Understanding the transmission of the virus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is crucial to control the spread of the COVID-19 disease. SARS-CoV-2 was first detected in December 2019 in Wuhan in China, and within a few months, it has spread worldwide, claiming the lives of over 979,000 individuals and infecting over 32 million to date. Excluding Antarctica, no continent is spared from the debilitating effects of this virus.
It is known that the SARS-CoV-2 infectious virions are viable on various surfaces for several hours. This is one of the primary ways by which high viral transmission leading to human infections is occurring. It presents an opportunity to break the transmission cycle by developing effective inactivation approaches.
Many studies are conducted to establish the transmission of this virus: how does it spread, how long does it survive without a host, what happens upon various decontamination efforts, etc. In one such effort, Zhitong Chen et al., in a recent medRxiv* preprint paper find that by employing an efficient cold atmospheric plasma (CAP) with argon feed gas on different surfaces inactivates the infectious SARS-CoV-2 virus.
Plasma is one of the four fundamental states of matter (i.e., solid, liquid, gas, and plasma). It comprises of gas of ions – atoms which have some of their orbital electrons removed – and free electrons that behave somewhat similar to the cellular components bound by blood plasma - hence the name. CAP operating at atmospheric pressure and room temperature is safe for treating a highly contaminated range of surfaces. Different features on the surface do not affect the efficiency of the treatment. The action of CAP is due to reactive oxygen and nitrogen species (RONS). It is known that CAP (ionized gas produced at atmospheric pressure) could be used in biomedical applications to prevent superinfections, especially in hospitals, helping patients to avoid secondary or opportunistic infections.

Temperature distribution of Ar and He plasma-treated 6-well plate. The highest temperature for each subject was found at the center of the circular thermal gradient immediately beneath the plasma discharge. The center temperature for the Ar plasma treated surface was approximately 32 °C and for the He plasma treatment was approximately 29 °C.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This team from the University of California employed argon (Ar)-fed CAP treatments to inactivate SARS-CoV-2 on various surfaces. They used plastic, metal, cardboard, basketball composite leather, football leather, and baseball leather surfaces for the test. The CAP device consisted of one powered needle electrode and another grounded outer ring electrode, connected to a high voltage transformer. They developed this using a 3D printer at UCLA. Using optical emission spectroscopy, the authors show the presence of CAP-generated ROS and RNS (such as nitric oxide [NO], nitrogen cation [N2+], atomic oxygen [O], and hydroxyl radicals [•OH]) species.
A time-dependent study on different surfaces was carried out. The discharge voltage for the feeding gases is reported at 16.6 kV at 12.5 kHz frequency. The rate of inactivation was tested in titer of recovered SARS-CoV-2 assessed in Vero-E6 cells, testing for viral cytopathic effect.
In less than 3 minutes, Ar-fed CAP treatment inactivated all SARS-CoV-2 virus on the surfaces, with excellent decontamination time, at 30 seconds of exposure, on metal and leather football surfaces. Plastic surfaces showed virus inactivation in 30 and 60-second treatments, whereas cardboard and basketball surfaces exhibited inactivation in 60 seconds.
