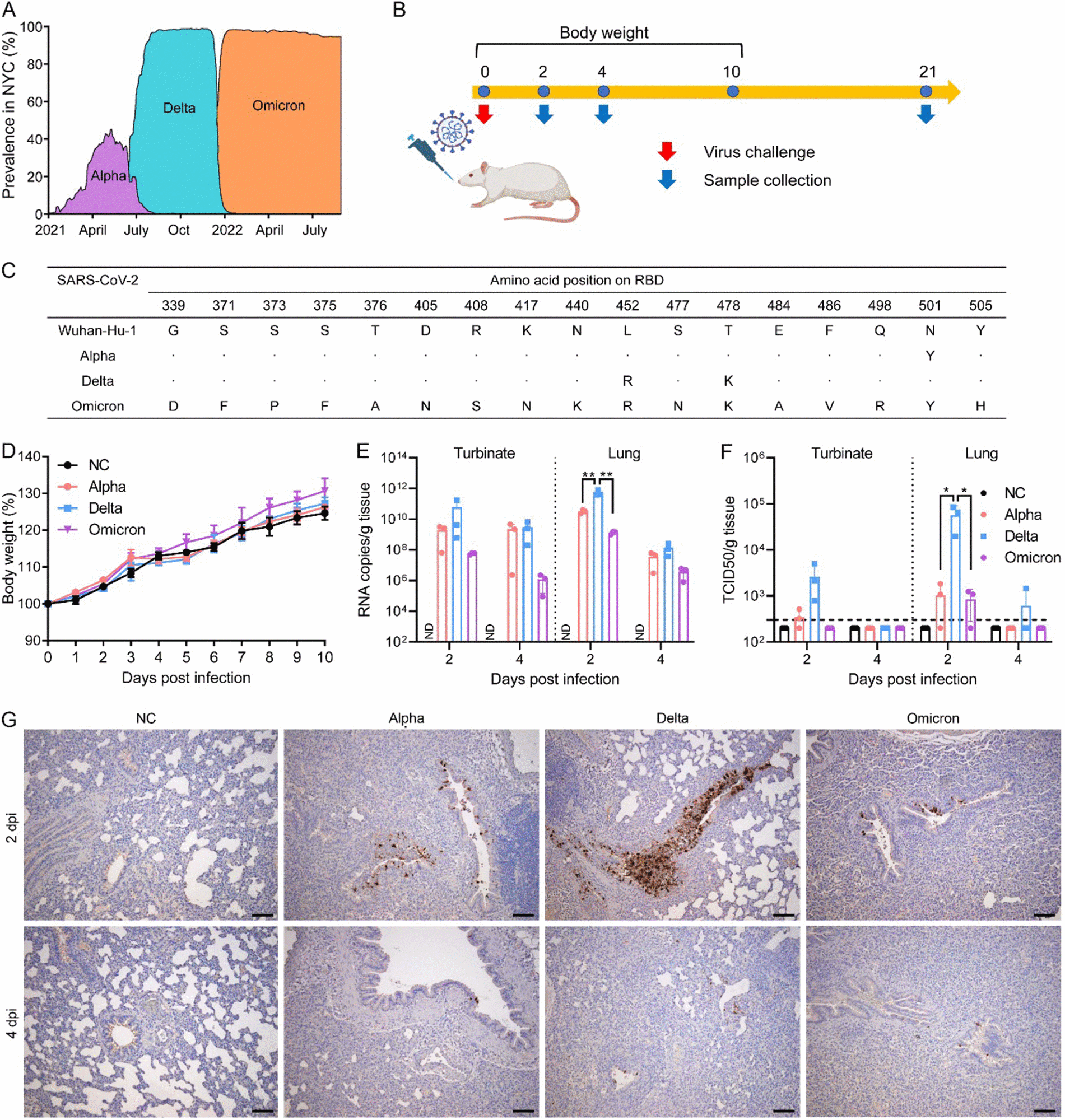 Sprague Dawley (SD) rats are susceptible to infection with Alpha, Delta, and Omicron variants. (A) Prevalence of Alpha, Delta, and Omicron variants in NYC. Figure adapted from https://outbreak.info. (B) Scheme of the virus challenge experiment using 6-week-old SD rats. (C) Amino acid changes of Alpha, Delta, and Omicron variants across the receptor binding domain (RBD) compared to Wuhan-Hu-1 (NCBI access no. MN908947.3). (D) Body weight of rats mock-infected or infected with either Alpha, Delta, or Omicron variant. (E and F) Viral RNA copies (E) and infectious viral titers (F) in the turbinate and lungs from rats infected with either Alpha, Delta, or Omicron variant at 2 or 4 days postinfection (dpi). *, P < 0.05; **, P < 0.01. (G) Detection of SARS-CoV-2 nucleocapsid protein in bronchial epithelial cells by immunohistochemistry at 2 and 4 dpi. Scale bar = 100 μm.
