Owing to the SPECTROstar Nanoand Brewing and Distilling Analytical Services, the past couple of years have seenadvancements in quality testing and analysis of alcoholic beverages. A distinct fingerprint spectral curve is shown by the UV/Vis absorbance spectra of the original spirit, which cannot be replicated in diluted or counterfeit samples. The SPECTROstar Nano is capable of performing measurements in both microplate and cuvette form, enabling flexibility, speed and consistency in measurements.
Testing for free amino nitrogen (FAN) is a part of standard brewing quality control analyses. This enables the protein content to be estimated and serves as an indicator of beer quality. The traditional way of determining protein content in beer is based on Kjeldahl assays, but these are costly and elaborate assays. The ninhydrin-based FAN assay for cuvettes is a suitable alternative to this assay and has been used as a standard method by the brewing industry for 40 to 50 years. Using ninhydrin, primary amino acids can be detected. As seen in Figure 2, the result is a blue dye showing an absorbance maximum at about 570nm. A standard substance for this assay is the amino acid glycine.
The next step was to decrease the volume for the standard cuvette assay and shift to a multi-well format. By scaling the test down, the need for costly reagents is reduced, waste is minimized and disposal becomes simpler and less costly. The integrated spectrometer is capable of taking spectra over time and overlay plots of all measurements are shown by the MARS data evaluation software.

Figure 1. MG LABTECH’s multidetection microplate reader SPECTROstar Nano. Image credits: BMG Labtech.
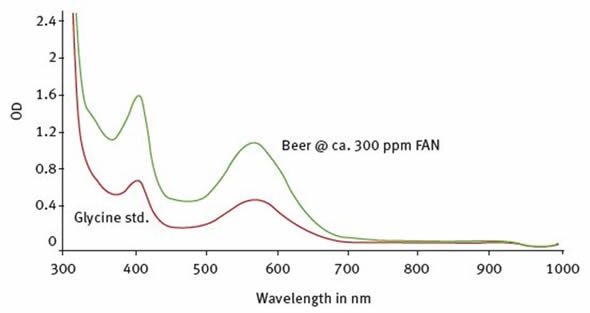
Figure 2. Spectra captured for a glycine standard (red line) and a beer sample (green line). Image credits: BMG Labtech.
Materials
The material used include the following:
- Clear 96-well plates from Greiner
- SPECTROstar Nano, BMG LABTECH, Ortenberg, Germany
Methods
Standard FAN Assay at pH 6.8
- Ninhydrin Color Reagent: 4g anhydrous Na2HPO4, 6g KH2PO4, 0.5g ninhydrin and 0.3g fructose are dissolved in 100 mL of distilled water. Stock can be stored refrigerated in an amber bottle for up to two weeks.
- Glycine Standard Stock: In order to form glycine standard stock, 107.2mg glycine is dissolved in distilled water and brought to 100mL exactly. To avoid mold growth, standard stock may be stored at 0 – 4°C.
- Glycine Standard Solution: 1mL glycine standard stock is diluted into a final volume of 100mL using distilled water. The standard contains 2mg amino-nitrogen/L. It should be freshly made for each daily assay run.
- Dilution solution: 2g of potassium iodate is dissolved in 600 mL distilled water and 400mL 96% ethanol is then added. With long term stability at 4°C, a close to room temperature should be used to prevent condensation forming on cuvettes.
- Assay: Dilution of 1mL of beer is performed to 50mL with distilled water and then 2 mL transferred to 16 X 150mm test tubes. 1mL of ninhydrin color reagent is added and the loosely covered tubes are heated for 16 minutes in a boiling water bath. To bring the temperature to 20°C (within 20 minutes) the tubes are moved to a cold water bath, and then 5mL of dilution reagent is added and mixed.
- The absorbance is then immediately recorded at 575nm against a blank containing 2mL water in place of the sample.
Microplate FAN Assay at pH 5.5
- Ninhydrin Stock Solution: 8g of ninhydrin is dissolved in 300mL of ethylene glycol and 100mL of 4N sodium acetate after which pH 5.5 buffer is added (*544g sodium acetate tetrahydrate + 400mL glacial acetic acid in 1L water). For at least six months, the stock is stable at room temperature
- Stannous Chloride Solution: 500mg SnCl2 is dissolved in 5mL ethylene glycol. The solution is stable at room temperature for a period of at least six months.
- Ninhydrin Reagent/Working Solution: Before the assay, 25µL of SnCl2 solution is added to every 1mL of ninhydrin stock solution and thoroughly mixed.
- Assay: 2µL (0.4µg N) of glycine standard or 2µL of beer or grape juice is introduced to a 96 well micro-well plate and 100µL of the pH 5.5 acetate buffered ninhydrin reagent is added. Heating of the plate is performed at 104°C for 10 minutes. For multiple samples, absorbance is recorded at the same time at 575nm in the SPECTROstar Nano microplate reader.
