Extracorporeal membrane oxygenation (ECMO) is a method of providing heart and lung support to patients, usually following a heart attack, when the heart is no longer capable of pumping blood around the body sufficiently.
Blood is pumped away from the body via a cannula that is inserted into a major vein and passed through a membrane tube that is surrounded with flowing oxygen gas to become oxygenated, before being returned to the body by cannula into either a major artery or vein.
Many patients infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) have presented respiratory failure, with around 10% of serious cases developing acute respiratory distress syndrome (ARDS).
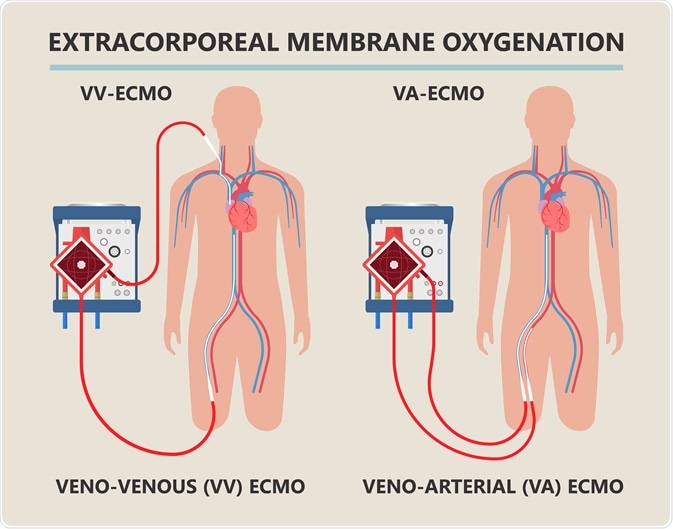 Diagram depicting how Extracorporeal membrane oxygenation (ECMO) works. Blood is pumped away from the body via a cannula that is inserted into a major vein and passed through a membrane tube that is surrounded with flowing oxygen gas to become oxygenated, before being returned to the body by cannula into either a major artery (VA ECMO) or vein (VV ECMO). Image Credit: rumruay / Shutterstock.com
Diagram depicting how Extracorporeal membrane oxygenation (ECMO) works. Blood is pumped away from the body via a cannula that is inserted into a major vein and passed through a membrane tube that is surrounded with flowing oxygen gas to become oxygenated, before being returned to the body by cannula into either a major artery (VA ECMO) or vein (VV ECMO). Image Credit: rumruay / Shutterstock.com
Is ECMO suitable for COVID-19 patients?
Namendys-Silva (2020) wrote in an open letter to the Heart and Lung journal, dated March 2020, that ECMO shows little usefulness for those with ARDS, with only around 18% of adults with ARDS due to COVID-19 undergoing ECMO surviving.
Further, another letter by Zhang and colleagues (2021) observes that many seriously ill COVID-19 patients present with coagulation abnormalities that are worsened by contact of circulating blood cells with the cannula and that determining the dosages of anticoagulants supplied to the patient is a balancing act.
The Extracorporeal Life Support Organization recommends that the decision to put a patient on ECMO should be left to the individual hospital, as the decision is highly dependent on capability, staffing, resource constraints, patient load, and local policy. Should placing patients on ECMO cause essential resources to be denied to other patients with more positive prognoses, then it should be denied. Only those without comorbidities such as advanced age, obesity, or organ failure should be considered.
Additionally, the staffing and training requirements to maintain an ECMO unit at a hospital are significant and expensive, so many hospitals either keep only a small team or rely on transfer to regional locations. Therefore, the Extracorporeal Life Support Organization recommends that hospitals do not undertake any additional training procedures or make putting patients on ECMO standard procedure in serious COVID-19 cases, particularly during times of resource strain.
Fears that the virus could pass the membrane used in ECMO have been raised by some, though Baldoino de Souza (2021) explains that there is little risk of this except in cases of improperly maintained equipment. For example, where the virus has been found in plasma leaking from the exhaust route in some hospitals in Japan. This is particularly concerning due to the flow of oxygen passing over the membrane, as an aerosol containing the virus could be produced.
In support of ECMO for COVID-19 patients, however, a large meta-study published by Schmidt et al. (2020) examined the outcome of 492 patients treated at a number of intensive care units and found generally positive results. The estimated probability of death was around 31% 60 days after ECMO initiation, comparable to the success rate of ECMO for any other non-covid related severe ARDS.
ECMO is only ever considered in cases where the patient has a very high probability of death anyway, often indicated at 80% or over, so a result of 31% that is in line with patients suffering from ARDS from other causes is promising.
An update from the Extracorporeal Life Support Organization (ELSO) in February 2021 highlighted the need for networks for sharing data about the use of COVID-19. The guideline states that most COVID-19 patients who have been treated with ECMO have received venovenous (V-V) ECMO for ARDS. From their analysis, although data was ongoing, mortality was similar between COVID-19 and non-COVID-19 ECMO patients; however, run duration times were longer for those with COVID-19.
 Closeup of an ECMO oxygenator. Image Credit: PIJITRA PHOMKHAM / Shutterstock.com
Closeup of an ECMO oxygenator. Image Credit: PIJITRA PHOMKHAM / Shutterstock.com
Are there any alternatives?
Mechanical ventilation is more commonly administered to COVID-19 patients presenting with ARDS, which is only of aid to the lungs rather than the heart. Several forms of mechanical ventilation exist, all of which have been employed to assist patients and have shown the ability to oxygenate the blood of ARDS-suffering COVID-19 patients.
However, mechanical ventilation is not without downsides, and doctors have noted that the mortality rate of COVID-19 patients placed on invasive mechanical ventilation remains worryingly high. The two methods would not normally be used consecutively, and the decision over which to apply to any particular patient would be left to the discretion of the hospital’s medical team.
However, it is important to acknowledge that ECMO fills a role that no other method can provide for critically ill but otherwise promising patients with few comorbidities.
References
- Badulak, J. et al. (2021). ECMO for COVID-19. ASAIO Journal. doi: 10.1097/MAT.0000000000001422 https://journals.lww.com/asaiojournal/Abstract/9000/ECMO_for_COVID_19__Updated_2021_Guidelines_from.98326.aspx
- Bartlett, R. H. et al. (2020) Initial ELSO Guidance Document: ECMO for COVID-19 Patients with Severe Cardiopulmonary Failure. American Society for Artificial Internal Organs, 66(5). https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7273858/
- Hua, J., Qian, C., Luo, Z., Li, Q. & Wang, F. (2020) Invasive mechanical ventilation in COVID-19 patient management: the experience with 469 patients in Wuhan. Biomedicalcentral, 24(348). https://ccforum.biomedcentral.com/articles/10.1186/s13054-020-03044-9
- Namendys-Silva, S. A. (2020) ECMO for ARDS due to COVID-19. Heart & Lung, 49(4). https://www.heartandlung.org/article/S0147-9563(20)30100-X/fulltext
- Schmidt, M. et al. (2020) Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome associated with COVID-19: a retrospective cohort study. The Lancet, 8. https://www.thelancet.com/pdfs/journals/lanres/PIIS2213-2600(20)30328-3.pdf
- Zhang, Y., Ji, B. & Zhou, Z. (2021) ECMO support for COVID-19: a balancing act. The Lancet, 397. https://www.thelancet.com/pdfs/journals/lancet/PIIS0140-6736(20)32515-0.pdf
Further Reading
Last Updated: Mar 23, 2021