A new global dataset of 239 human-infective RNA viruses shows how animal hosts, vector transmission, surveillance gaps, and viral traits shape the path from spillover to epidemic threat.

Study: A complete catalogue of human-infective RNA viruses. Image Credit: Andrzej Rostek / Shutterstock
A recent review published in the journal Scientific Data presents an updated global catalog that brings the number of ribonucleic acid (RNA) viruses known to infect humans to 239 species, 25 more than in 2018, offering new insight into emergence and spread.
Rather than appearing at random, most viruses cluster within a few families, are linked to non-human hosts, particularly mammals, and are detected at varying rates over time as taxonomy, reporting, surveillance, and sequencing technologies evolve.
While spillover into humans is common worldwide, only a minority of species reach epidemic or endemic levels in humans, highlighting a critical bottleneck between exposure and epidemic spread.
RNA viruses remain a growing threat to global health today, driving diseases such as measles, influenza, and AIDS caused by HIV, and causing new outbreaks. Recent events involving the Oropouche virus and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) underscore the epidemic potential of these viruses. Yet the viral landscape keeps changing rapidly.
Researchers identify new human-infective species almost every year, revise classifications, and expand genomic and ecological data. As evidence accumulates on transmission, host range, and spread, the need for an updated catalog becomes critical for tracking what is known and anticipating future risks.
Human RNA Virus Catalog Methods
In this paper, researchers developed an updated, expanded dataset of RNA viruses known to infect humans, capturing current knowledge through December 2024.
Building on earlier catalogs from 2001 and 2018, they conducted systematic literature searches every 1–3 years using databases such as Web of Science, PubMed, Scopus, and Google Scholar, supplemented by sources including the World Health Organization (WHO), Centers for Disease Control and Prevention (CDC), ProMed, and genome records from the National Center for Biotechnology Information (NCBI).
The dataset included only peer-reviewed primary reports providing robust evidence that RNA viruses recognized by the International Committee for the Taxonomy of Viruses (ICTV) infect humans under natural or real-world conditions, excluding intentional experimental inoculation or in vitro evidence.
The team resolved ambiguities through independent assessments and consensus, and in some cases inferred missing traits from closely related viruses. They compiled species-level data by integrating information across known subtypes and linked each virus to its first reported human case, genome sequence, and geographic origin.
The researchers recorded key traits, including transmissibility, host range, and transmission routes, using standardized criteria. They classified transmissibility into Levels 2, 3, and 4, ranging from zoonotic infections without human spread to viruses capable of epidemic or endemic spread in humans.
Lastly, the team mapped discovery dates and locations, enabling temporal and spatial analyses of virus emergence. By integrating genomic, ecological, and epidemiological data into a single framework, the updated dataset provides a robust foundation for studying viral diversity, evolution, and public health risk.
The annual count of new and (currently) ICTV-recognised, human-infective RNA virus species is shown in Figure 2a. Figure 2b depicts the accumulation of species over time, as well as the accumulation of genera and of families containing one or more human-infective RNA virus species. The first human RNA virus – Yellow fever virus – was reported in 1901. The number of species increases slowly up to the mid-1950s and somewhat faster thereafter. By the end of the 20th century 178 species had been identified, and in the 21st century so far, a further 61 have been added. By decade, the 1960s contributed markedly the greatest number of new species (42). Next was the 2000s (31) but the rate fell again in the 2010s.
