Introduction
Neurobehavioral and metabolic effects of GLP-1 RAs
How GLP-1 RAs affect appetite
Changes in cravings, mood, and behavior
How GLP-1 drugs may reshape addiction pathways
Clinical and public health implications
Conclusions
References
Further reading
The widely used obesity and diabetes GLP-1 drugs may do far more than reduce weight. Emerging evidence suggests they alter food cue reactivity, reward signaling, and addictive behaviors, while leaving key questions about mood and emotional eating unresolved.
 Image Credit: Edugrafo / Shutterstock.com
Image Credit: Edugrafo / Shutterstock.com
Introduction
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) have transformed the treatment of metabolic conditions such as obesity and diabetes. In addition to their weight loss benefits, these drugs also act through gut-brain signaling and central nervous system pathways involved in appetite, reward, and feeding behavior, although the extent to which individual agents cross the blood-brain barrier remains an active area of investigation.3
Obesity currently affects over one billion individuals worldwide and, in 2024, was directly implicated in over 3.7 million deaths.1 Metabolic diseases like obesity and overweight have traditionally been treated with combinations of behavioral therapy and pharmacotherapy, the latter of which was often associated with poor efficacy and weight regain after medication cessation.2
Endogenous GLP-1 is a gut hormone released during and soon after food intake to modulate blood glucose levels and satiety. To prolong these effects therapeutically, GLP-1 RAs were designed to resist rapid degradation, allowing these medications to remain active in the human bloodstream for days.3 GLP-1 RAs are widely used to treat obesity and several other metabolic conditions, with the American Psychological Association (APA) reporting that over 12% of all U.S. adults have used at least one GLP-1 medication since 2020.2
GLP-1 receptors are expressed in brain regions involved in satiation, satiety, and reward, including the hypothalamus, brainstem, and mesolimbic pathways.3 Thus, the significant weight loss and metabolic benefits of GLP-1 RAs are partly due to neurological mechanisms that may have therapeutic implications beyond weight loss and diabetes management.3
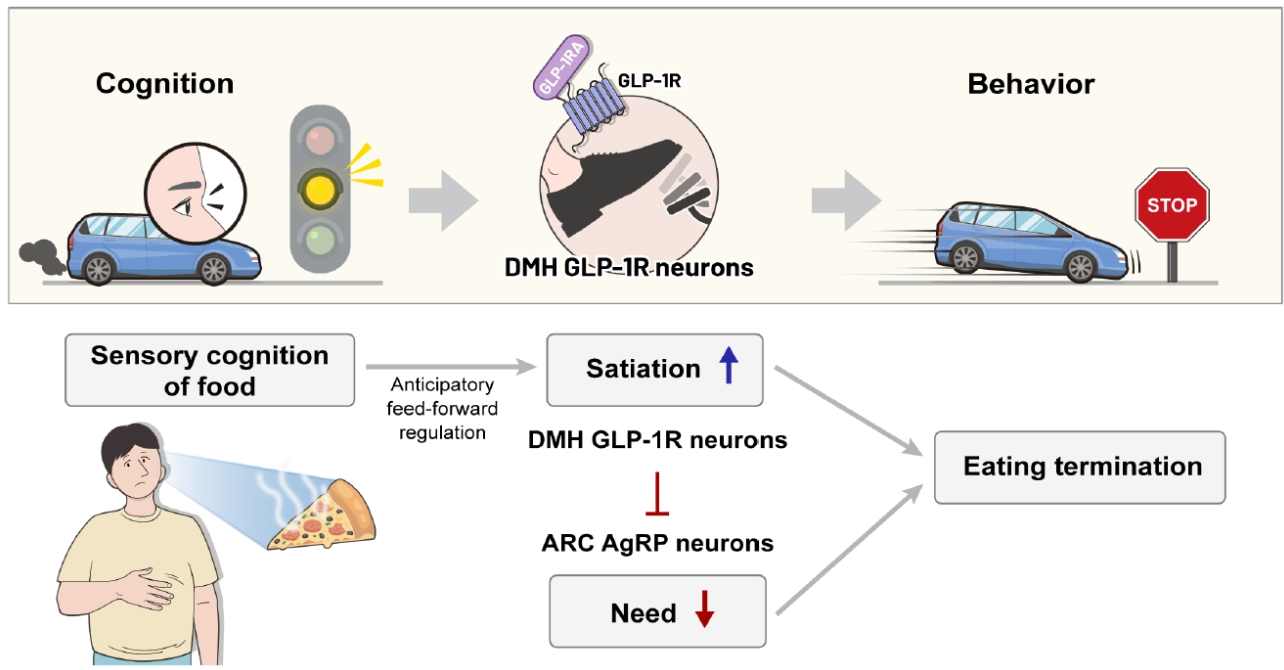
Feed-forward cognitive control of satiation. The role of dorsomedial hypothalamus (DMH) glucagon-like peptide-1 receptor (GLP-1R) neurons regulating cognitive satiation can be likened to stepping on a vehicle’s brake in response to traffic signals - a preemptive action taken before reaching the actual stop goal, reflecting a feed-forward control mechanism. These neurons project inhibitory signals to arcuate nucleus (ARC) agouti-related peptide (AgRP) neurons, ultimately contributing to meal termination. Notably, glucagon-like peptide-1 receptor agonists (GLP-1RAs) can potentiate the activity of DMH GLP-1R neurons.
