SOLASCURE Ltd (SolasCure), a biotechnology company developing a novel treatment to transform chronic wound healing, today announced the successful completion of its second Phase II clinical trial, CLEANVLU2. The study demonstrated that its investigational product, Aurase Wound Gel (AWG), heals chronic wounds at a faster rate than current standard of care, providing clinical validation of its ability to deliver continuous enzymatic debridement while simultaneously activating wound healing.
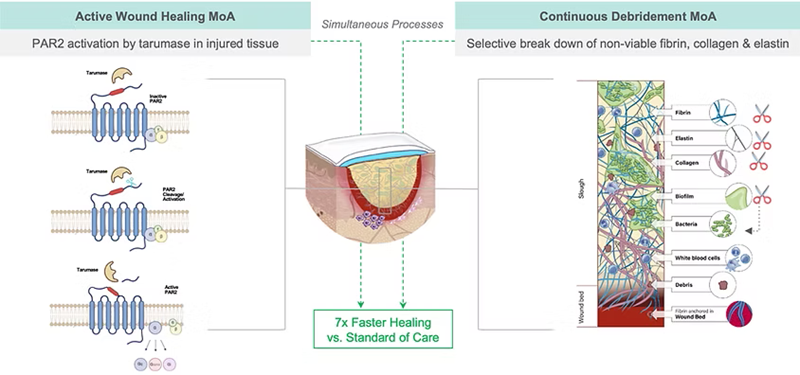 Image Credit: SolasCure
Image Credit: SolasCure
AWG combines a proprietary hydrogel formulation with the active pharmaceutical ingredient (API) tarumase. Following earlier Phase IIa clinical study, CLEANVLU, which established proof of concept and demonstrated a strong safety profile and pain-free application, CLEANVLU2 further evaluated efficacy and dose response in patients with treatment-resistant venous leg ulcers. At higher concentrations of tarumase, AWG demonstrated significantly enhanced clinical activity, debriding sloughy wounds 22 times faster and achieving rates of healing seven times faster than standard of care (hydrogel in combination with advanced moist wound healing dressings and compression bandaging systems). After 26 days, AWG achieved 65 % debridement versus 9 % in control (p=0.005) and 58 % wound area reduction versus 15 % in control (p=0.022).
The results indicate a dual mechanism of action where, in addition to continuously removing necrotic tissue, API tarumase activates PAR2 receptors involved in tissue repair, initiating healing pathways from the first application. AWG maintained an excellent safety profile, caused no additional pain to patients, and was associated with improvements in quality of life across physical, psychological, and daily living measures.
Chronic wounds remain a significant and growing unmet medical need, affecting millions of patients worldwide and placing a substantial burden on healthcare systems. Current therapies primarily focus on wound bed preparation rather than directly stimulating the biological processes required for healing. This clinical milestone highlights the potential of AWG to address wound bed preparation, debridement, and sustained healing within a single therapeutic, supporting further development and engagement with investors and strategic partners.
Full clinical data from CLEANVLU2 will be submitted for peer-reviewed publication.
These Phase II results represent a highly encouraging advance in the treatment of chronic wounds. From a clinical perspective, the data demonstrate very effective debridement alongside meaningful activation of the biological processes leading to healing."
Rob Kirsner, M.D., PhD, Head of Medical Advisory Board at SolasCure, Chairman and Harvey Blank Professor of Dermatology, University of Miami