Alkylated mercury (CH3Hg+), inorganic Hg(II) and elemental Hg(0) are some of the various forms of the element mercury. Methylmercury is the most toxic of the most common mercury species existing in the environment. Methylmercury is categorized as a neurotoxin; it bio-accumulates quickly and even small amounts can lead to serious health issues or death. According to the US Food and Drug Administration (FDA), the consumption of contaminated fish is the major exposure pathway to methylmercury in humans and wildlife.

Image Credits: Alexey V Smirnov/shutterstock.com
In addition to fish, mercury can also be tested in end consumers. It can be tested for in human hair, for example, or in potential polluters such as crude oil. The US Environmental Protection Agency (EPA) has set a reference dose for methylmercury of 0.1 μg/kg of body weight per day and the World Health Organization (WHO) has set the dose at 1.6 μg/kg of body weight per week (Levenson, C. W., 2006).
Mercury and arsenic speciation test using IC-ICP/ MS
Specific isotope dilution mass spectrometry (SIDMS) is a technique used to analyse the species transformations of mercury that occur during several sample preparation methods. This paper explains the firm advantage that SIDMS (as detailed in Environmental Protection Agency [EPA] Method 6800) provides in the analysis of mercury species transformations that occur during the preparation of fish tissue samples. The use of SIDMS is anticipated to rise, given the unique features and advantages of the EPA Method 6800. This tool is valuable for validating and optimizing trace-metals-speciated sample preparation and it is expected to become widely accepted by analytical chemists.
Results
It took less than five minutes to achieve separation and the retention times were 1.87 ± 0.02 and 2.98 ± 0.03 minutes, as shown in Figure 1. The linear calibration curves for CH3Hg+ and Hg(II) were achieved in the 1 to 20 μ g/L range. Detection limits for Hg(II) and CH3Hg+ were 0.46 ± 0.02 and 0.78 ± 0.08 μg/L, respectively.
Non-quantitative recoveries and species transformations observed during extraction can be overcome using the SIDMS tool. SIDMS enables mercury species to be determined with high accuracy, precision and correction for species transformations. Inorganic mercury contamination in the extracting reagent could be observed, depending on the extraction method used, although alkaline extraction methods produced results that agreed well with the 95% confidence levels of the certified values.
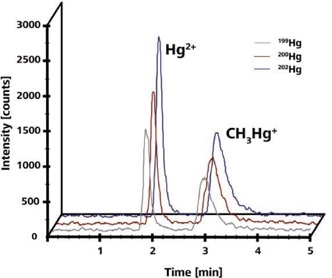
Figure 1. 10 μg/L Hg(II) and CH3Hg+. Chromatograms obtained at different masses were shifted for clarity. Column: DVB-C18 column, 150 × 4.6 mm, 2 μm; eluent: 50 mmol/L pyridine, 0.5% (w/v) mmol/L cysteine, 5% (v/v) methanol (pH = 3); flow rate: 1 mL/min, isocratic; m/z 199, 200, 201, 202.
Mercury species in crude oil by speciated isotope dilution LC-ICP/MS
Mercury, which arises from many anthropogenic and natural sources, is present in various physical and chemical forms in the environment. One of the processes that releases mercury into the environment is the burning of fossil fuels, which is a major concern. Sources include petroleum, crude oil, gas condensate, and natural gas and these contain significant amounts of mercury. Depending one where it is produced, crude oil can contain mercury ranging from 0.1 to 20,000 μg/kg.
Results
By following ccf-SIDMS in accordance with the EPA Method 6800, HPLC-ICP/MS analysis of mercury species can be applied successfully for crude oil samples (Figure 2). The method detection limit for C2H5Hg+, Hg2+, and CH3Hg+ was below 0.1 ng/mL and therefore below 1ng/g for crude oil samples.
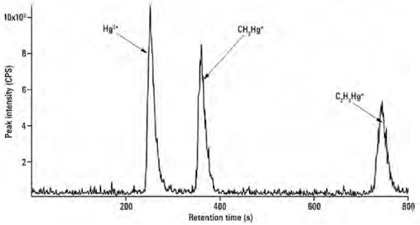
Figure 2. Three mercury species (10 μg/L each). Column: Supelcosil LC-18 (300 mm × 4.0 mm, 5 μm); eluent: 0.4% (w/v) L-Cysteine, 0.05% (v/v) 2-mercaptoethanol, 0.06 mol/L ammonium acetate, 5% (v/v) methanol; flow rate: 1 mL/min; m/z 199, 200, 201, 202.
