The coronavirus disease (COVID-19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), continues to spread globally. Understanding how the virus affects the body is crucial to find effective therapies and drugs.
A team of researchers at the Brigham and Women’s Hospital, Harvard Medical School, shows the cascade of vascular and tissue damage tied to SARS-CoV-2 plasma viremia, the frequency of virus isolation from cell-free plasma. This can be used to predict COVID-19 outcomes.
The SARS-CoV-2 plasma viremia has been linked to severe disease and death in COVID-19 cases in small-scale cohort studies. The current study, published on the pre-print server medRxiv*, aimed to study the mechanisms behind this relationship.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
SARS-CoV-2 plasma viremia
There is increasing evidence that COVID-19 manifests pathology beyond the pulmonary tract. As the virus spread across the globe, many people experienced symptoms showing that the disease does not only impact the respiratory tract but also other parts of the body, including the cardiovascular system.
SARS-CoV-2 nucleic acids have been detected in a wide range of body parts like the spleen, heart, liver, and digestive system in both immunocompromised and immunocompetent hosts.
Further, vascular and organ damage have been reported, while viremia has been implicated in transplacental transmission.
Plasma viremia occurs when free viruses, the ones not present within cells, are detected in the plasma of humans infected with SARS-CoV-2. Detecting plasma viremia in infection is optimal for virus detection.
All these show that infection occurs not only in the respiratory tract, and how it moves into the circulatory system is a critical step for the COVID-19 disease process. Knowing how this happens may help in formulating new therapies to combat the coronavirus pandemic.
The study authors have previously demonstrated that SARS-CoV-2 plasma viremia in hospitalized COVID-19 patients is tied to severe disease and death. These studies, however, have been limited due to small sizes and sampling late during the disease process.
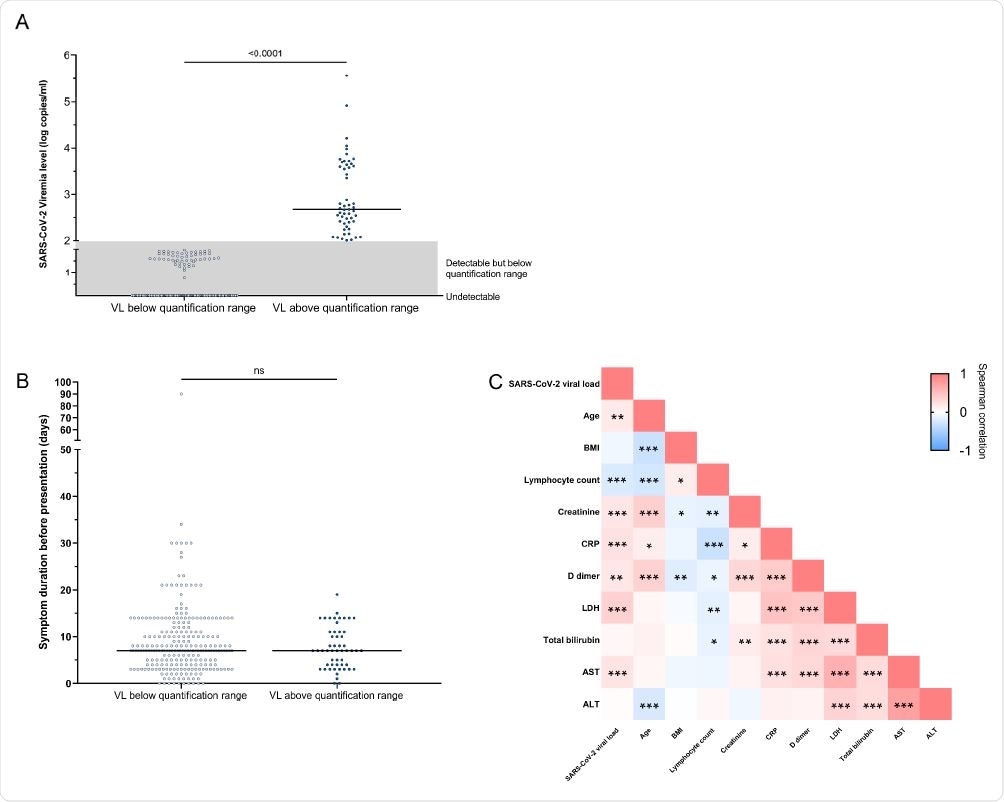
SARS-CoV-2 viremia at Day 0. (A) Distribution of SARS-CoV-2 viral load. 53 participants had viremia within the quantification range with median viral load 2.68 log copies/ml; 247 participants had viral loads (VLs) below the range of quantification or detection. (B) Duration between symptom onset and ED presentation was comparable between the viremic (quantifiable) and the aviremic/viremic (unquantifiable) group. (C) Pairwise correlation heatmap between viral load and baseline factors (Spearman's rank correlation coefficient). *, P<0.05; **, P<0.01; ***, P<0.001.
The study
In the current study, the researchers aimed to determine whether levels of SARS-CoV-2 plasma viremia could predict COVID-19 disease outcomes.
To arrive at the study findings, the team conducted plasma viral load quantification, proteomic analysis, and assessed neutralizing antibody titers in a large group of patients in the emergency department (ED).
The researchers quantified the levels of plasma viremia and how it could help predict COVID-19 outcomes after adjusting for several confounders.
The study is the largest longitudinal cohort to report a comprehensive analysis of SARS-CoV-2 plasma viremia. The findings showed that SARS-CoV-2 plasma viremia at the time when the patients are in the ED predicts maximal COVID-19 disease severity and death within 28 days.
The team also revealed that they discovered proteomic signatures upregulated in the setting of SARS-CoV-2 viremia, including evidence of damage caused by the virus. The virus causes systemic tissue damage, a pro-inflammatory response, tissue fibrosis, repair, and perturbed hemostasis.
Plasma viremia represents the relationship between extrapulmonary multiorgan involvement and severe disease outcomes. In other respiratory viruses, systemic invasion of the infection from the respiratory tract is not new. These include the respiratory syncytial virus, influenza virus, and adenovirus.
One strength of the study is, it enrolled all severely ill patients upon their arrival at the ED, minimizing bias selection. The study results revealed that when the patients were in the ED, their plasma SARS-CoV-2 viral load levels predicted severe illness and death within the next 28 days.
The patients with higher levels of plasma viremia demonstrated a “perfect storm,” wherein they exhibited a proteomic pattern of broad tissue damage. The damage affected the lungs, gastrointestinal tract, tissues, endovascular regions, and there was an increased level of pro-inflammatory markers.
“Our findings provide key insights into SARS-CoV-2 pathogenesis and identify potential therapeutic targets to mitigate COVID-19 disease severity,” the researchers concluded.
The current study can help clinicians and health experts properly assess patients to help predict outcomes. At the same time, it can help scientists in developing new therapies to combat the spreading infection.
To date, the virus has already infected more than 114 million people globally. There are over 2.54 million deaths tied to the infection.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Source:
Journal references:
- Preliminary scientific report.
Li, Y., Schneider, A., Mehtam, A. et al. (2021). SARS-CoV-2 Viremia is Associated with Distinct Proteomic Pathways and Predicts COVID-19 Outcomes. medRxiv. https://www.medrxiv.org/content/10.1101/2021.02.24.21252357v1
- Peer reviewed and published scientific report.
Li, Yijia, Alexis M. Schneider, Arnav Mehta, Moshe Sade-Feldman, Kyle R. Kays, Matteo Gentili, Nicole C. Charland, et al. 2021. “SARS-CoV-2 Viremia Is Associated with Distinct Proteomic Pathways and Predicts COVID-19 Outcomes.” The Journal of Clinical Investigation 131 (13). https://doi.org/10.1172/JCI148635. https://www.jci.org/articles/view/148635.