How GLP-1 RAs affect appetite
Within the gastrointestinal tract, GLP-1 RAs delay gastric emptying, thereby sustaining mechanical fullness signals through vagus nerve activity.3 In the brain, GLP-1 RAs target areas like the brainstem and hypothalamus to modulate appetite, food cravings, and hunger signals.
Mechanistic studies reveal that GLP-1 RAs activate neurons in the arcuate nucleus, which are linked to hunger suppression. These drugs also activate GLP-1 receptors in the hypothalamus, which increases the release of anorexigenic peptides like pro-opiomelanocortin while reducing the release of orexigenic peptides like neuropeptide Y and agouti-related peptide. Much of this circuit-level evidence comes from animal and translational studies, supported by a smaller body of human neuroimaging work.3
Recent reviews also highlight the dorsomedial hypothalamus as an important target for pre-ingestive cognitive satiation, alongside broader hypothalamic and hindbrain circuits that shape meal initiation, satiation, and satiety. Although GLP-1 signaling interacts with other metabolic systems, evidence in the supplied papers supports a primary role in reducing food intake and externally driven eating rather than a simple generalized increase in energy expenditure.3
 Image Credit: Juan Gaertner / Shutterstock.com
Image Credit: Juan Gaertner / Shutterstock.com
Changes in cravings, mood, and behavior
The most cited GLP-1 RA-associated behavioral shift is the loss of obsessive and/or intrusive rumination about acquiring and eating food.GLP-1 RAs have been widely linked anecdotally to reduced “food noise,” a colloquial and still-evolving concept often framed as heightened and/or persistent food cue reactivity rather than a standardized clinical endpoint.5
Although research into the psychological effects of GLP-1 RA use remains nascent, emerging evidence suggests that these drugs impact mood and stress responses.4 At present, however, the evidence is mixed and comes from different tiers of data.4,6,7 Large observational analyses have reported lower risks for several neuropsychiatric outcomes, including substance use and psychotic disorders.4 Meanwhile, the European Medicines Agency’s Pharmacovigilance Risk Assessment Committee concluded that available evidence does not support a causal link between GLP-1 receptor agonists and suicidal or self-injurious thoughts and actions.6 By contrast, social media analyses have identified self-reported experiences such as insomnia, anxiety, and depressed mood, which are useful for signal detection but do not establish causality.7
Nevertheless, after reviewing over 43,000 comments across Reddit, YouTube, and TikTok, GLP-1 RA consumers appear to frequently report insomnia, unprovoked anxiety, and depressive episodes shortly after initiating pharmacotherapy. These reports should be interpreted as hypothesis-generating rather than clinically confirmed adverse-effect estimates. The pathophysiological mechanisms that contribute to these symptoms remain unclear; however, they may include delayed gastric emptying that disrupts sleep, rapid blood sugar fluctuations that trigger panic, or the psychological stress of losing food as a primary coping tool.7
How GLP-1 drugs may reshape addiction pathways
GLP-1 RAs interact with the mammalian mesolimbic reward system and have been proposed to modulate dopaminergic signaling in circuits involving the ventral tegmental area, nucleus accumbens, and other reward-related regions, thereby reducing the reinforcing value of food and some substances in preclinical models. The evidence provided in these papers is stronger for dopamine-related reward modulation than for direct serotonin-mediated mood effects.8
Evidence for addiction-related benefits is promising but still preliminary.8 In a Swedish nationwide cohort study of people with alcohol use disorder, semaglutide and liraglutide were associated with lower risks of alcohol use disorder hospitalization and substance use disorder-related hospitalization during periods of use, and these associations were numerically stronger than those seen for approved alcohol use disorder medications in the same analysis.10 Separately, the Nature Medicine Veterans Affairs study found reduced risk of substance use disorders overall.4 Although these findings are promising, randomized clinical trials are needed to clarify the potential efficacy of GLP-1 RAs for AUD and SUD treatment.