Mercury species transformations in human hair using speciated isotope dilution mass spectrometry
By testing hair certified reference material (IAEA-085) approved for the content of monomethyl mercury and total mercury, nine different analytical extraction procedures commonly used for mercury speciation in human hair were assessed. For each procedure, the extraction efficiencies, recoveries, and potential for species transformations during sample processing were assessed using SIDMS as a quantitative and diagnostic tool. The extraction methods evaluated were based on acid leaching with HNO3 and HCl. ICP/MS and high-performance liquid chromatography-ICP/MS were used to detect total mercury and mercury species, respectively, for all extraction procedures.
Results
To prevent relative biases from being interpreted as accurate species measurement, all the methods tested could be corrected by applying isotopic correction using SIDMS protocol (Figure 3). This shows that, providing isotopic equilibration is achieved, the sample preparation and extraction errors can be corrected to ensure precise and legally justifiable and accurate results.
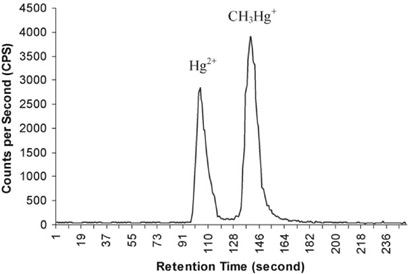
Figure 3. Speciation of inorganic mercury and monomethyl mercury. Column: Nova-Pak C18 RP column, 60 Å, 4 μm, 3.9 mm × 150 mm; eluent: 0.4% L-Cysteine (pH 5); flow rate: 1 mL/min; m/z 199, 200, 202
Microwave-assisted extraction protocol for analysis of total mercury and methylmercury in fish tissues
To extract methylmercury (CH3Hg+), inorganic mercury (Hg2+), and total mercury (Hg) from fish tissues, a fast and efficient closed vessel microwave-assisted extraction (MAE) method based on acid leaching was developed.
Humans are mainly exposed to methylmercury (CH3Hg+) through the intake of fish (85–90% of total Hg is present in aquatic organisms). Microbial action on Hg2+ in aquatic ecosystems leads to the formation of CH3Hg+, one of the most toxic forms of Hg. Readily bioavailable and able to bioaccumulate in the food chain, CH3Hg+ reaches relatively high concentrations in large predator fish such as swordfish, marlin, shark and certain tuna species. In the past, the intake of contaminated fish has led to severe neurological damage in growing fetuses and children (Aschner, T., 2005; Gochfeld, M., 2005; Burger, J., 2005).
Results
A quick and efficient MAE method was developed for extracting total Hg, Hg(II) and CH3Hg+ in fish tissues. Optimum conditions for the method were 0.5 g of sample, 10 mL of solvent containing 0.25 mol/L NaCl and 5 mol/L HCl, and an irradiation time of 10 minutes at 60 °C, as illustrated in Figure 4. Analyzing both total Hg and Hg species in different biological certified reference materials proved the accuracy of the method. SIDMS was successful as a diagnostic tool for validating the MAE method.
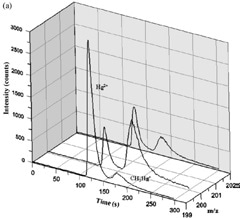
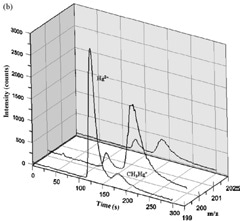
Figure 4. Dog fish liver DOLT-3 (top) and marlin fish tissue (bottom) samples spiked with 199Hg(II) and CH3 200Hg+ before MAE. Column: PS-DVB C18 RP (150mm × 4.6mm, 2 μm); eluent: 50 mmol/L pyridine, 0.5% (w/v) L-cysteine, and 5% (v/v) methanol (pH 3); flow rate: 1mL/min.
Mercury – further applications with IC-ICP/MS
Comparison of methods with respect to efficiencies, recoveries, and quantitation of mercury species interconversions in food demonstrated using tuna fish. Reyes, L. H.; Mizanur Rahman, G. M.; Fahrenholz, T.; 'Skip' Kingston, H. M. (2008) Anal. Bioanal. Chem. 390(8), 2123–2132
References
- Aschner, M.; Syversen, T. (2005) Methylmercury: Recent advances in the understanding of its neurotoxicity. Ther. Drug Monit. 27, 278–283.
- Bianchi, A.; Calabi, L.; Corana, F.; Fontana, S.; Losi, Maiocchi, A.; Paleari, L.; Valtancoli, B. (2000) Thermodynamic and structural properties of Gd(III) complexes with polyaminopolycarboxylic ligands: Basic compounds for the development of MRI contrast agents. Coord. Chem. Rev. 204, 309–393
- Burger, J.; Gochfeld, M. (2005) Heavy metals in commercial fish in New Jersey. Environ. Res. 99(3), 403–412
- Dietary Supplement Fact Sheet: Selenium. US National Institutes of Health; Office of Dietary Supplements. Retrieved Oct 11, 2016.
