The use of ion chromatography with amperometric detection for simple and reliable determination of lactulose and mannitol in urine is described in this article. Diagnosis of the leaky gut syndrome and associated chronic inflammatory bowel diseases can be achieved with the lactulose-mannitol test. Lactulose is a disaccharide and mannitol or mannite is a sugar alcohol of the monosaccharide mannose (Figure 1).
Figure 1. Chemical structure of mannitol and lactulose.
The Role of the Intestinal Mucosa
The main function of the intestinal mucosa is absorption of essential nutrients, other critical substances, and fluids and electrolytes from the intestinal cavity or lumen. Another vital function of an intact intestinal mucosa is protecting the blood stream from absorbing antigens like bacteria, heavy metals, and bacterial endotoxins, as well as luminal toxins.
The highly sensitive lactulose-mannitol test on the urine can help detect leaky gut syndrome or impaired intestinal permeability and other diseases related to it. This test is performed by oral administration of lactulose and mannitol. Since human body does not use these substances, they remain unchanged and are excreted in the urine.
The transcellular absorption of smaller molecule mannitol via the intestinal epithelial cells overcomes the barrier, whereas the larger disaccharide molecule lactulose could pass through the barrier paracellularly through the gaps present between the cells.
With an intact intestinal mucosa and a normal intestinal permeability, paracellular transport is prevented and controlled by the close cell-to-cell connections or tight junctions, which, in turn, results in minute quantities of lactulose absorption and detection in urine.
Increased Permeability due to Damaged Intestinal Mucosa
The intestinal mucosa can be damaged due to immunological disorders, genetic disposition, psychological factors and environmental influences, such as virus, nicotine, bacteria and food, resulting in loss of its barrier function. Consequently, intestinal permeability increases due to loss of the sealing capability of the tight junctions.
As a result, the absorption of macromolecules and luminal antigens increases as they are allowed to freely pass through the intestinal mucosa, which, in turn, activates the mucosal immune system, and causes food allergies and intestinal inflammation. Celiac disease, Crohn’s disease and ulcerative colitis are some of the other health disorders caused by the loss of the barrier function of the intestinal mucosa. However, it has not been established clearly whether the loss of barrier function is the reason behind or the consequence of these diseases.
Detection of Increased Intestinal Permeability via Urine Analysis
Paracellular transport of larger lactulose molecules from the intestinal lumen to plasma and finally into the urine is fostered due to an increase in intestinal permeability because of impaired intestinal mucosa. As a result, the urine is detected to have higher concentrations of these molecules. However, there is a dip in the functional capacity and the number of the intestinal epithelial cells, which facilitate transcellular movement of the smaller mannitol molecule.
Consequently, the urine is detected to have lower concentrations of mannitol molecules. After oral administration of a mixture of mannitol and lactulose, the two decisive factors in the diagnosis of increased intestinal permeability are rate of recovery of these sugars from the urine sample and the ratio of lactulose to mannitol.
Urine Analysis Using Ion Chromatography
A number of analytical techniques are suggested in scientific publications for quantitative analysis of sugars and sugar alcohols in urine, but most of the methods have their own limitations. For instance, techniques, such as enzymatic determination, colorimetry and chromatography have less specificity and time-intensive.
Other techniques like gas chromatography, including GC-MS, involve elaborate derivatization steps. In comparison with these techniques, ion chromatography with pulsed amperometric detection (IC-PAD) enables easy and more accurate determination of even trace amounts of sugar. Figure 2 depicts the results of using an ion chromatograph with pulsed amperometric detection for determining trace level concentrations of lactulose and mannitol in urine.
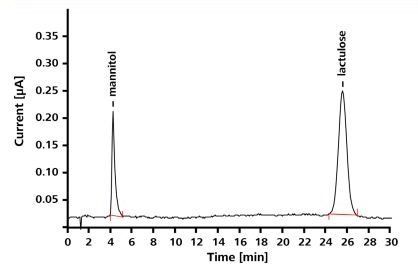
Figure 2. Chromatogram of a standard solution of mannitol and lactulose (1mg/L mannitol, 5mg/L lactulose); column: Hamilton RCX-30 - 250/4.6; eluent: 50mmol/L NaOH, 1.5mL/min; column temperature: 30°C; detector: PAD mode, WE: Au, RE: Pd, working potential: 0.05V, temperature: 30°C; sample volume: 20µL.
Optimization of the chromatographic parameters for separation was performed on the RCX-30 column, the stationary phase of which comprised of a styrenedivinyl benzene copolymer made up of quaternary ammonium groups.
The Metrosep Carb 1 - 150/4.0 column is another option for carrying out separation. The mobile phase consists of NaOH eluent. The readings obtained with it and the linearity, accuracy and selectivity of the method were studied.
It was observed that the calibration curves were linear and the correlation coefficient ranges between 0.99990 and 0.99995 for the concentration range of 0.1-10mg/L. The sample was spiked with known concentration of lactulose-mannitol standard for validating the accuracy of the method (Figure 3). The range of recovery rates was found to be 95-104%.
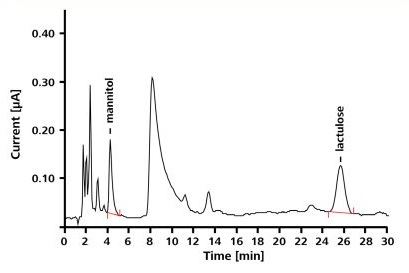
Figure 3. Chromatogram of a urine sample spiked with 1.5 mg/L mannitol and 2.5 mg/L lactulose; separation conditions correspond to those indicated in Figure 2.
Conclusion
Accurate and fast determination of mannitol and lactulose in urine samples is possible through the ion chromatographic method equipped with amperometric detection. Intestinal permeability can be effectively characterized using this diagnostic tool.
Acknowledgements
Produced from materials authored by Benedikt Galliker and Alfred Steinbach from Metrohm International Headquarters, Herisau, Switzerland.
About Metrohm
At Metrohm is one of the world’s most trusted manufacturers of high-precision instruments for chemical analysis. Metrohm was founded in 1943 by engineer Bertold Suhner in Herisau, Switzerland. Today, Metrohm is represented in 120 countries by subsidiaries and exclusive distributors. The global Metrohm Group also includes the Dutch companies Metrohm Applikon and Metrohm Autolab, manufacturers of online analyzers and instruments for electrochemical research, respectively. Recently, the Metrohm Group was joined by Metrohm Raman, a leading manufacturer of handheld Raman spectrometers.
Metrohm is the global market leader in analytical instruments for titration. Instruments for ion chromatography, voltammetry, conductivity, and stability measurement make the Metrohm portfolio for ion analysis complete. Instruments for Near-infrared and Raman spectroscopy are another, strongly growing segment of the Metrohm portfolio.
Metrohm is a problem solver, both in the laboratory and within the industrial process. To this end, the company offers their customers complete solutions, including dedicated analytical instrumentation as well as comprehensive application know-how. More than 30% of the company’s employees at the Metrohm international headquarters in Herisau work in R&D.
Metrohm has been owned 100% by the non-profit Metrohm Foundation since 1982. The Metrohm Foundation, which does not exert any influence on the company’s business operations, sponsors gifted students in the natural sciences, supports charitable and philanthropic purposes and, above all, ensures the independence of the company.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.