Organ transplantation has transformed the lives of many patients with kidney disease, lung disease and heart disease. However, transplantation still requires immunosuppressive medications to prevent attack from the immune system of the recipient.
In addition to increasing the risk of infection, these can cause side effects, such as loss of appetite, nausea, vomiting. Furthermore, there are insufficient organs donated to allow all those in need of a transplant to receive one1.
In some diseases, it is not necessary for the recipient to receive an entire organ in order for the missing functionality to be restored. It therefore followed that it may be possible to help more patients with the available donated organs.
With improved biotechnologies, it became possible to obtain purified cells from donated tissue and cell transplantation became a reality. This brought with it the potential to harvest the cells for transplantation from the patient in order to circumvent immunological rejection of the transplanted cells.
Transplantation of haematopoietic stem cells, usually derived from bone marrow, peripheral blood, or umbilical cord blood, is commonly used in the treatment of blood diseases, including cancers such as leukaemia and multiple myeloma1. It can also be used to replenish cells destroyed during chemotherapy.
Another emerging area in which cell transplantation is being used is the treatment of certain patients with type 1 (insulin-dependent) diabetes.
Pancreatic islet transplantation
Patients with type 1 diabetes are unable to produce insulin. This renders them unable to maintain levels of glucose in their blood within the healthy range. Untreated diabetes results in dangerously high blood sugar levels that can cause a range of life-threatening conditions including kidney failure and cardiovascular disease.
© GodoiJE/Shutterstock.com
Type 1 diabetes is treated with regular insulin injections, which effectively prevent elevated glucose levels. However, the quantity of insulin injected needs to be balanced with the amount of carbohydrate ingested.
If sufficient carbohydrate is not eaten after insulin has been administered, glucose levels fall below the normal range (hypoglycaemia) and there is not adequate energy available for the body to function normally.
The brain is particularly energy hungry and lack of adequate glucose can result in a diabetic coma, which can be fatal if not treated. Usually, the individual experiences warning signs, such as headache, tiredness, confusion, that their blood sugar levels have become too low and can take in more glucose to remedy the situation.
However, some patients with type 1 diabetes have hypoglycaemia unawareness and cannot feel the onset of symptoms of hypoglycaemia.
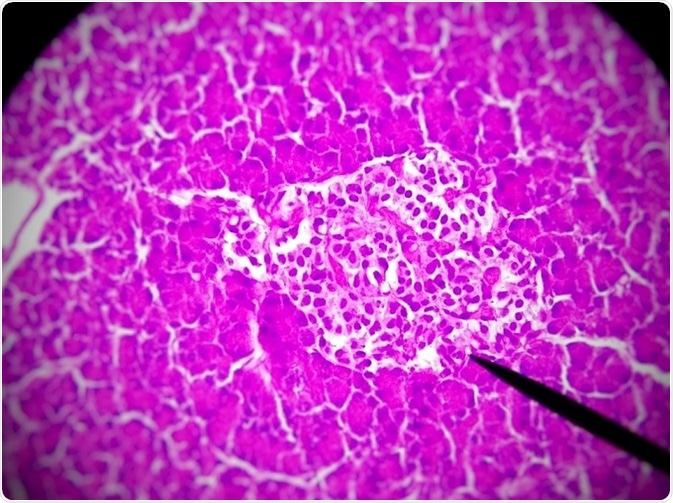
An experimental treatment being used to help these individuals is pancreatic islet transplantation2. Pancreatic islets, also called islets of Langerhans, are small clusters of cells within the pancreas.
Cells of the pancreatic islets are responsible for producing insulin when levels of glucose in the blood rise, eg, after eating. It is these cells that do not function in type 1 diabetes.
The patient typically receives an infusion of 400,000 to 500,000 purified islets extracted from a donated pancreas. The cells of the transplanted islet will then produce and release insulin into the bloodstream of the recipient.
Pancreatic islet transplantation has been shown to result in control of blood sugar levels, or to at least prevent hypoglycaemia unawareness2. However, the autoimmune response that destroyed the transplant recipient's own islets in the first place to cause diabetes may reoccur and destroy the transplanted islets.
Consequently, ongoing monitoring of the transplanted cells is essential to ensure that glucose levels continue to be adequately controlled.
Monitoring outcomes after pancreatic islet transplantation
The monitoring of transplanted pancreatic islets in vivo is facilitated by in vitro labelling of the cells before they are introduced to the recipient3.
The type of label used depends on the imaging technique to be used, for example, a radionuclide would be used for positron emission tomography (PET), whereas a paramagnetic core is needed for 1H magnetic resonance imaging (MRI).
Since a single imaging technique typically does not provide the necessary specificity, sensitivity and spatial resolution, a combination of labels is often used.
The 19fluorine (19F) label, incorporated by endocytosis, has successfully been used in the quantification of transplanted stem cells by MRI4. However, the structure of pancreatic islets makes them particularly difficult to label without reducing their function3.
Researchers have recently modified a technique typically used for gene delivery into stem cells—microporation— to achieve improved labelling outcomes for pancreatic islets5. Pancreatic islet cells were labelled via modified microporation with bimodal positively charged poly(lactic-coglycolic acid) nanoparticles with encapsulated perfluoro crown ethers and indocyanine green dye.
The success of the labelling procedure was demonstrated by nuclear magnetic resonance imaging with a Bruker Avance III 400 MHz spectrometer and compared with the passive endocytosis methodology. Imaging of the pancreatic islet was performed using a 4.7 T Bruker BioSpin imager.
The modified microporation labelling methodology was shown to be a safer way to label pancreatic islet cells than the standard microporation procedure5. It was also fast, but the efficiency achieved was lower than with endocytosis. This novel methodology may thus be a suitable procedure for labelling when it is necessary to obtain results quickly.
References
- Watt FM, Driskell RR. The therapeutic potential of stem cells. Philos Trans R Soc Lond Ser B Biol Sci. 2010;365:155–63.
- Ichii H, Ricordi C. Current status of islet cell transplantation. J Hepato-Biliary-Pancreat Surg. 2009;16:101–12.
- Shapiro AMJ. Islet Transplantation in Type 1 Diabetes: Ongoing Challenges, Refined Procedures, and Long-Term Outcome. Rev Diabet Stud. 2012;9(4): 385–406.
- Gaudet JM, et al. Tracking the fate of stem cell implants with fluorine-19 MRI. PLoS One. 2015;10:e0118544.
- Herynek V, et al. Pre-Microporation Improves Outcome of Pancreatic Islet Labelling for Optical and 19F MR Imaging. Biological Procedures Online 2017;19:6. doi: 10.1186/s12575-017-0055-4
About Bruker BioSpin - NMR, EPR and Imaging

Bruker BioSpin offers the world's most comprehensive range of NMR and EPR spectroscopy and preclinical research tools. Bruker BioSpin develops, manufactures and supplies technology to research establishments, commercial enterprises and multi-national corporations across countless industries and fields of expertise.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.