- Gochfeld, M.; Burger, J. (2005) Good fish/bad fish: A composite benefit–risk by dose curve. Neurotoxicology 26(4), 511–520
- Herrmann, T. Einsatz der On-line-Kopplung von Ionenchromatographie und ICP-MS zur Bestimmung von Anionen. Diploma thesis, Philipps-Universität Marburg, Germany, 2006
- Knöll, J. Ph.D. Ultratrace determination of aminopolycarboxylic acid based chelating agents using inverse on-line coupling of IC with ICP-MS. PhD thesis, Philipps-Universität Marburg, Germany, 2013
- Knöll, J.; Seubert, A. (2012) Indirect ultra trace determination of aminopolycarboxylic acids in surface water using ion exchange chromatography coupled on-line to inductively coupled plasma mass spectrometry. Journal of Chromatography A 1270, 219-224.
- Kümmerer, K.; Ed. Pharmaceuticals in the environment: Sources, fate, effects, and risks. Springer-Verlag Berlin Heidelberg, 2008; 3rd ed.
- Künnemeyer, J.; Terborg, L.; Meermann, B.; Brauckmann, C.; Möller, I.; Scheffer, A.; Karst, U. (2009) Speciation analysis of gadolinium chelates in hospital effluents and wastewater treatment plant sewage by a novel HILIC/ICP-MS method. Environ. Sci. Technol. 43(8), 2884–2890.
- Künnemeyer, J.; Terborg, L.; Nowak, S.; Telgmann, L.; Tokmak, F.; Krämer, B. K.; Günsel, A.; Wiesmüller, G. A.; Waldeck, J.; Bremer, C.; Karst, U. (2009) Analysis of the contrast agent Magnevist and its transmetalation products in blood plasma by capillary electrophoresis / electrospray ionization time-of-flight mass spectrometry. Anal. Chem. 81(9), 3600–3607
- Levenson, C. W.; Axelrad, D. M. (2006) Too much of a good thing? Update on fish consumption and mercury exposure Nutr. Rev. 64(3), 139–145.
- Montaser, A.; Golightly, D. W.; Eds. Inductively coupled plasmas in analytical atomic spectrometry. VCH Publishers, Inc.: New York, 1992.
- Rahman, G. M. M.; Martone, N.; 'Skip' Kingston, H. M. (2012) Determination of hexavalent chromium in NIST SRM 2701 by speciated isotope dilution mass spectrometry (EPA Method 6800) using IC-ICP-MS. In Handbook of hyphenated ICP-MS applications, 2nd edition; Agilent, 2012; pp 33–35. http://www.agilent.com/cs/library/applications/5990-9473EN_icpmsSpeciationHB_lr.pdf (accessed Oct 7, 2016)
- Saranko, C. J. et al. Fact Report for toxicity of arsenite and arsenate, Florida Dept. of Health, Nov 6, 1998
- Schedlbauer, O. F.; Heumann, K. G. (2000) Biomethylation of thallium by bacteria and first determination of biogenic dimethylthallium in the ocean. Appl. Organomet. Chem.14(6), 330-340
- Wilber, C. G. (1980). Toxicology of selenium: A review.Clin. Toxicol. 17 (2), 171–230.
About Metrohm
At Metrohm is one of the world’s most trusted manufacturers of high-precision instruments for chemical analysis. Metrohm was founded in 1943 by engineer Bertold Suhner in Herisau, Switzerland. Today, Metrohm is represented in 120 countries by subsidiaries and exclusive distributors. The global Metrohm Group also includes the Dutch companies Metrohm Applikon and Metrohm Autolab, manufacturers of online analyzers and instruments for electrochemical research, respectively. Recently, the Metrohm Group was joined by Metrohm Raman, a leading manufacturer of handheld Raman spectrometers.
Metrohm is the global market leader in analytical instruments for titration. Instruments for ion chromatography, voltammetry, conductivity, and stability measurement make the Metrohm portfolio for ion analysis complete. Instruments for Near-infrared and Raman spectroscopy are another, strongly growing segment of the Metrohm portfolio.
Metrohm is a problem solver, both in the laboratory and within the industrial process. To this end, the company offers their customers complete solutions, including dedicated analytical instrumentation as well as comprehensive application know-how. More than 30% of the company’s employees at the Metrohm international headquarters in Herisau work in R&D.
Metrohm has been owned 100% by the non-profit Metrohm Foundation since 1982. The Metrohm Foundation, which does not exert any influence on the company’s business operations, sponsors gifted students in the natural sciences, supports charitable and philanthropic purposes and, above all, ensures the independence of the company.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.