As the most frequently altered gene in human cancers, p53 is possibly the most important gene in cancer biology. Indeed, the gene for p53 is damaged or missing in at least half of all cancers. Yet, despite its relevance to the field, we are still understanding how p53 functions, and recent research - with the help of nuclear magnetic resonance (NMR) spectroscopy - demonstrates how it continues to surprise us.
The Guardian of the Genome
p53 is a tumor suppressor gene, meaning that its normal function is in preventing the development of cancer. It is a key player in the p53 network, a molecular cascade within a cell that ultimately leads to cell death, or apoptosis. The effective working of this process is incredibly important within the body where the number of cells is tightly controlled by the regulation of cell division and cell death. Cancer ensues when these two processes become imbalanced, resulting in overproliferation.
It was previously thought that the p53 protein worked only in the nucleus. It is a transcription factor, meaning that it activates other genes. The proteins produced by these genes are then involved in signaling to the cell to undergo apoptosis.
However, more recently, research has shown that p53’s role extends outside the nucleus and into the cytosol where it has a more direct role in initiating apoptosis.
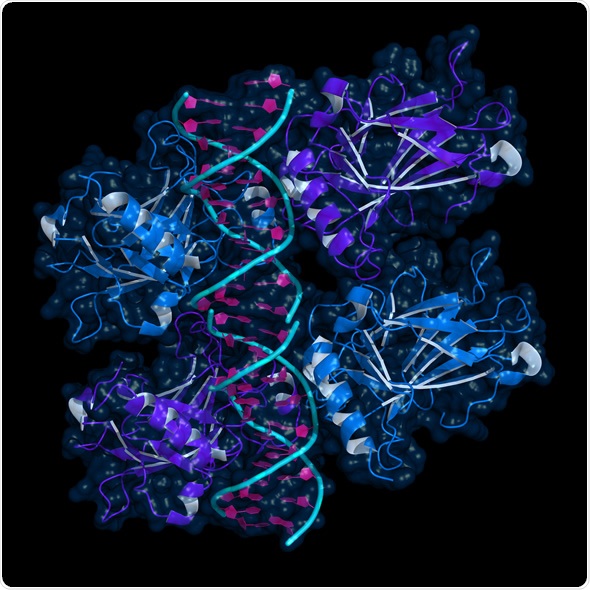
Ribbon model of p53 protein bound to DNA molecule. p53 (aka tumor protein 53) is a transcription factor whose inactivation can trigger the onset of cancer. Image Copyright: petarg, Image ID: 254933110 via shutterstock.com
Stepping into the cytosol
Around 10 years ago, research began to show that p53 was capable of inducing apoptosis even in cells that had no nucleus or in cells where transcription was blocked. These findings clearly indicated a role for p53 in apoptosis, independent of its effects on gene transcription. Then in 2004, a team of US researchers, led by Douglas Green, showed that p53 could directly trigger apoptosis in the cytosol by activating a protein called Bax.
However, it was not until this year that the team were able to create a molecular model, using NMR, explaining the mechanism by which p53 could do this. The findings could have the potential to lead to new types of therapies for cancer.
Observing a bond
First, the researchers found out that it was a particular amino acid within the p53 protein that allowed it to interact with BAX. They added BAX to radioactively labeled fragments of p53. The NMR spectra during this process showed “satellite” peaks for the resonance of a particular proline amino acid within p53 and revealed that it was undergoing a process called cis-trans isomerization. This is where the amino acid converts between its two structural forms: from the trans isomer, its usual state, to the cis isomer, which became much more common when BAX was added to p53.
Furthermore, the researchers were able to demonstrate specifically how this conformational change in a single amino acid led p53 to bind to the BAX protein.
They separately added fragments of two parts of the p53 protein – its structured region and its unstructured region – to radioactively labeled BAX. The NMR spectra showed that this created chemical shift perturbations (CSPs) in distinct regions of the BAX protein. When they repeated this with a complete p53 protein, all these CSPs combined were observed. This showed that the structured and unstructured regions of p53 bind independently to BAX.
Then, by titrating p53 fragments into radioactively labeled BAX, the researchers were able to observe the kinetics of this binding which confirmed that it arose through the conformational change of proline from its trans to its cis isomer.
Using the NMR data, they were then able to create a molecular model showing how the entire process takes place. Interestingly, this process is distinct from the way BAX is usually activated in the apoptotic pathway, so this research revealed not only a new dimension to how p53 works, but also a previously unknown way that BAX could be activated.
The researchers say that the finding could be used to reactivate the p53 apoptotic pathway that becomes inactivated in so many cancers, such as by creating molecules that could reactivate this newly discovered BAX-dependent apoptosis or reactivate the cis form of proline within p53.
Facilitating discovery with Bruker Avance
To conduct their research, Follis et al used a Bruker Avance spectrometer coupled to cryogenically cooled triple resonance probes. The latest Bruker Avance III HD delivers an unprecedented level of digital control, NMR speed and the most advanced RF generation in the industry.
 Nanobay 400 HD with BCU SmartCooler™, Prodigy BBO CryoProbe, SampleXpress™ and Ascend 400 MHz NMR magnet.
Nanobay 400 HD with BCU SmartCooler™, Prodigy BBO CryoProbe, SampleXpress™ and Ascend 400 MHz NMR magnet.
Meanwhile, cryoprobes have allowed one of the largest increases in NMR sensitivity over recent decades, meaning scientists can use sample amounts that even recently would have been considered too small.
Bruker offers two lines of cryogenic probe technologies: CryoProbe, which is based on a closed cycle helium cryocooler and CryoProbe Prodigy which is an open cycle liquid nitrogen cooling system. Both use a range of automated features including cool-down, warm-up and timer, and are compatible with Bruker’s automatic sample changer.
References:
- Bradley D. (2015) NMR reveals cancer clue: p53 activity. Available at: http://www.spectroscopynow.com/nmr/details/ezine/14f25d494fb/NMR-reveals-cancer-clue-p53-activity.html. Date accessed: 06 September 2015.
- Chipuk JE, et al. Direct activation of Bax by p53 mediates mitochondrial membrane permeabilization and apoptosis. Science 2004; 303: 1010-1014.
- Chipuk JE, et al. Pharmacological activation of p53 elicits Bax-dependent apoptosis in the absence of transcription. Cancer Cell 2003; 4: 371-381.
- Follis AV, et al. The DNA-binding domain mediates both nuclear and cytosolic functions of p53. Nature Structural & Molecular Biology 2014; 21: 535-543.
- Follis AV, et al. Pin1-Induced Proline Isomerization in Cytosolic p53 Mediates BAX Activation and Apoptosis. Molecular Cell 2015; 59: 677-684.
- Vogelstein B (2010). p53: The most frequently altered gene in human cancers. Available at: http://www.nature.com/scitable/topicpage/p53-the-most-frequently-altered-gene-in-14192717. Date accessed: 6th September 2015.
About Bruker BioSpin - NMR, EPR and Imaging

Bruker BioSpin offers the world's most comprehensive range of NMR and EPR spectroscopy and preclinical research tools. Bruker BioSpin develops, manufactures and supplies technology to research establishments, commercial enterprises and multi-national corporations across countless industries and fields of expertise.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.