Introduction
Studies on assisted fertilization via micromanipulation techniques have gradually evolved from 1988 to the present day. The technique of intracytoplasmic sperm injection or ICSI (Figure 1) is a great benefit in providing treatment for infertile couples, especially those patients with male factor.
The limitations in in vitro fertilization (IVF) have been overcome in ICSI, where couples can have their own child, and is currently the preferred method of assisted reproduction treatment. Similar to IVF, with ICSI, the oocytes are first retrieved through follicle aspiration. This is followed by hyaluronidase treatment to remove the corona radiata and cumulus cells.
Parallel preparation of the spermatozoa is done as per the guidelines and procedures proposed by the WHO. A single spermatozoon is selected and injected into an oocyte via an injection pipette, which is a thin glass capillary. If fertilization takes place, the embryo thus formed is transferred after 2 to 5 days of microinjection into the uterus.
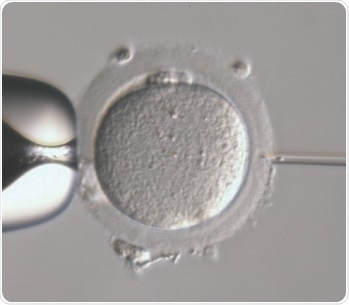
Figure 1. IntraCytoplasmic Sperm Injection (ICSI). (Picture from Centre of Reproductive Medicine and Andrology, IVF laboratory, Münster, Germany).
In order to carry out such delicate manipulations involved in ICSI, Eppendorf has developed the micromanipulator TrasnferMan 4m (Figure 2). This system provides many unique features that facilitate the workflow process. For instance, the time required for the ICSI process, when utilizing the Eppendorf TransferMan 4m1, is lower than previous models. This factor is vital to the entire procedure as the exposure of oocytes to external variances, which may be unfavorable, is minimized.

Figure 2. Workstation for ICSI with Eppendorf micromanipulator TransferMan 4m, CellTram Air and CellTram vario
1The TransferMan 4m is registered in the European Union as a medical device (according to Medical Device Directive 93/42/EEC). The TransferMan 4m is classified in the United States as an assisted reproduction micromanipulator and microinjector medical device under 21 CFR 884.6150.
Materials and Methods
Devices
- Differential interference contrast (DIC) or modulation contrast-based inverted microscope integrated with objective lenses 10x, 20x and 40x
- Two Eppendorf TransferMan 4m micromanipulators (one to move the holding capillary and the other to collect and transfer the spermatozoa)
- Inverted microscope adapter
- CellTram® Air microinjector to hold the oocyte.
- CellTram® Vario-microinjector to transfer the sperm
Consumables and media
- Tested embryo (e.g., M-8410 (Sigma-Aldrich® or others), light mineral oil
- Tissue-culture-grade (e.g., no. 353655 dish 50mm IVF low wall (BD Falcon®) or others), shallow cell culture dishes,
- Eppendorf VacuTips holding capillaries or Gynemed Nr. 001-100-30
- Eppendorf TransferTip® (ICSI) injection capillaries or Origio® (MIC-50-30)
- Culture media (HEPES-buffered, supplemented with pyruvate, protein and antibiotics)
- PVP (polyvinylpyrrolidone) or similar formulations
Preparation of microinjection dish
The media and the oil must be heated to 37°C before use. Droplets of the medium, ranging between 5µL and 25µL, are placed in a Petri dish’s center. The droplets are then supplemented with PVP prior to sample addition. Additional PVP-containing droplets may be required to store the isolated spermatozoa prior to injection. The droplets are then coated using light mineral oil to aid in maintenance of stability, pH, and temperature. After preparation, the microinjection dish is placed in an incubator until further use.
Micromanipulator set up
The TransferMan 4m micromanipulator helps in selecting five types of application masks with function keys that are optimized for the respective applications (Figure 3). The feature included in these functions is position storage. While four masks contain fixed keys of function with a few definable free soft keys, the “My app” mask can be programmed to cater to specific requirements. For spermatozoa injection, the “ICSI” application mask is selected.

Figure 3. Display of the TransferMan 4m control panel: choose your application
Microinjection capillary preparation
The two microcapillaries must be positioned into the CellTram microinjector holding system. They are then equilibrated and aligned before starting the ICSI procedure. Since the diameter of the holding pipette is greater than that of the injection pipette, it is used to guide the steps on equilibration and positioning.
The universal capillary holder is first integrated with the micropipettes. This is connected by a tube to the microinjector. While using systems filled with oil, care must be taken that air bubbles are not present. The capillaries are then pushed gently into the holder tool past the sealing rings. The universal capillary holder is then attached to the head of the TransferMan 4m micromanipulator and checked for alignment. The angle of individual injection can be corrected using a knurled screw (Figure 4).
