The chemical compound bromate is formed when drinking water is disinfected through ozonation. At mg/L levels, bromate is potentially carcinogenic to mice and rats. Based on recent toxicological studies, the International Agency for Research on Cancer (IARC) has classified bromate as a group-2B carcinogen to humans, with concentrations greater than 0.05 g/L associated with kidney tumor risk. In view of this, the US Environmental Protection Agency (USEPA) has called for comments on setting bromate’s maximum contaminant level goal to zero.
Ultratrace determination of bromate in drinking water
In this study, the goal was to establish a method for routine determination of bromate in drinking water using IC with mass-spectrometric detection. The IC-ICP/MS technique was selected for its simplicity compared with other mass-spectrometric techniques. Using this technique, the presence of bromate in drinking water can be determined at concentrations in the sub-μg/L range. The sample does not have to be pre-treated and it takes 15 minutes to analyze a single sample.
Results
A technique was developed to determine ultra-trace levels of bromate in drinking water by IC-ICP/MS. In this method, the microbore column method is used in combination with a self-made high-performance and high-capacity anion-exchanger. Direct analysis of almost all water samples was possible without matrix elimination, owing to the high capacity of the separation column and an optimized elution system based on NH4NO3. In addition, large injection volumes can be used. Due to the sensitivity of ICP/MS detection, there is no need for trace enrichment, meaning the sample does not have to be pre-treated.
In the mineral and drinking waters analyzed, the method detection limits for bromate were in the range of 50–65 ng/L, corresponding to absolute detection limits of 44 to 58 pg. The sample composition influenced the retention of bromate, as well as the signal-to-background (SBR) and signal-to-noise (SNR) ratios. At a concentration of 500 ng/L bromate, the within-run imprecision of the presented IC-ICP/MS coupling was 5%.
As shown in Figure 1, complete analysis took 8 to 15 minutes, depending on the sample’s bromide content. Given the sensitivity, precision, sensitivity and analysis duration of the described IC-ICP/MS coupling, it is well suited for accurate routine analysis of bromate content in drinking water at sub-μg/L levels.
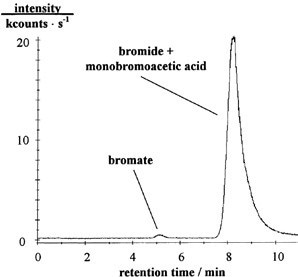
Figure 1. Separation of 1 μg/L bromate, 100 μg/L bromide, and 100 μg/L monobromoacetic acid. Sample volume: 585 μL; column: self-made; eluents: 100 or 60 mmol/L nitric acid or 100 mmol/L hydrochloric acid, pH adjusted to 6 with ammonia (25% w/w); m/z 79
Nowak, M.; Seubert, A. (1998) Anal. Chim. Acta 359, 193–204
Bromate – further applications with IC-ICP/MS
Comparison of ion chromatographic methods based on conductivity detection, post-column-reaction and on-line-coupling IC-ICP/MS for the determination of bromate Schminke, G.; Seubert, A. (2000) Fresenius J. Anal. Chem. 366(4), 387–391.
Trace analysis of bromate in drinking waters by means of online coupling IC-ICP/MS Seubert, A.; Nowak, M. (1998) Fresenius J. Anal. Chem. 360(7), 777–780.
Further applications with IC-ICP/MS
Sulfur
Measurement of sulphur isotope ratio (34S/32S) in uranium ore concentrates (yellow cakes) for origin assessment - Han, S:H.; Varga, Z.; Krajko, J.; Wallenius, M.; Song, K.; Mayer, K. (2013) J. Anal. At. Spetrom. 28(12), 1919–1925.
Aluminum
Characterization of an aluminium(III) citrate species by means of ion chromatography with inductively coupled plasma-atomic emission spectrometry detection Peukert, A.; Seubert, A. (2009) J. Chrom. A 1216(45), 7946–7949.
References
- Aschner, M.; Syversen, T. (2005) Methylmercury: Recent advances in the understanding of its neurotoxicity. Ther. Drug Monit. 27, 278–283.
- Bianchi, A.; Calabi, L.; Corana, F.; Fontana, S.; Losi, Maiocchi, A.; Paleari, L.; Valtancoli, B. (2000) Thermodynamic and structural properties of Gd(III) complexes with polyaminopolycarboxylic ligands: Basic compounds for the development of MRI contrast agents. Coord. Chem. Rev. 204, 309–393
- Burger, J.; Gochfeld, M. (2005) Heavy metals in commercial fish in New Jersey. Environ. Res. 99(3), 403–412
- Dietary Supplement Fact Sheet: Selenium. US National Institutes of Health; Office of Dietary Supplements. Retrieved Oct 11, 2016.