Sprague Dawley (SD) rats are susceptible to infection with Alpha, Delta, and Omicron variants. (A) Prevalence of Alpha, Delta, and Omicron variants in NYC. Figure adapted from https://outbreak.info. (B) Scheme of the virus challenge experiment using 6-week-old SD rats. (C) Amino acid changes of Alpha, Delta, and Omicron variants across the receptor binding domain (RBD) compared to Wuhan-Hu-1 (NCBI access no. MN908947.3). (D) Body weight of rats mock-infected or infected with either Alpha, Delta, or Omicron variant. (E and F) Viral RNA copies (E) and infectious viral titers (F) in the turbinate and lungs from rats infected with either Alpha, Delta, or Omicron variant at 2 or 4 days postinfection (dpi). *, P < 0.05; **, P < 0.01. (G) Detection of SARS-CoV-2 nucleocapsid protein in bronchial epithelial cells by immunohistochemistry at 2 and 4 dpi. Scale bar = 100 μm.
Further, immune responses to infection were determined by measuring the expression of cytokines/chemokines at 2 and 4 dpi and antibodies at 21 dpi. All variant infections elicited a pro-inflammatory expression profile, particularly at 2 dpi. Consistently, Delta-infected rats had the highest expression. IgG and nAbs were detectable against all variants at 21 dpi. IgM was not detected, irrespective of the infecting variant.
No significant differences between Alpha and Delta infections were observed in IgG titers. Nonetheless, the Delta variant induced higher IgG titers than Omicron. Delta-induced nAb titers were higher than those elicited by Alpha or Omicron. Sequencing of the virus from the animal lungs revealed no adaptive amino acid substitutions across the RBDs of three variants.
However, N74K substitution in the spike was observed in animals infected with the Alpha variant. Other substitutions were observed in the nucleoprotein, non-structural protein 6 (NSP6), and NSP13 in some rats infected with Delta/AlphaFinally, the researchers computationally modeled interactions between variant RBDs and rat angiotensin-converting enzyme 2 (ACE2).
Structural models showed that residue 452 was not directly engaging with ACE2 and was surrounded by many adjacent residues. Thus, L452R substitution in the Delta variant might alter structural conformation and modulate binding affinity. In line with this, in vitro binding assays revealed a two-fold higher binding affinity of Delta RBD with double L452R/T478K mutations than the original Wuhan-Hu-1 RBD.
Conclusions
Taken together, the authors demonstrated that rats in NYC were exposed to SARS-CoV-2. Over 16% of tested rats were seropositive, and 5% were positive for the viral RNA. Genomic analyses revealed that sequences were related to lineage B, which was prevalent in the early pandemic in NYC. The team speculated that lineage B viruses were enzootic in rats. Further, the team showed that SARS-CoV-2 Delta and Omicron could infect rats. Overall, the findings underscored the potential risk of secondary zoonosis from rats and the need to continue surveillance in rat populations.