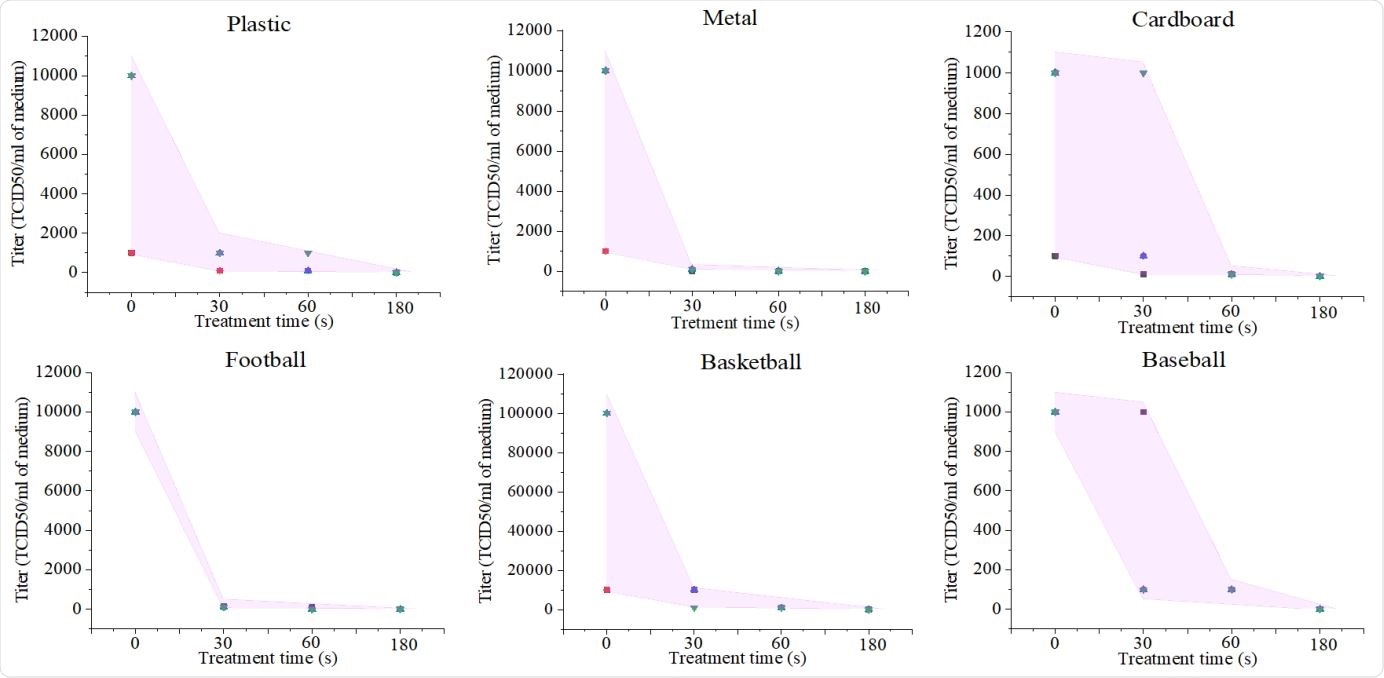
Ar-fed CAP disinfecting SARS-CoV-2. Panel A shows Ar-fed CAP treatment of a plastic surface and the optical emission spectrum of reactive oxygen and nitrogen species (RONS) (Exposure: 250 ms). Panel B shows a Bright-field image of SARS-CoV-2 infected Vero-E6 cells showing viral cytopathic effect (CPE). Uninfected (Mock) cells included as control. Panel C shows SARS-CoV-2 titer response to CAP treatment times of 0, 30, 60, and 180 seconds on surfaces of plastic, metal, cardboard, leather football, composite leather basketball, and leather baseball. Error bar in each graph is shown as shaded area.
The authors also tested the virus inactivation on cotton cloth material used for face masks, observing similar results. It is important to note that in place of Ar-fed plasma, when the authors used He-fed plasma, the disinfection of SARS-CoV-2 on metal and plastic surfaces did not occur, even at 300 seconds.
This study throws important light on the successful disinfection method of a wide range of surfaces commonly touched by people daily – the Ar-fed CAP treatment is effective in this objective. Therefore, this method has great potential in the prevention of virus transmission and curbing the disease. The cold plasma is significantly safer than most other treatments such as alcohol and UV radiation; therefore, this method has great potential as a safe and effective means to control virus transmission.
CAP can render SARS-CoV-2 harmless on the surfaces. The authors report this study to be the first one to demonstrate plasma inactivation of the SARS-CoV-2 virus – a significant milestone for the biotech community. The authors believe this work opens a wide range of opportunities for the scientific, engineering, and medical communities. Further investigations need to be initiated to carry on the study to the next level.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources