Herbal supplements have been recently included in the International Conference on Drug Regulatory Authorities (ICDRA). The World Health organization (WHO) introduced Traditional Medicine Program (TMP) to integrate traditional medicine into national health care systems. Similar to the synthetic medicines, the herbal medicines were governed by strict regulations according to the TMP guidelines for the assessment of quality, safety, and efficacy as well as important requirements for labelling and the package insert for consumers' information [1].
Application of Vis-NIRS to Determine the Concentration of Baicalin
Baicalin, isolated from the roots of the species Scutellaria baicalensis, is one of the most widely used Chinese herbal extracts in Eastern and Western medicines. This compound has shown to possess anti-inflammatory, anti-viral, and anti-tumor properties [2]. High performance liquid chromatography (HPLC) is used to estimate the Baicalin concentration in scutellaria baicalensis powder.
However, this analytical technique requires extensive sample preparation and well-trained operator, resulting in relatively high costs for routine analysis. Visible near Infrared spectroscopy (Vis-NIRS) is an excellent and faster alternative to HPLC to determine the concentration of Baicalin.
Experimental Set Up and Validation
To investigate the validity of using Vis-NIRS as an alternative to HPLC, 132 powder samples of Scutellaria baicalensis, with varying concentrations of Baicalin ranging from 11.54– 15.40% were evaluated using NIRS DS2500 Analyzer over the full wavelength range (400–2500 nm).
The samples were filled in flat quartz glass vessels and analyzed in triplicates to determine the correlation between the obtained values and reference values was determined. Vision Air 2.0 software was used for acquisition and management of data, as well as for the development of the quantification method. To correct for spectral baseline shifts, partial Least Squares regression (PLS) was performed on 90 Scutellaria baicalensis powder samples using 2nd derivative as data pre-treatment.

Figure 1. The NIRS DS2500 Analyzer was used for spectral data acquisition over the full range from 400 nm to 2500 nm.
Table 1. Used equipment and software.
| Equipment |
Metrohm number |
| NIRS DS2500 |
2.922.0010 |
| NIRS transflection vessel, optically flat |
6.7401.000 |
| Vision Air 2.0 Complete |
6.6072.208 |
Results
At a wavelength region of 720–920 nm, the change in absorbance with the change in concentration change was obvious. To verify the performance of the derived quantitative models during method development, internal cross-validation and external validation on an independent data set of 42 samples was applied.
For the quantification of Baicalin in Scutellaria baicalensis powder, a 3-factor-model with a Standard Error of Calibration (SEC) of 0.35%, a Standard Error of Cross Validation (SECV) of 0.48% and a Standard Error of Prediction (SEP) of 0.53% was developed. The R2 = 0.75 value indicated high correlation between the provided reference values and the calculated values through Vis-NIRS.
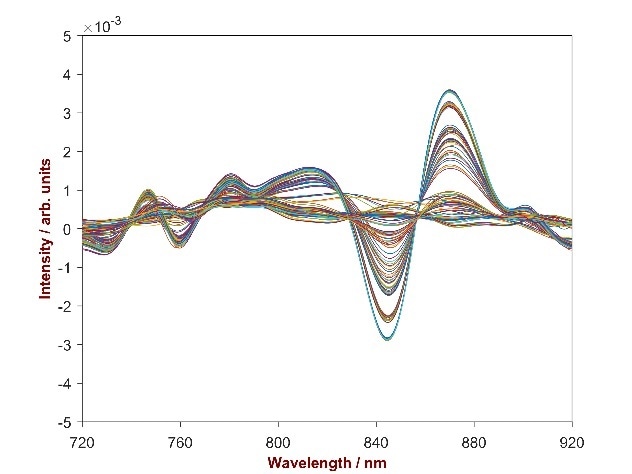
Figure 2. 2nd derivative spectra of 132 scutellaria baicalensis powder samples with the Baicalin concentration differing from 11.54–15.40%. Displayed is the wavelength region of 720–920 nm were the correlation between absorbance change and concentration change is obvious.
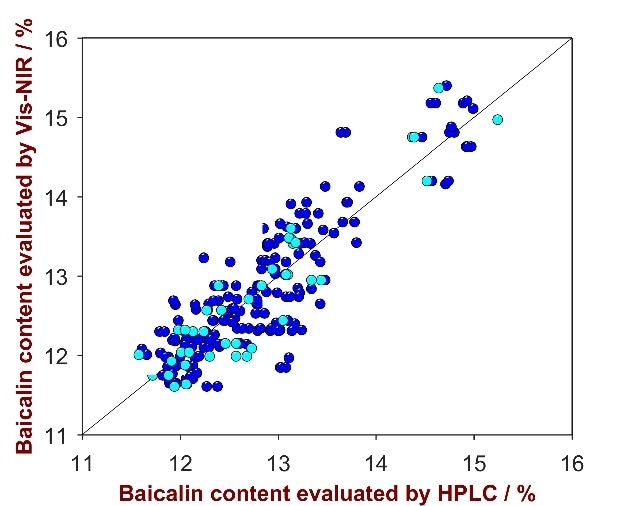
Figure 3. Correlation plot of the predicted Baicalin product strength by NIRS versus the HPLC reference values. Displayed are the calibration (blue) and validation data (turquois).
Table 2. Results of the quantitative method development for Baicalin content.
| . |
. |
| Regression model |
PLS with 3 factor |
| Pre-treatment |
2nd derivative |
| Wavelength range |
780–1080 nm
1120–2500 nm |
| R2 |
0.75 |
| SEC |
0.36% |
| SECV |
0.48% |
| SEP |
0.53% |
Conclusion
Taken together, Vis-NIRS can be conveniently used for the quantification of Baicalin content in a Scutellaria baicalensis powder. The standard errors obtained with Vis-NIRS were similar to the traditionally used HPLC method, underlining the suitability of Vis-NIR spectroscopy as a highly reliable and faster quality control method for the quantification of Baicalin. The use of Vis-NIRS for this purpose can save time and money as compared to the HPLC method.
References
- https://nccih.nih.gov/health/whatiscam/chinesemed.htm
- https://en.wikipedia.org/wiki/Baicalein
About Metrohm
At Metrohm is one of the world’s most trusted manufacturers of high-precision instruments for chemical analysis. Metrohm was founded in 1943 by engineer Bertold Suhner in Herisau, Switzerland. Today, Metrohm is represented in 120 countries by subsidiaries and exclusive distributors. The global Metrohm Group also includes the Dutch companies Metrohm Applikon and Metrohm Autolab, manufacturers of online analyzers and instruments for electrochemical research, respectively. Recently, the Metrohm Group was joined by Metrohm Raman, a leading manufacturer of handheld Raman spectrometers.
Metrohm is the global market leader in analytical instruments for titration. Instruments for ion chromatography, voltammetry, conductivity, and stability measurement make the Metrohm portfolio for ion analysis complete. Instruments for Near-infrared and Raman spectroscopy are another, strongly growing segment of the Metrohm portfolio.
Metrohm is a problem solver, both in the laboratory and within the industrial process. To this end, the company offers their customers complete solutions, including dedicated analytical instrumentation as well as comprehensive application know-how. More than 30% of the company’s employees at the Metrohm international headquarters in Herisau work in R&D.
Metrohm has been owned 100% by the non-profit Metrohm Foundation since 1982. The Metrohm Foundation, which does not exert any influence on the company’s business operations, sponsors gifted students in the natural sciences, supports charitable and philanthropic purposes and, above all, ensures the independence of the company.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.