Scientists propose that Parkinson’s disease may begin far from the brain, where environmental toxins, microbiome disruption, and intestinal barrier damage interact to ignite the biological cascade leading to neurodegeneration.
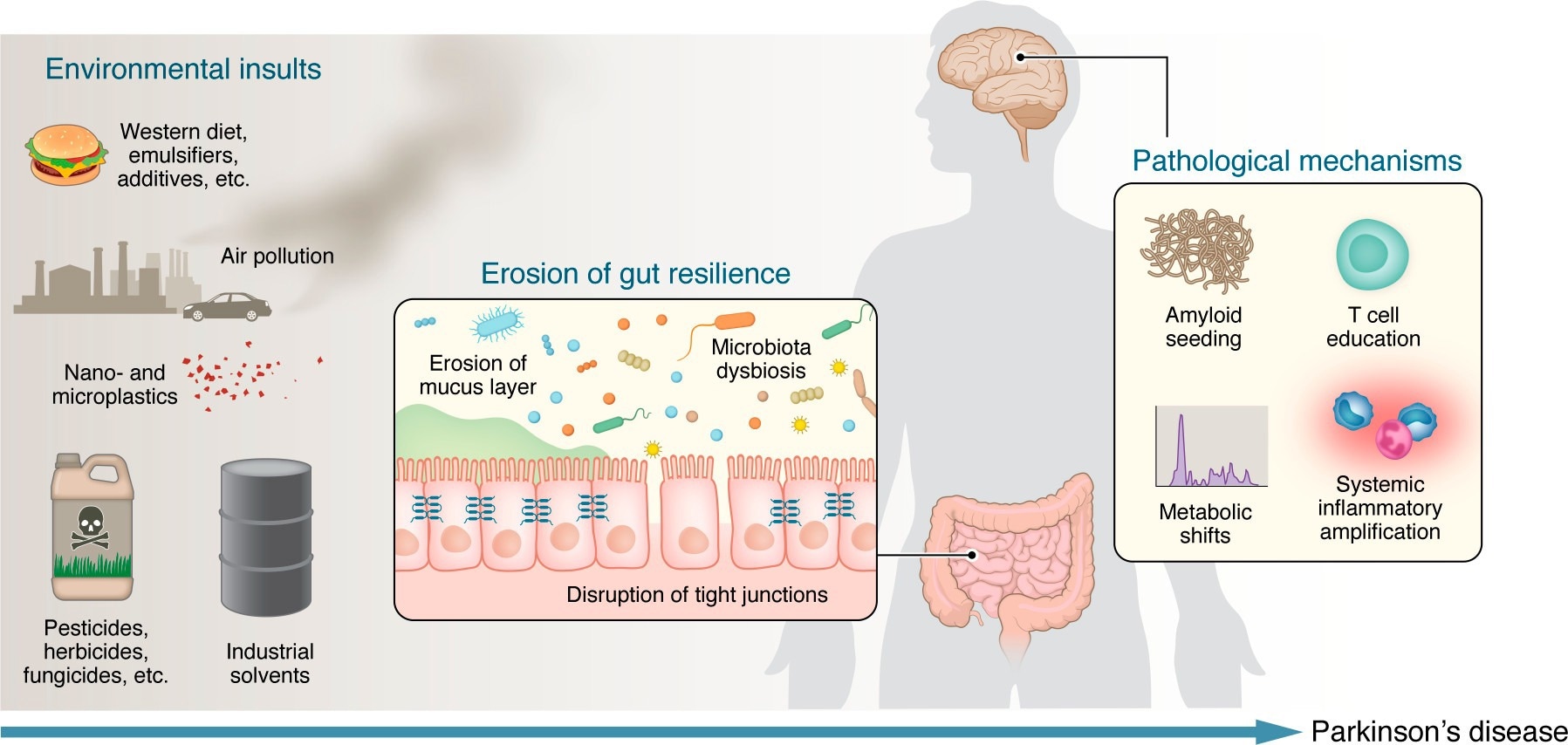
Environmental insults reduce gut resilience and initiate convergent mechanisms that drive Parkinson’s disease. A lifetime of environmental insults (including Western diets and food additives, nano- and microplastics, pesticides and herbicides, industrial solvents, and air pollution) act on the intestinal microbiome and barrier. These exposures promote microbiota dysbiosis, disrupt tight junctions, and erode the mucus layer, collectively reducing gut resilience. Once this peripheral defense is compromised, several mechanistic pathways propagate pathology to the brain: (a) amyloid seeding by bacterial functional amyloids, (b) maladaptive T cell education and autoimmune responses, (c) microbiome-driven metabolic shifts that generate neurotoxic metabolites and reduce short-chain fatty acids, and (d) systemic inflammatory amplification. Together, these processes lower the threshold for α-syn misfolding, neuroinflammation, and neurodegeneration.
A recent perspective published in the Journal of Clinical Investigation suggests that PD may, in some individuals, begin in the gut, driven by environmental exposures and changes in the microbiome.