- Gochfeld, M.; Burger, J. (2005) Good fish/bad fish: A composite benefit–risk by dose curve. Neurotoxicology 26(4), 511–520
- Herrmann, T. Einsatz der On-line-Kopplung von Ionenchromatographie und ICP-MS zur Bestimmung von Anionen. Diploma thesis, Philipps-Universität Marburg, Germany, 2006
- Knöll, J. Ph.D. Ultratrace determination of aminopolycarboxylic acid based chelating agents using inverse on-line coupling of IC with ICP-MS. PhD thesis, Philipps-Universität Marburg, Germany, 2013
- Knöll, J.; Seubert, A. (2012) Indirect ultra trace determination of aminopolycarboxylic acids in surface water using ion exchange chromatography coupled on-line to inductively coupled plasma mass spectrometry. Journal of Chromatography A 1270, 219-224.
- Kümmerer, K.; Ed. Pharmaceuticals in the environment: Sources, fate, effects, and risks. Springer-Verlag Berlin Heidelberg, 2008; 3rd ed.
- Künnemeyer, J.; Terborg, L.; Meermann, B.; Brauckmann, C.; Möller, I.; Scheffer, A.; Karst, U. (2009) Speciation analysis of gadolinium chelates in hospital effluents and wastewater treatment plant sewage by a novel HILIC/ICP-MS method. Environ. Sci. Technol. 43(8), 2884–2890.
- Künnemeyer, J.; Terborg, L.; Nowak, S.; Telgmann, L.; Tokmak, F.; Krämer, B. K.; Günsel, A.; Wiesmüller, G. A.; Waldeck, J.; Bremer, C.; Karst, U. (2009) Analysis of the contrast agent Magnevist and its transmetalation products in blood plasma by capillary electrophoresis / electrospray ionization time-of-flight mass spectrometry. Anal. Chem. 81(9), 3600–3607
- Levenson, C. W.; Axelrad, D. M. (2006) Too much of a good thing? Update on fish consumption and mercury exposure Nutr. Rev. 64(3), 139–145.
- Montaser, A.; Golightly, D. W.; Eds. Inductively coupled plasmas in analytical atomic spectrometry. VCH Publishers, Inc.: New York, 1992.
- Rahman, G. M. M.; Martone, N.; 'Skip' Kingston, H. M. (2012) Determination of hexavalent chromium in NIST SRM 2701 by speciated isotope dilution mass spectrometry (EPA Method 6800) using IC-ICP-MS. In Handbook of hyphenated ICP-MS applications, 2nd edition; Agilent, 2012; pp 33–35. http://www.agilent.com/cs/library/applications/5990-9473EN_icpmsSpeciationHB_lr.pdf (accessed Oct 7, 2016)
- Saranko, C. J. et al. Fact Report for toxicity of arsenite and arsenate, Florida Dept. of Health, Nov 6, 1998
- Schedlbauer, O. F.; Heumann, K. G. (2000) Biomethylation of thallium by bacteria and first determination of biogenic dimethylthallium in the ocean. Appl. Organomet. Chem.14(6), 330-340
- Wilber, C. G. (1980). Toxicology of selenium: A review.Clin. Toxicol. 17 (2), 171–230.
About Metrohm
At Metrohm is one of the world’s most trusted manufacturers of high-precision instruments for chemical analysis. Metrohm was founded in 1943 by engineer Bertold Suhner in Herisau, Switzerland. Today, Metrohm is represented in 120 countries by subsidiaries and exclusive distributors. The global Metrohm Group also includes the Dutch companies Metrohm Applikon and Metrohm Autolab, manufacturers of online analyzers and instruments for electrochemical research, respectively. Recently, the Metrohm Group was joined by Metrohm Raman, a leading manufacturer of handheld Raman spectrometers.
Metrohm is the global market leader in analytical instruments for titration. Instruments for ion chromatography, voltammetry, conductivity, and stability measurement make the Metrohm portfolio for ion analysis complete. Instruments for Near-infrared and Raman spectroscopy are another, strongly growing segment of the Metrohm portfolio.
Metrohm is a problem solver, both in the laboratory and within the industrial process. To this end, the company offers their customers complete solutions, including dedicated analytical instrumentation as well as comprehensive application know-how. More than 30% of the company’s employees at the Metrohm international headquarters in Herisau work in R&D.
Metrohm has been owned 100% by the non-profit Metrohm Foundation since 1982. The Metrohm Foundation, which does not exert any influence on the company’s business operations, sponsors gifted students in the natural sciences, supports charitable and philanthropic purposes and, above all, ensures the independence of the company.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.