Automatic testing of vast numbers of chemical and/or biological compounds searching for a specific biological target is part of a drug discovery process known as high-throughput screening (HTS).
Such methods are widely utilized in the pharmaceutical industry and makes use of robotics and automation to test the biological or biochemical activity of a number of molecules - potential drugs – very quickly.
Large-scale compound libraries can be quickly screened cost-effectively, thus speeding up target analysis. High-throughput screening is a valuable tool for evaluating pharmacological targets, pharmacologically profiling agonists and antagonists for receptors (such as GPCRs) and enzymes, for example.
High-Throughput Screening in Drug Discovery
The primary objective of high-throughput screening is to identify candidates that affect the target in an anticipated way, so-called “hits” or “leads” by screening compound libraries. This is accomplished using liquid handling devices, robotics, plate readers as detectors, and devoted software for instrumentation control and data processing.
High-throughput screening is not usually utilized to identify drugs: many properties crucial to the advancement of new drugs cannot be evaluated by compound library screening, such as toxicity and bioavailability. Instead, the primary role of high-throughput screening assays is to recognize “leads” and offer suggestions for their optimization.
High-throughput screening assay results provide a starting point from which additional stages in the drug discovery pipeline such as drug design can be followed, and allow scientists to understand the interaction or role of a particular biochemical process.
High-throughput screening should, therefore, be considered a rapid examination of biological processes, through which compounds with a poor or no effect can be quickly excluded from the analysis pipeline.

PHERAstar FSX plate reader integrated in a Labcyte Access lab workstation at Cancer Research UK, Manchester Institute. Image credits: BMG Labtech.
High-Throughput Screening Services: Pharma, CROs and Universities
Modern advances in robotics, liquid handling and plate reader detection, as well as high-speed computers have allowed high-throughput screening to come to the fore. To run efficiently, high-throughput screening still necessitates a highly focussed and costly screening facility not afforded by every lab.
Instead of setting up their own facility, institutions with restricted budgets regularly access high-throughput screening services via third party providers like Contract Research Organisations (CRO) or, especially in academia, core facilities.
PHERAstar FSX supports the diverse and changing needs of CROs
The High-Throughput Screening Process: Basic Steps
From a technical point of view, the key points in high-throughput screening are miniaturization, automation and assay readout. Generally, high-throughput screening involves the following stages:
- Preparation of samples and compound libraries
- Formation of a method suitable for automation
- Formation of a robotic workstation
- Gaining and handling of data
Preparation of Samples and Compound Libraries
Samples are normally cellular or biochemical in nature, depending on the assay required. High-throughput screening demands that samples are prepared in an arrayed format - the key platform being the microplate.
Typical formats include 384-, 1536-, or 3456-well plates. Which microplate format and color are chosen is likely to be dictated by the nature of the sample and of the detection assay. Screening facilities keep compound library collections stored in so-called “stock plates”. These are not directly used in experiments, but “copied” to an assay plate through a pipetting station when needed.
Establishment of a Method Suitable for Automation
A key element of high-throughput screening is automation, as the conversion of a benchtop to an automated high-throughput screening assay imposes specific constraints which affect the practical assay design.
Preferably, a high-throughput screening assay is done in a single well, with low amounts of reagents (miniaturization), and minimal or no additional handling other than the injection of the sample/compound under examination. Consequently, the choice of the optimal detection mode and assay has to be subordinated to automation issues.
Major issues when setting up an automated screen are establishing a stringent assay and an effective quality control method. The clearer the distinction between negative and positive controls, the lower it is to produce false negatives and especially false positives.
Configuration of a Robotic Workstation
The robotic platform is designed to independently manage multiple plates simultaneously, hastening data acquisition significantly. Robotic platforms for high-throughput screening vary from simple automated liquid handling machines to multidimensional workstations executing multiple functions with the support of one or more mechanical arms. A robotic system usually manages microplates from station to station for several stages such as reagent addition, mixing, incubation, and detection.

CLARIOstar integrated in an automated biosynthetic production pipeline (Hamilton workstation), aimed at the production of fine and speciality chemicals from bacterial hosts at SYNBIOCHEM. Image credits: BMG Labtech.
Acquisition and Handling of Data
Data acquisition is typically carried out using an optical measurement which enumerates the amount of light “produced” by the sample. Different readouts such as fluorescent or luminescent detection, colorimetry, or light scatter (turbidity) are offered. Common detection modes include fluorescence intensity and polarization, FRET, time-resolved fluorescence (e.g.: HTRF, LANCE, etc), luminescence (e.g.: NanoBRET), and AlphaScreen.
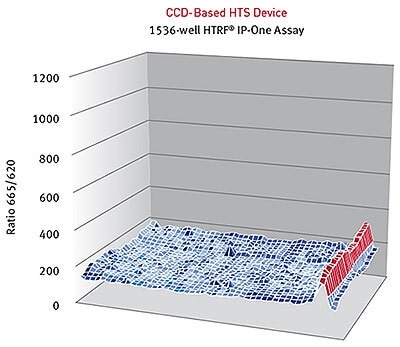
HTRF® ratios obtained for the IP-One assay with a CCD-based high-throughput screening Device. Image credits: BMG Labtech.
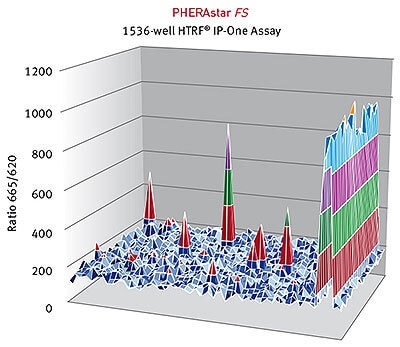
HTRF® ratios obtained for the IP-One assay with the PHERAstar FS. Image credits: BMG Labtech.
The light-based detection mode that is chosen for readout depends on the biological question required, data quality and cost-effectiveness. Specific instrumentation, such as multi-mode microplate readers, can consecutively carry out different tests or apply different detection procedures on the wells. The output is a grid of numeric values.