Global RNA Virus Discovery Patterns
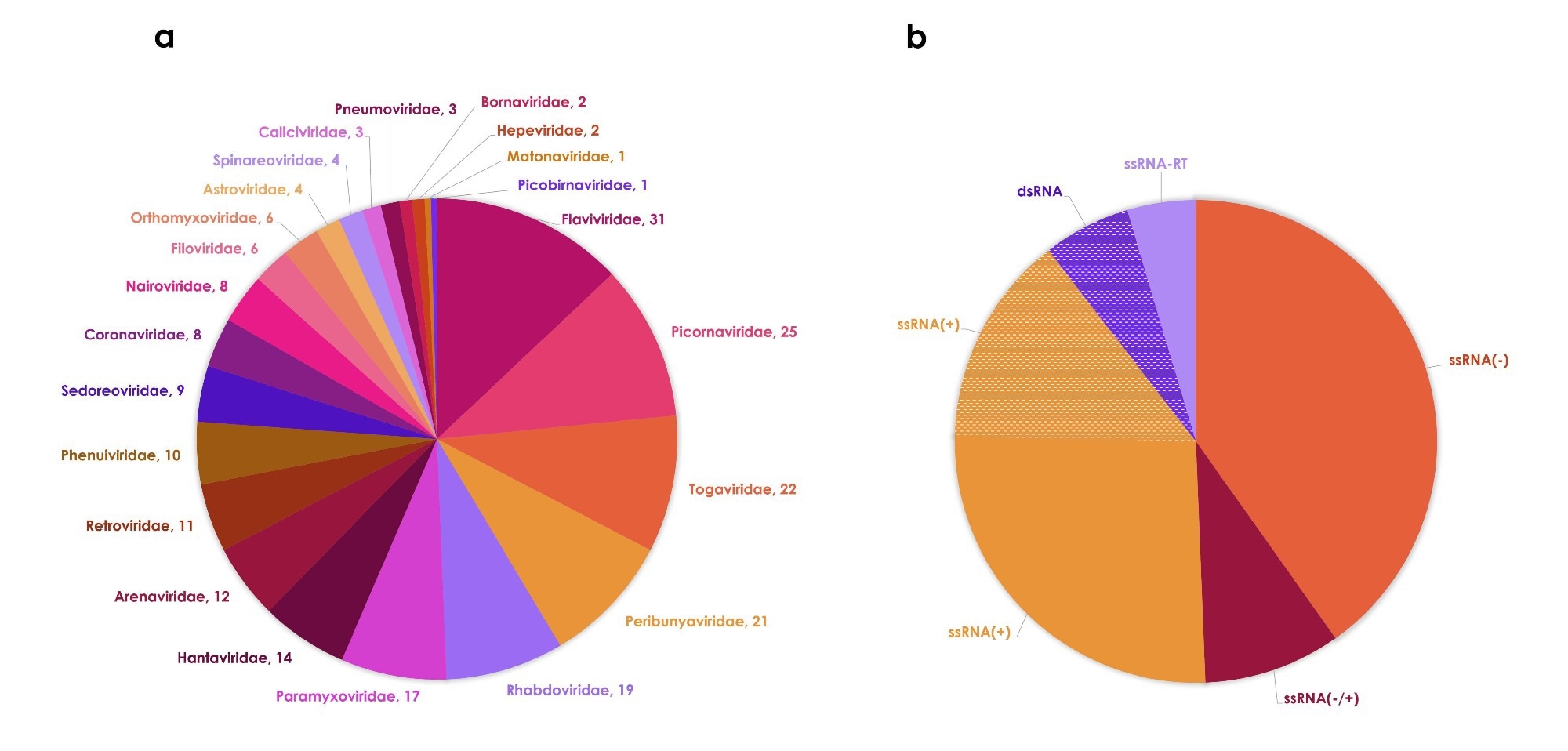
The updated dataset includes 239 RNA viruses known to infect humans, as classified by the ICTV. Compared with 2018, this reflects 25 additional species identified through new discoveries and taxonomic updates.
These species span 61 genera and 23 families, although diversity remains concentrated within a few families, and most viruses share common genomic features, particularly single-stranded RNA genomes.
Over time, discoveries have increased from the mid-20th century, though the authors note that formal analysis is needed to determine whether discovery rates are rising or falling overall.
After minimal growth in the early 20th century, identification rates rose sharply from the mid-1900s, with notable peaks in the 1960s and early 2000s. However, most newly identified species extend existing genera and families rather than introducing entirely new taxonomic groups.
Geographically, the first reported human cases have occurred across all inhabited continents, with clusters in regions with stronger surveillance systems. This pattern highlights both the global nature of viral spillover and the influence of detection capacity on discovery.
Spillover, Transmission, and Epidemic Potential
Ecologically, the majority of viruses (62%) are strictly zoonotic (Level 2) and do not sustain human-to-human transmission. Only 60 species reach Level 4, meaning they are endemic in humans or capable of epidemic spread, and many of these still maintain animal reservoirs.
Most viruses are associated with non-human mammalian hosts, reinforcing their central role in emergence. Transmission routes are diverse, but vector-borne spread, primarily via mosquitoes and ticks, dominates, followed by inhalation and direct contact pathways.
Notably, the transmission routes of a subset of viruses remain uncertain, reflecting persistent knowledge gaps. Together, these findings clearly underscore a landscape defined by repeated documented spillover, expanding discovery, and limited adaptation to sustained human transmission.
RNA Virus Surveillance and Risk Prediction
These findings point to a more targeted and proactive approach to emerging viral threats. Rather than searching broadly for entirely novel pathogens, future efforts could use this dataset to examine high-risk viral families, mammalian reservoirs, and regions with limited surveillance, where undetected spillover is most likely to occur.
Expanding genomic sequencing, metagenomics, and real-time surveillance will be critical to closing persistent knowledge gaps, particularly around transmission routes and host range.
At the same time, the dataset provides a valuable foundation for modeling trends in discovery and identifying traits linked to epidemic potential. As it continues to evolve, it can help refine risk prediction and guide early warning systems.
Ultimately, the challenge is not only to discover new viruses, but to understand which ones are most likely to adapt, spread, and pose the next global health threat.