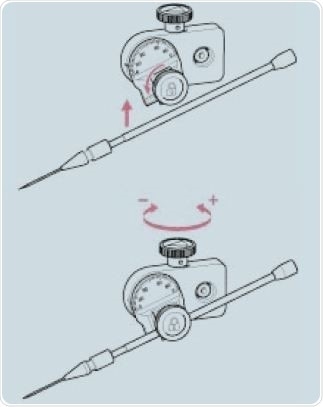
Figure 4. Adjustment of the injection angle.
For vertical alignment of the capillary, the holder is to be rotated, even while the pipette is held tightly in place. The two pipettes are to be aligned straight. Horizontal alignment should be done carefully. However, tilting of the holding pipette is to be avoided as it is required to lie flat on the base of the Petri dish to facilitate oocyte aspiration.
The injection pipette, in contrast, must slightly tilt downwards to allow the spermatozoon’s tail to break properly. The micropipettes are to be primed with medium prior to use so as to ensure that the gametes that are manipulated do not come in contact with oil or air. This equilibration can be achieved using the ICSI medium.
Position Storing
The application mask of ICSI can store about three positions. Two of these are reserved already (Pos. 1 and Pos. 2), while the third position can be programmed individually (Pos. 3). The joystick is to be pressed twice to reset the required position of the capillary (Figure 5) during the course of the ICSI process.
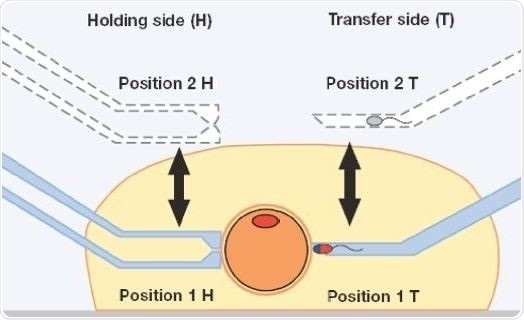
Figure 5. Storing of positions within and above the injection droplet (schematic representation).
Spermatozoa selection using the dual speed joystick
TransferMan 4m’s DualSpeed joystick can be declutched to reposition if maximum displacement of the path’s actual radius has been achieved. This feature is used to move the needle carefully in the fine or extra fine speed mode, while it is still able to do a quick motion range on entry into the joystick’s dynamic outer zone (Figure 6).
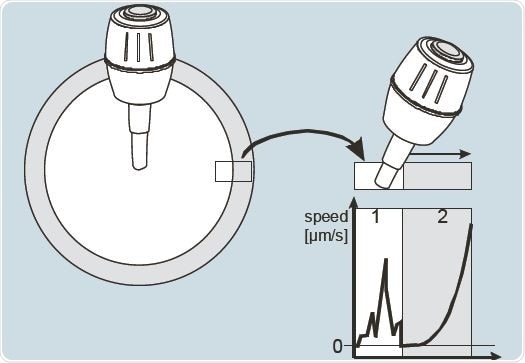
Figure 6. Dual Speed joystick with proportional and dynamic deflection
Microinjection
The sample sperm is placed into a pre-filled PVP drop, while the oocytes are transferred to the designated drops of the medium. The spermatozoon is then aspirated into the ICSI Transfer Tip capillary tail-first, while the CellTram Vario knob is rotated gently. The Petri dish containing the oocyte and cell is then focused. The other TransferMan 4m joystick is pressed in order to push the holding capillary from the 2H position to 1H position. Attachment of the oocyte to the holding capillary is facilitated by the CellTram Air device.
Capillary injection is then focused. The knob is next rotated to move the spermatozoon along the capillary till it is fixed at the tip (Figure 7A). The transfer capillary is pushed across the zona pellucida (Figure 7B) and then into the ooplasm. The oocyte is to be pricked at the center in order to break the membrane of the oolemma atraumatically and gently. To ensure this, a tiny amount of the ooplasm is aspirated into the capillary of the injection to denote membrane rupture (Figure 7C). The ooplasm that was aspirated was deposited at the center of the oocyte (Figure 7D).
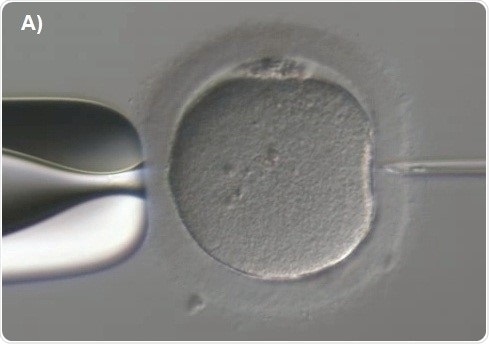
Figure 7A. ICSI procedure - Position the injection needle with spermatozoon at the 3 o’clock position.
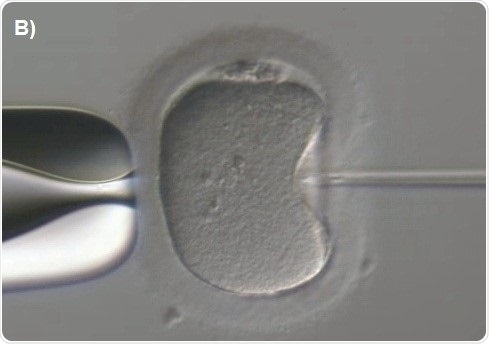
Figure 7B. Pushing of the needle through the zona pellucida.