4 PHERAstar FSX plate readers integrated in a HighRes Biosolutions modular robotic workstation at AstraZeneca UK. Image credits: BMG Labtech.
In a single day, one HTS-dedicated plate reader can measure hundreds of plates, producing a vast amount of data points. Data management is therefore critical to automated HTS given a large number of compounds tested, the diversity of chemical libraries and the requirement to correlate and compare results from different screening operations.
Devoted data analysis and management platforms (e.g.: Genedata) entwine data obtained from different screening campaigns with compound structures, in addition to completed assays to each other, enabling the extraction of detailed information from different perspectives.
PHERAstar FSX: The Dedicated High-Throughput Screening Plate Reader
Compound library screenings must be measured quickly, in low volumes (a few µL) and yield strong results. Sensitive and precise instrumentation is required in addition to ease of use and the flexibility to perform different assays.
HTS on the PHERAstar
The PHERAstar FSX, through its past generations, has been considered the gold standard reference reader for high-throughput screening for the following reason:
- Highest sensitivity for fluorescence intensity and polarization on the market
- Option to measure in 384-, 1536- as well as 3456-well plate formats
- Simultaneous Dual Emission detection for quick and robust detection of fluorescence polarization assays, BRET, FRET and TR-FRET as well as AlphaScreen assays
- Devoted AlphaScreen/AlphaLISA excitation laser
- High-frequency TRF laser for efficient measurement of 1536-well plates on the fly (1 flash) in just 36 seconds
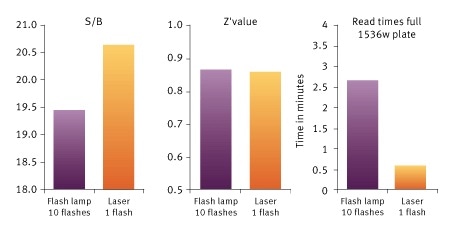
Signal to Blank (S/B), Z´value and read times for a Histone-BRD4 interaction assay measured on the PHERAstar FSX. The use of the TRF laser reduces read times, increases S/B, while keeping a very high Z´value. Image credits: BMG Labtech.
High-Throughput Screening Applications
G-Protein Coupled Receptors (GPCRs) are one of the most targeted groups of receptors in drug screening, but not all benchtop assays can be converted to an automated HTS, especially when using live cells. BMG Labtech’s Application Note 287 describes how NanoBRET was effectively employed to monitor ligand-GPCRs binding in living cells for the first time.
Miniaturization is a significant problem in HTS: samples must be as small as possible in order to reduce costs, while still delivering consistent results. The sensitivity of the microplate reader and its capability to consistently measure samples in high-density plate formats is of great importance.
In Application Note 229, DiscovereX PathHunter reporter cell lines were measured with the PHERAstar FS in both 1536- and 3456-well plates. The reader perceived a signal emanating from as few as 1,000 cells in a 1,536- and 250 cells in a 3,456-well plate. Application Note 260 illustrates how an AlphaLISA assay was miniaturized down to 2 µL per sample in a 1536-well plate for detection on the PHERAstar FS.
In drug screening, speed is as vital as miniaturization. Application Note 295 explains how the PHERAstar FSX equipped with the TRF excitation laser, can detect HTRF assays in 1536-well plates “on the fly” in 36 seconds, delivering a Z` value > 0.8.
About BMG Labtech

BMG LABTECH has been committed to producing microplate readers for more than twenty years. By focusing on the needs of the scientific community, the company’s innovative microplate readers have earned the company the reputation of being a technology leader in the field.
BMG LABTECH has developed a wide range of dedicated and multi-mode microplate readers for life sciences applications and high-throughput screening.
All BMG LABTECH microplate readers are "Made in Germany" and are conceived, developed, assembled, and tested entirely at our headquarters in Germany.
Since our establishment in Offenburg, Germany in 1989, BMG LABTECH has expanded to offer a worldwide sales and support network with offices in the USA, UK, Australia, Japan and France. Our subsidiaries, regional offices and distributors are committed to bringing you innovative microplate reader technology with the quality and reliability you expect from a German company.
Our staff includes engineers and scientists from the fields of biology, biochemistry, analytical chemistry, and physics.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.