GLP-1 agonists may exert a centrally mediated effect to reduce addictive behavior at least partly via dopamine modulation.10”
Blocking the reward value of food may expose latent behavioral vulnerabilities in high-risk patients. For example, one multicenter observational study found that GLP-1 RAs successfully and sustainably reduced eating triggered by sights or smells; however, these drugs did not significantly affect emotional eating. In that study, emotional eating scores declined only transiently and were not significantly associated with long-term changes in body weight or glycemic control9. This supports a more cautious interpretation than claiming that emotional eating becomes biologically impossible during treatment.9
Study finds drugs like Ozempic could help with substance addiction
Clinical and public health implications
The neurobehavioral complexity of GLP-1 RAs and limited data on their psychological outcomes urge caution in their prescription and necessitate routine psychological support during their sustained use.2 The APA article notes that many patients discontinue treatment within a year because of side effects, cost, access issues, or other challenges, but this source does not establish mental distress as a uniform or primary reason across all patients.2
Recognizing the need for comprehensive, person-centered obesity care, the World Health Organization's 2026 global guidelines explicitly recommend pairing GLP-1 therapies with intensive behavioral interventions, including weekly counseling sessions and structured goal setting for diet and physical activity.1 The APA emphasizes the importance of mental health professionals to help patients navigate body image changes, identity shifts, stigma, and evolving relationships with food during treatment.2
Conclusions
GLP-1 RAs are multisystemic interventions capable of simultaneously addressing both physiological and some psychological barriers to weight loss. Although these medications can silence food noise, reduce externally cued eating, and show early promise in addiction-related outcomes, current evidence does not show that they reliably resolve emotional eating or uniformly improve mood across all users.4,9
References
- Celletti, F., Farrar, J., & De Regil, L. (2026). World Health Organization Guideline on the Use and Indications of Glucagon-Like Peptide-1 Therapies for the Treatment of Obesity in Adults. JAMA 335(5); 434. DOI: 10.1001/jama.2025.24288. https://jamanetwork.com/journals/jama/fullarticle/2842199
- American Psychological Association. (2025). Weight-loss drugs and mental health. APA Monitor on Psychology. https://www.apa.org/monitor/2025/07-08/weight-loss-drugs-mental-health. Accessed 6th March 2026.
- Park, J. S., Kim, K. S., & Choi, H. J. (2025). Glucagon-Like Peptide-1 and Hypothalamic Regulation of Satiation: Cognitive and Neural Insights from Human and Animal Studies. Diabetes & Metabolism Journal 49(3); 333-347. DOI: 10.4093/dmj.2025.0106. https://e-dmj.org/journal/view.php?doi=10.4093/dmj.2025.0106
- Xie, Y., Choi, T., & Al-Aly, Z. (2025). Mapping the effectiveness and risks of GLP-1 receptor agonists. Nature Medicine 31(3); 951-962. DOI: 10.1038/s41591-024-03412-w. https://www.nature.com/articles/s41591-024-03412-w
- Hayashi, D., Edwards, C., Edmond, J. A., et al. (2023). What Is Food Noise? A Conceptual Model of Food Cue Reactivity. Nutrients, 15(22), 4809. DOI: 10.3390/nu15224809. https://www.mdpi.com/2072-6643/15/22/4809
- European Medicines Agency. (2024). GLP-1 receptor agonists: available evidence does not support a link with suicidal and self-injurious thoughts and actions. https://www.ema.europa.eu/en/news/meeting-highlights-pharmacovigilance-risk-assessment-committee-prac-8-11-april-2024. Accessed 6th March 2026.
- Arillotta, D., Floresta, G., Guirguis, A., et al. (2023). GLP-1 Receptor Agonists and Related Mental Health Issues; Insights from a Range of Social Media Platforms Using a Mixed-Methods Approach. Brain Sciences 13(11); 1503. DOI: 10.3390/brainsci13111503. https://www.mdpi.com/2076-3425/13/11/1503
- Völker, K. M., Prechtl, B. L. H., Bormann, N. L., & Choi, D. S. (2026). The potential role of GLP-1 receptor agonists in substance use disorders – a systematic review. Frontiers in Pharmacology 16. DOI: 10.3389/fphar.2025.1702448. https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2025.1702448/full
- Koide, Y., Kato, T., Hayashi, M., et al. (2025). Association between eating behavior patterns and the therapeutic efficacy of GLP-1 receptor agonists in individuals with type 2 diabetes: a multicenter prospective observational study. Frontiers in Clinical Diabetes and Healthcare 6. DOI: 10.3389/fcdhc.2025.1638681. https://www.frontiersin.org/journals/clinical-diabetes-and-healthcare/articles/10.3389/fcdhc.2025.1638681/full
- Lahteenvuo, M., Tiihonen, J., Solismaa, A., et al. (2025). Repurposing Semaglutide and liraglutide for Alcohol Use Disorder. JAMA Psychiatry 82(1); 94-98. DOI: 10.1001/jamapsychiatry.2024.3599. https://jamanetwork.com/journals/jamapsychiatry/fullarticle/2825650.
Further Reading
Last Updated: Mar 17, 2026