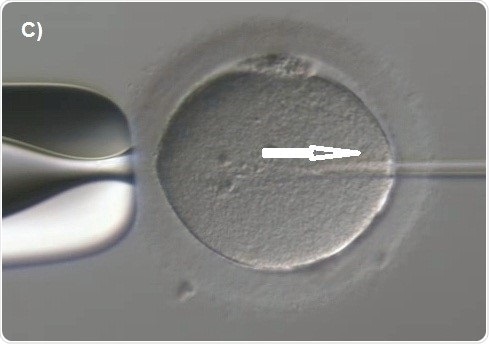
Figure 7C. Aspiration of cytoplasm (arrow)
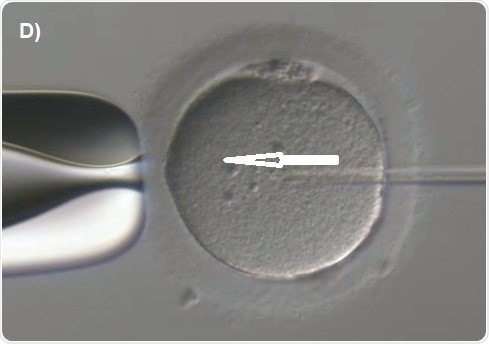
Figure 7D. Pushing out the cytoplasm together with the spermatozoon into middle of the oocyte (arrow).
The amount of medium is to be minimized by introducing the PVP into the cytoplasm. The capillary is then removed carefully. The oocyte that has been injected is then released. In case of multiple oocyte injection, 3 to 6 oocytes are to be kept in the Petri dish to prevent stressing of the oocytes. Following the addition of the culture medium, the Petri dish is incubated overnight.
Fertilization assessment and transfer of embryo
About 15 to 18 hours post microinjection, fertilization assessment of the oocytes is done. The oocytes that have been fertilized must possess two polar bodies and two pronuclei. Finally, gamete uterine transfer is done 2 to 5 days post microinjection.
Acknowledgments
Produced from materials originally authored by Verena Nordhoff from the Centre of Reproductive Medicing and Andrology, University Hospital of Münster, Germany.
References
- Lanzendorf SE, Maloney MK, Veeck LL, Slusser J, Hodgen GD, Rosenwaks Z. A preclinical evaluation of pronuclear formation by microinjection of human spermatozoa into human oocytes. Fertil Steril 1988 May; 49(5):835–42.
- Palermo G, Joris H, Devroey P, Van Steirteghem AC. Pregnancies after intracytoplasmic injection of single spermatozoon into an oocyte. Lancet 1992 Jul 4; 340(8810):17–8.
- WHO. (2010) Laboratory Manual for the Examination and Processing of Human Semen, 5th edn. Cambridge University Press, Cambridge, UK.
About Eppendorf NA
Eppendorf NA, with headquarters in New York, are a subsidiary of Eppendorf AG (Hamburg, Germany) – manufacturer of laboratory instruments and consumables for the life sciences.
To make it easy for labs in the United States to benefit from Eppendorf quality, they provide total customer support and service: taking/tracking orders; product installation and training; application support; calibration services; general product maintenance and repair, and more!
Eppendorf's products
Eppendorf products are used in all types of life science research and testing settings – from basic laboratory applications to highly specialized cell and molecular biology applications. They are highly regarded for their quality design and performance – beginning with extensive research and development, adding state-of-the-art technology and ending with strict quality-controlled manufacturing are what make their products stand out from the rest. It is what has made them a brand you have been able to rely on for over 70 years.
Over the years Eppendorf has improved upon and added to their flagship products – pipettes and pipette tips, centrifuges and microcentrifuge tubes – to now include the most ergonomic liquid handling devices and automated pipetting stations, quiet centrifuges, fast and fully flexible thermal cyclers, sample-protecting deepwell plates, cell manipulation systems and microcapillaries, ultra-low temperature freezers, shakers, incubators and bioprocessing equipment.
Eppendorf's services
Customer Support Representatives are available M–F, 8:30 AM – 8:00 PM (EST).
Local Sales Representatives cover the entire US to give you the fastest best possible attention. They know their products – and better yet, they know your applications. Feel confident in their abilities to assess your needs and determine the best products and systems to give you the best results.
Field Specialists also cover the entire US to provide post-sales installation and training on their specialty systems and automation.
The Applications Hotline is manned by degreed scientists working in their own fully functional life science laboratory. They are on call and email-accessible to help you troubleshoot your application or answer any technical question you may have. The Eppendorf Services group provides a wide variety of support services such as pipette calibration, instrument maintenance programs, and expedited repair.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.