Researchers at the University of Oxford have conducted a study of UK healthcare workers showing how antibody responses to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection can wane over time, depending on age, previous symptoms and ethnicity.
SARS-CoV-2 is the agent responsible for the coronavirus disease 2019 (COVID-19) pandemic that continues to sweep the globe, threatening public health and devastating the economy.
The team’s longitudinal seroprevalence study of more than 3,200 healthcare workers found that immunoglobulin G (IgG) antibodies against the SARS-CoV-2 nucleocapsid protein waned within months, falling more quickly among young people and asymptomatic individuals.
The team also observed higher antibody titers and moderate evidence of longer antibody half-lives among Asian healthcare workers, compared with White healthcare workers.
“Relatively short-term antibody responses have two epidemiological consequences,” says Sheila Lumley and colleagues.
Firstly, seroprevalence testing could miss previous infections, particularly among younger people who had an asymptomatic or mild infection. Secondly, testing may be unable to determine whether SARS-CoV-2 has circulated historically, within a particular geographic region, for example.
“Antibody dynamics have significant implications for the course and management of pandemic,” say the researchers. “Ongoing longitudinal studies are required to determine the long-term kinetics of antibody-mediated response to SARS-CoV-2, and responses to re-exposure.”
A preprint version of the paper is available on the server medRxiv* server, while the article undergoes peer review.
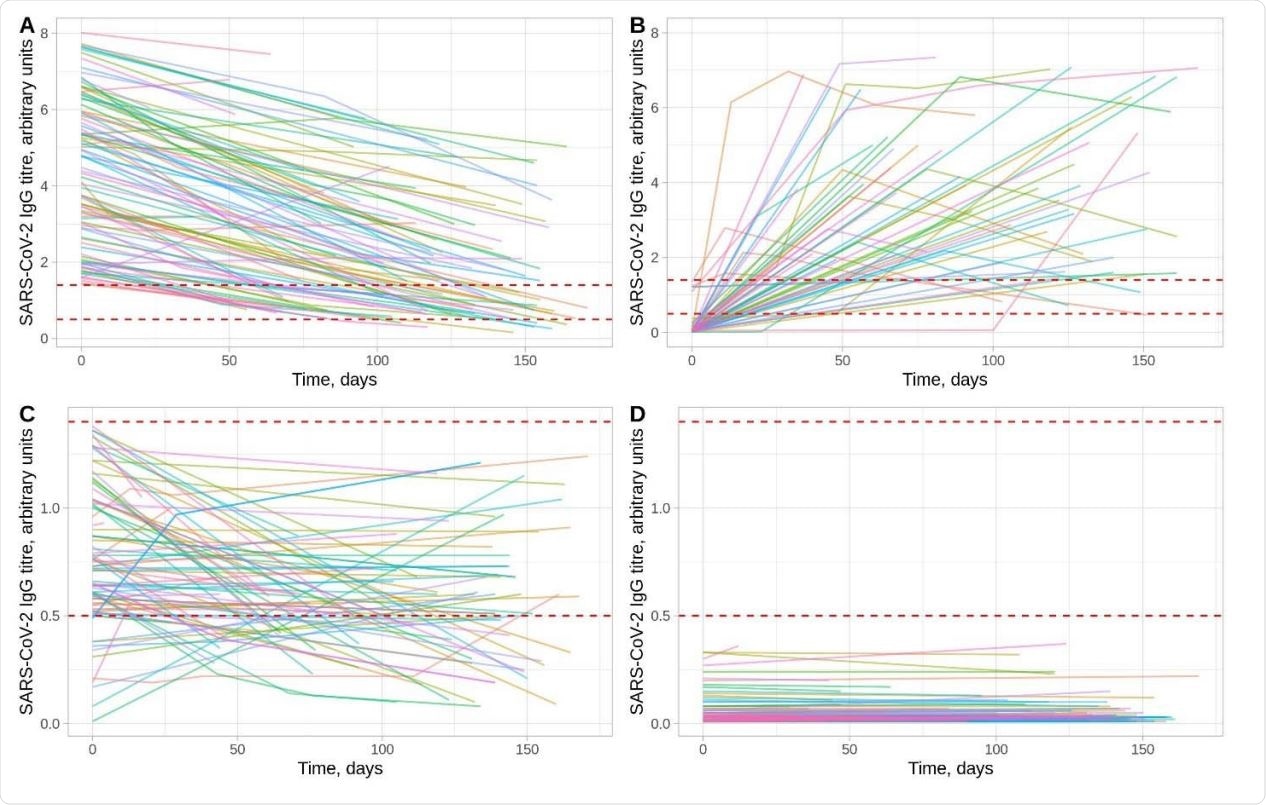
SARS-CoV-2 antibody trajectories in 3217 HCWs. Panels A and B show trajectories for HCWs with a positive result (≥1.40 arbitrary units) at some time. Panel A shows those whose first measurement was positive (n=466, only data from 100 randomly sampled individuals is shown to assist visualisation) and Panel B the remainder (n=56) in whom seroconversion was observed. Panel C shows those with a maximum titer that was equivocal (0.50-1.39, n=90). Panel D shows results from HCWs with a maximum titre that was negative (<0.50, n=2605, only data from 100 randomly sampled individuals is shown to assist visualization). The dashed lines indicate the thresholds for a positive and equivocal result, note the different y-axis scales in panels A and B versus panels C and D.
Previous estimates of antibody duration vary
Antibodies against SARS-CoV-2, which are generally detectable within 1 to 3 weeks, can be assayed to help estimate the proportion of a population that has been exposed to or infected with the virus.
