A new Perspective argues that long-acting siRNA therapies could ease the daily burden of hypertension treatment, but only if health systems turn each dose into a structured checkpoint for lifestyle advice, home BP review, and ongoing cardiovascular risk management.
Perspective: From daily burden to scheduled protection: the “vaccine-like” shift in hypertension
In a recent perspectives article published in the Journal of Human Hypertension, researchers discussed emerging evidence from recent trials of long-acting RNA interference (RNAi) therapy targeting hepatic angiotensinogen (AGT) to evaluate their potential to improve the real-world efficacy (and adherence) of pharmacological interventions for hypertension.
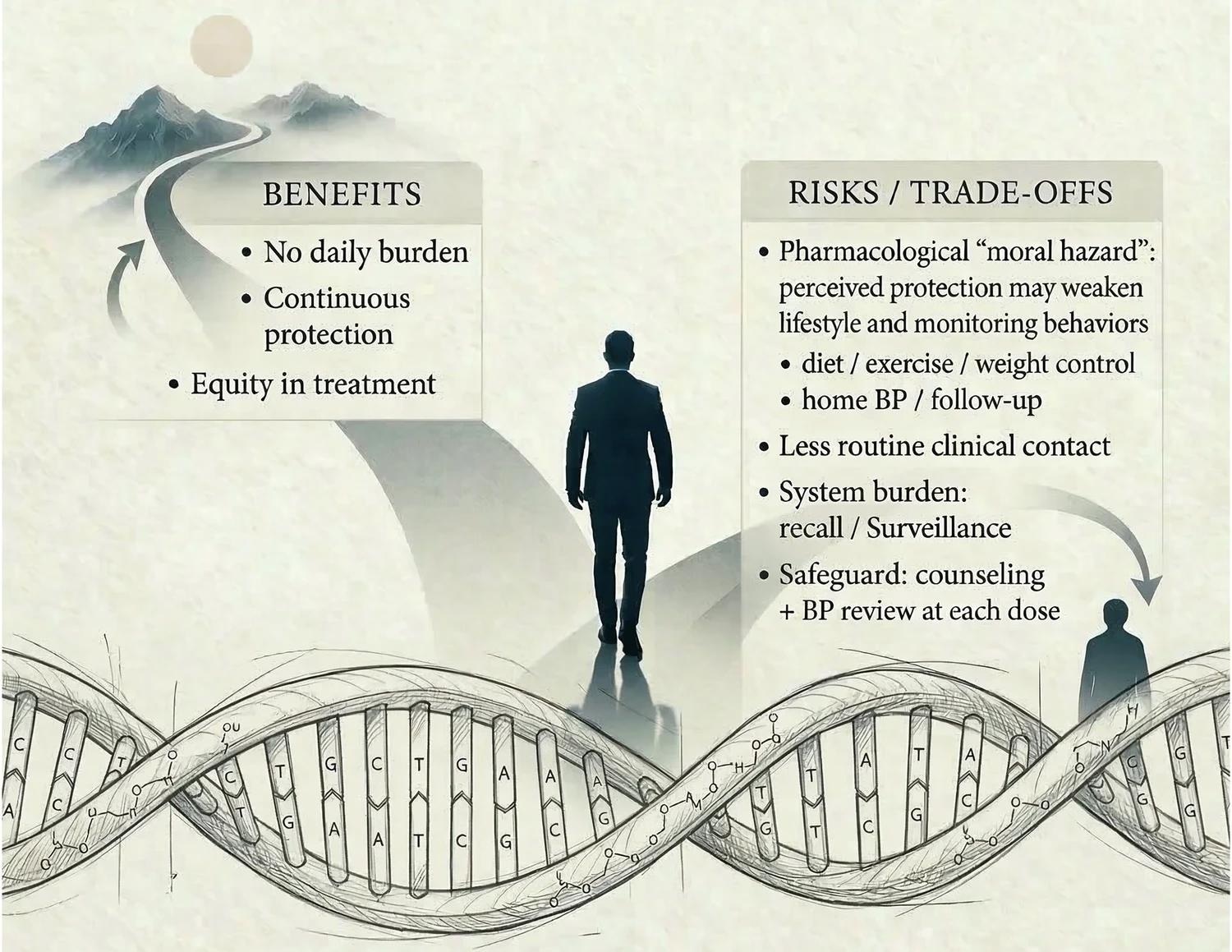
The article analyzes a "vaccine-like" shift in hypertension care responsibilities caused by using small-interfering RNA (siRNA) therapies, such as zilebesiran. These novel drugs can suppress blood pressure for months with a single dose. The authors emphasize that while this model shifts the burden of care from the patient’s memory to the healthcare system’s reliability, it also introduces "pharmacological moral hazard", the risk that patients may neglect vital lifestyle changes due to an inaccurate sense of total pharmacological security.
Hypertension Adherence and Care Model Background
Modern clinical practice often considers hypertension to represent one of medicine’s most significant paradoxes: it is highly solvable with several effective behavioral and pharmacological interventions proven to successfully mitigate the condition, yet remains a leading cause of death and disability worldwide. Pooled global analyses covering trends from 1990 to 2019 reveal that fewer than 25% of hypertensive individuals achieved controlled blood pressure (BP) levels in 2019.
Researchers have attributed the primary vulnerability in the current system to the fact that BP control is traditionally designed as a “behavioral achievement" to be carried out daily. Specifically, because hypertension frequently presents as asymptomatic, patients lack the immediate physiological feedback necessary to reinforce daily pill-taking, resulting in adherence significantly eroding over time.
Furthermore, health systems often outsource therapeutic success to the patient, who is inadvertently responsible for making daily decisions to maintain ongoing preventive care, leading to the hypothesis that the current system effectively turns cardiovascular protection into a social filter that tracks life stability rather than clinical need.
Zilebesiran Trial Evidence and Perspective Scope
The present perspective aims to examine these ideas and inform future hypertension policy by evaluating the recent operational and clinical shifts in the field catalyzed by long-acting RNAi therapeutics.
The article primarily discusses “zilebesiran”, a relatively novel small-interfering RNA (siRNA) drug that targets hepatic angiotensinogen (AGT). By suppressing this upstream rate-limiting step in the renin-angiotensin-aldosterone system (RAAS), the drug has been shown to enable sustained BP lowering for months following only a single subcutaneous dose.
Study data were discussed from several key zilebesiran-testing clinical trials, particularly KARDIA-1 (a phase 2 clinical trial demonstrating that dosing every three or six months resulted in persistent reductions in systolic BP), KARDIA-3 (a higher-risk patient-specific evaluation which found that the month-3 primary endpoint, placebo-adjusted office systolic BP lowering, did not meet statistical significance after multiplicity adjustment), and planned investigations like ZENITH.
ZENITH is an upcoming global phase 3, event-driven trial that the researchers expect to enroll approximately 11,000 patients to determine if twice-yearly angiotensinogen silencing can reduce cardiovascular death, nonfatal myocardial infarction, nonfatal stroke, and heart failure events when added to standard care.
Vaccine-Like Protection and Moral Hazard Concerns
The perspective’s arguments support the idea that zilebesiran and similar RNAi therapeutics will lead to the development of a "vaccine-like" hypertension care model, which may reduce the observed variability in global BP control caused by missed doses and fragmented routines. For example, in the KARDIA-1 trial, the pharmacological coverage was aligned with a quarterly or biannual cadence, making protection system-mediated rather than patient-dependent.
However, the authors identify a critical "therapeutic trade-off"; relocating risk from the patient to the health system means the health system must now reliably perform recall, outreach, documentation, and safety surveillance.
The authors’ major concern is the "pharmacological moral hazard": the potential for perceived protection to reduce engagement in other risk-preventing health behaviors. Specifically, there is a risk that patients may de-prioritize sodium restriction, weight management, home BP monitoring, and physical activity. The authors note that this remains a plausible behavioral concern, rather than a directly demonstrated effect in long-acting siRNA antihypertensive therapy.
Furthermore, infrequent injections (e.g., twice yearly) might exacerbate this concern by reducing routine clinical touchpoints, potentially making blood pressure less "visible" to both patients and clinicians and reducing opportunities for shared monitoring and decision-making. The authors stress that these vulnerabilities are not arguments against siRNA therapy, but design specifications for care pathways.
Hypertension Policy and Clinical Follow-Up Implications
This perspective article posits that the promise of long-acting siRNA therapeutics lies in their ability to democratize cardiovascular protection by removing the "adherence trap", likely the most persistent current cause for the global hypertension pandemic.
However, the analyses caution that realizing this potential requires resisting a "fire-and-forget" mentality. While RAAS silencing stabilizes hemodynamics, it does not inherently cultivate the healthier activity patterns or dietary habits necessary for holistic cardiovascular health.
Consequently, the authors stress that a twice-yearly injection should not be a "cure" that ends the conversation between doctor and patient, but a "security floor" that creates space for better dialogue about holistic health. They propose that each dosing visit should become a high-value touchpoint for lifestyle reinforcement, home BP review, medication reconciliation, and proactive adverse-event monitoring.
As the field moves toward the 11,000-patient ZENITH trial, the focus shifts from BP durability to whether this "vaccine-like" model can ultimately improve major cardiovascular outcomes while maintaining the visibility of hypertension as a manageable chronic condition.