Rising Parkinson’s Disease Incidence and Early Non-Motor Symptoms
The incidence of PD has more than doubled over the past generation, with projections estimating an increase of over 50% by 2040. This rise exceeds what can be explained by aging populations, improved detection, or genetics alone. PD typically manifests with progressive motor symptoms, such as rigidity, resting tremor, gait disturbance, and bradykinesia. However, these symptoms appear only after substantial degeneration of the dopaminergic neurons in the substantia nigra, often after roughly half of these neurons have been lost.
Prodromal symptoms, including anosmia, rapid-eye movement sleep behavior disorder, and constipation, precede motor symptoms by 10 to 20 years. Since pathological changes appear in the periphery long before they do in the brain, understanding the rising disease burden will require looking beyond the central nervous system. As such, the authors propose that the gut may act as an early interface where environmental exposures interact with host biology, rather than the brain alone being the site of PD's initial development.
Environmental Exposures and Erosion of Host Resilience
Environmental exposures, including solvents (e.g., trichloroethylene), air pollution, and pesticides (e.g., rotenone, paraquat), are implicated in PD pathogenesis. Yet, individual exposures cannot fully account for the rising incidence of PD, suggesting that the disease may arise from cumulative environmental pressures that reduce host resilience. The authors propose that the gut modulates resilience through epithelial barrier integrity and microbial function and composition.
Toxins exert selective pressure, favoring taxa with traits that confer a detrimental effect. For instance, paraquat produces reactive oxygen species (ROS), which decrease microbial diversity and enrich Enterobacteriaceae that generate functional amyloids called curli. Likewise, trichloroethylene inhibits mitochondrial complex I while restructuring the microbiota to favor sulfate-reducing bacteria, e.g., Desulfovibrio, over short-chain fatty acid–producing, neuroprotective taxa.
Several environmental factors can also disrupt the intestinal barrier. Dietary emulsifiers, e.g., polysorbate-80 and carboxymethylcellulose, erode the mucin layer and weaken the barrier. Additionally, ingested microplastics and fine particulate matter (PM2.5) compromise barrier function and trigger inflammation. These particles also concentrate pesticides and heavy metals on their surface, facilitating the transport of environmental toxins across the gut barrier.
Gut Microbiota Changes and Neurodegenerative Mechanisms
Enterobacteriaceae contribute to α-synuclein pathology by generating curli subunits (CsgB and CsgA). These amyloids are structurally similar to α-synuclein. Curli can accelerate the accumulation of α-synuclein, and it is also a pathogen-associated molecular pattern that stimulates inflammation. This dual role makes curli an important link between environmental exposures and neurodegeneration. Further, gut dysbiosis directs metabolism in different neurotoxic pathways.
The kynurenine switch upregulates indoleamine 2,3-dioxygenase, diverting tryptophan metabolism away from serotonin synthesis towards kynurenine-derived neurotoxins. Meanwhile, sulfate-reducing bacteria produce excess hydrogen sulfide, which inhibits mitochondrial cytochrome c oxidase (complex IV), impairs mitochondrial function, and exacerbates α-synuclein accumulation. The loss of short-chain fatty acids, like butyrate, further compounds these shifts.
Immune Activation Linking Gut Dysfunction to Brain Degeneration
Consequently, barrier dysfunction allows the translocation of bacterial components into systemic circulation, where innate immune sensing through Toll-like receptors (TLR2 and TLR4) can activate the NLRP3 inflammasome and trigger the release of interleukin (IL)-18 and IL-1β. These cytokines cross the blood-brain barrier and prime microglia into a hyperreactive state. Once primed, the microglia show exaggerated responses to subsequent insults from endogenous α-synuclein aggregates, viral infections, or low-level exogenous toxins.
The authors also highlight a potential role for gut-driven immune education, in which α-synuclein–reactive CD4+ T cells may be primed in the intestine before migrating to the brain, where pro-inflammatory T helper responses, including Th1 and Th17 activity and cytokines such as interferon-γ, could contribute to dopaminergic neuron injury.
Conclusions and Implications for Parkinson’s Disease Prevention
If PD results from a lifetime of environmental pressure, then effective interventions must focus on strengthening biological resilience and decreasing environmental burden. Research has focused on central PD mechanisms for decades: mitochondrial dysfunction, α-synuclein aggregation, dopaminergic neuron vulnerability, and neuroinflammation. While these remain essential targets, they represent downstream disease processes.
By the time they are detectable in the brain, the disease may have already passed a pivotal inflection point. By contrast, the gut is where environmental exposures intersect with the host. It is where epithelial barrier integrity and microbial ecology determine whether immune tolerance is eroded, misfolded α-synuclein seeds form, and systemic inflammation primes the brain for degeneration.
Overall, the gut represents a potentially clinically modifiable target in PD, one that is mechanistically upstream and therapeutically accessible even after disease onset, with early studies suggesting improvements in some symptoms and biological markers but not definitive disease reversal. Moving upstream may therefore be essential to reverse global PD trends. Strengthening peripheral resilience is one of the most promising, scientific, and scalable strategies. Preventing initiation in the gut may ultimately help modify PD's trajectory.
Journal reference:
- Palushaj B, Voigt RM. (2026). The Parkinson’s pandemic: prioritizing environmental policy and biological resilience via the gut. The Journal of Clinical Investigation, 136(5), e205275. DOI: 10.1172/JCI205275, https://www.jci.org/articles/view/205275