Itching is among the most persistent of skin sensations, requiring an immediate response. However, scientists say it is a protective sensation that stimulates the individual to eliminate parasites and other irritating substances from the skin. The reason for this curious sensation is the presence of irritant chemicals like histamine and serotonin, or light touch as when an insect moves across the skin, stimulating touch receptors. However, these two stimuli act through separate nerve pathways.

Image Credit: NamtipStudio / Shutterstock
It is important that the brain distinguish between light touch, which is essential for a host of tasks, and a mechanical stimulus that causes an itch which requires a scratching response. How this task is accomplished has been a bit of a mystery so far.
The pathway by which chemicals cause itching has been documented in some detail, beginning from the A delta and C fibers which do not respond to mechanical stimuli. These nerve fibers carry the impulse to the dorsal horn of the spinal cord, and via the cells carrying the gastrin-releasing peptide receptor (GRPR), to the thalamus and other areas in the brain. However, not much is known about how mechanical itch is caused.
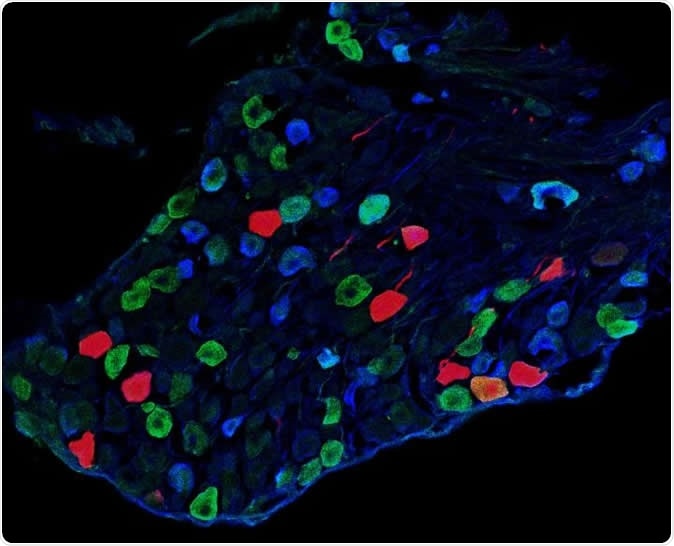
This is a cross-section from a dorsal root ganglion showing sensory neurons that transmit light-touch information from the skin to Y1 neurons within the spinal cord. Image Credit: Salk Institute
How does an itch begin?
The mechanical itch begins with low-threshold mechanoreceptors (LTMRs) in the skin that respond to light mechanical stimulation. Previous research by the same team showed that impulses from these cells are regulated in the spinal cord’s dorsal horn by inhibitory interneurons (INs) that express a chemical called neuropeptide Y::Cre (NPY::Cre INs).
When these INs are removed from mice, they develop chronic scratching without any obvious cause, and also with very light mechanical stimulation. Therefore, these neurons inhibit the passage of mechanical itch impulses. This was unchanged despite removal of the GRPR neurons, which shows that mechanical itch is transmitted quite apart from the GRPR-expressing neuron pathway. Thus the NPY::Cre INs gate the onward transmission of mechanical itch stimuli from the spinal cord.
The researchers then looked for the nerve cells that receive the mechanical itch signal from the LTMRs. Among the cells in the dorsal horn that receive these impulses, there are some neurons that secrete the excitatory peptide glutamate. A new study showed mechanical itch is processed via these peptide-signaling neurons, which transmit the signal to certain dorsal horn neurons expressing two proteins called Y1 and Lbx1 (Y1Cre neurons). Most neurons of this type in the spinal cord are excitatory.
When Y1Cre neurons are removed in the above NPY::Cre-ablated mice, the chronic scratching disappears. In normal mice too, the removal of Y1Cre neurons prevented itching in response to mechanical stimuli. Activation of Y1Cre neurons by drugs increased their scratching response to a mechanical itch, and induced spontaneous scratching. That is, the Y1Cre neurons form an excitatory pathway in the dorsal horn which is necessary to express mechanical itch. They are the cells that promote the onward transmission of the mechanical itch stimuli.
Why don’t we itch when something touches us?
The scientists found that NPY produced by the body is essential to suppress signaling by the dorsal horn Y1Cre cells. In this way it inhibits responses to light touch like that produced by a hair, for instance, as well as to mechanical itching. Mice with Lbx1Cre/ Y1f/f conditional knockout start scratching spontaneously and react excessively to itch-causing mechanical stimuli (but not chemical itch and pain), due to the lack of endogenous NPY. To prevent this, NPY release must be stimulated by activating either Y1 receptors or NPY::Cre interneurons. This shows that NPY-Y1 signals gate the mechanical itch impulses in the spinal dorsal horn.
Activating NPY::Cre IN specifically suppresses mechanical itch and sensitivity to light punctate touch, because NPY suppresses Y1+ cells in the dorsal horn. There are different types of cells that express Y1Cre of which only a few are needed for mechanical itch response.
The lack of any effect on chemical itch following the removal of these neurons shows that mechanical and chemical itch follow distinct pathways from the level of the receptors to the spinal cord dorsal horn. This is despite the fact that both types of itch feel the same and set off the same response, viz., scratching. This suggests that both pathways eventually meet in the brain.
Why is this discovery important?
The presence of the NPY-Y1 pathway is crucial to regulating LTMR receptor-mediated sensations, enabling us to distinguish light touch from itching sensations. This could be operating together with other neurotransmitter pathways like glycine or GABA that act in shorter timescales, to provide both rapid and long-lasting inhibition of the mechanical itch pathways. One mechanism might be the production of other stimuli to ‘drown’ the mechanical itch, a phenomenon called counterstimulation.
The presence of this gated pathway for mechanical itch allows humans to enjoy the advantages of light touch. When this gating mechanism fails, chronic neuropathic itch may result. Patients with psoriasis, for instance, have lower levels of serum NPY which is related to an increased itch severity. Chronic spontaneous itching occurs in eczema, diabetes and some cancers. It is a very distressing condition that severely impacts the quality of life in the sufferers. Thus Y1 neuron signals could be targeted to relieve chronic itch.
The study was published in the journal Cell Reports on July 16, 2019.
Journal reference:
Spinal neuropeptide y1 receptor-expressing neurons form an essential excitatory pathway for mechanical itch. David Acton, Xiangyu Ren, Stefania Di Costanzo, Antoine Dalet, Steeve Bourane, Ilaria Bertocchi, Carola Eva, & Martyn Goulding. Cell Reports. DOI:https://doi.org/10.1016/j.celrep.2019.06.033. https://www.cell.com/cell-reports/fulltext/S2211-1247(19)30799-5