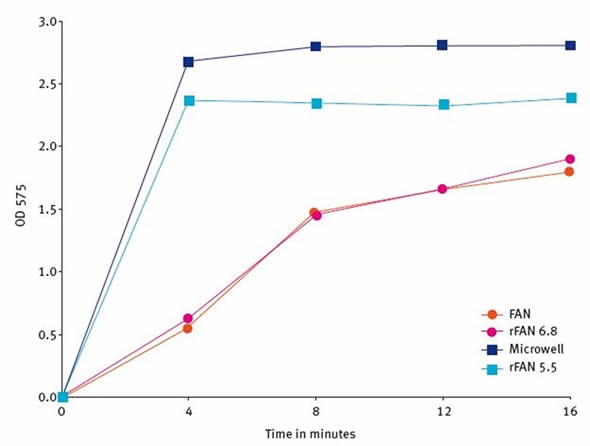
Figure 3. Reaction times required for completion of 4 different FAN assays. Image credits: BMG Labtech.
Reduced Volume Cuvette FAN Assay at pH 5.5
Assay: To 200 µl of the pH 5.5 acetate buffered ninhydrin reagent, beer or grape juice (30 µL) or 20 µL (4 µg N) glycine standard is added and placed for 10 minutes in a boiling water bath. The samples are taken out after 10 minutes and 2.8mL cold water is added. The tubes are then vortexed and the absorbance at 575nm is recorded in the SPECTROstar Nano against a blank containing 30µL water in place of the sample.
Controls and Calculations
For each sample, water blanks and the glycine standard, tests should be performed in duplicate or triplicate. From each sample and the standard glycine, the average blank value is subtracted. To obtain the FAN content in ppm, the blank corrected sample net (average) absorbance is divided by the net absorbance of the standard glycine and multiplied by 2 and the dilution factor (50 for beer diluted 1mL into 50mL or 100 for beer wort diluted 1mL into 100mL).
Results and Discussion
Optimization of Wavelength
The optimal wavelength for measuring this assay can be acquired by SPECTROstar Nano spectrometer’s scanning capabilities. Either with a beer or glycine sample (Figure 2), spectra were acquired from the ninhydrin reaction. The spectra obtained using the SPECTROstar Nano showed that there is a clear absorbance maximum at around 570 to 575nm.
Reaction Times Required for FAN Assay Completion
Comparison of the reaction time of four distinct ninhydrin-based FAN assays was performed. In addition to the three assays already explained (standard FAN pH 6.8, microplate FAN pH 5.5 and reduced volume rFAN cuvette pH 5.5), another reduced cuvette FAN assay at pH of 6.8 was studied. Figure 3 shows the result. It emerged that reaction rates differ between the assays. The standard FAN and rFAN pH 6.8 reactions needed at least 16 minutes to reach, at best, maximum absorbance. The rFAN pH 5.5 and the microplate FAN assay attained stable plateaus and maximal values within 8 minutes. When incubation was performed for up to 20 minutes, there was no absorbance loss in any of the assays.
Conclusion
This experiment showed that the SPECTROstar Nano is highly suitable for measuring the FAN microplate and cuvette assays for beer samples. There is no need for an extended cooling time or dilution/quench solution. There is also no need to run dilutions on beer/wort samples before sampling. Compared to the 40 to 50 year old standard FAN cuvette assay, these are all clear benefits. The assays were further optimized in terms of buffers and pH and should be beneficial to a number of brewing, distilling, enology and food laboratories for routinely determining free or total useable nitrogen.
References
- Lie, S. (1973) The EBC-Ninhydrin Method for Determination of Free Alpha Amino Nitrogen. J. Inst. Brew. 79, 37-41.
- Abernathy, D.G., Spedding, G. and Starcher, B. (2009) Analysis of Protein and Total Usable Nitrogen in Beer and Wine Using a Microwell Ninhydrin Assay. J. Inst. Brew. 115(2), 122-127.
Acknowledgement
Produced from articles authored by Gary Spedding and Nathan R. Harrison, Brewing and Distilling Analytical Services; Franka Ganske and EJ Dell, BMG LABTECH.
About BMG Labtech

BMG LABTECH has been committed to producing microplate readers for more than twenty years. By focusing on the needs of the scientific community, the company’s innovative microplate readers have earned the company the reputation of being a technology leader in the field.
BMG LABTECH has developed a wide range of dedicated and multi-mode microplate readers for life sciences applications and high-throughput screening.
All BMG LABTECH microplate readers are "Made in Germany" and are conceived, developed, assembled, and tested entirely at our headquarters in Germany.
Since our establishment in Offenburg, Germany in 1989, BMG LABTECH has expanded to offer a worldwide sales and support network with offices in the USA, UK, Australia, Japan and France. Our subsidiaries, regional offices and distributors are committed to bringing you innovative microplate reader technology with the quality and reliability you expect from a German company.
Our staff includes engineers and scientists from the fields of biology, biochemistry, analytical chemistry, and physics.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.