Serological responses can also provide some information about the risk and severity of future infection.
“Most epidemiological outbreak models assume that SARS-CoV-2 infection leads to the development of post-infection immunity for a defined duration,” writes Lumley and colleagues.
However, measurable IgG antibodies against SARS-CoV-2 antigens do not always develop following infection, and previous estimates of the duration of antibody responses vary.
What did the current study involve?
Now, Lumley and the team have presented findings from a longitudinal seroprevalence study of 3,217 UK healthcare workers who underwent quantitative serial measurements of IgG antibodies against SARS-CoV-2 nucleocapsid protein.
Polymerase chain reaction (PCR) testing and antibody testing were offered to both symptomatic workers (from March 27th onwards) and asymptomatic workers (from April 23rd onwards) by the Oxford University Hospitals NHS Foundation Trust across all four of its teaching hospitals in Oxfordshire, UK.
“39% of HCWs [healthcare workers] were asymptomatic throughout, and therefore our data also represent an important contribution to the literature, which to date has mainly focused on trajectories following symptomatic infection,” write the researchers.
A Bayesian statistical model was used to investigate the trajectory of SARS-CoV-2 IgG antibody levels and any association with age, ethnicity, previous symptoms and gender.
What did the study find?
Among this cohort of working-age healthcare workers (up to 69 years), antibody levels peaked 24 days following the first positive PCR test, before then beginning to fall.
“By following quantitative antibody responses, we could separately analyze changes in initial antibody levels and rates of waning,” says the team.
Among 452 IgG-seropositive healthcare workers tested over a median of 121 days, the estimated mean antibody half-life was 85 days and the estimated mean time until loss of seropositivity was 137 days.
Higher maximum IgG levels were correlated with longer estimated IgG half-lives (a slower fall in IgG over time).
Associations between antibody duration and age, ethnicity, symptoms
The team reports that older age was associated with higher maximum IgG titers and longer IgG half-lives.
“This study highlights that sero-epidemiological surveys performed several months into this pandemic are likely to underestimate prior exposure in younger adults, as they tend to lose detectable antibody faster,” says the researchers.
Healthcare workers of Asian ethnicity had higher maximum antibody levels, compared with their White counterparts, along with marginal evidence for longer antibody half-lives.
Prior self-reported symptoms were associated with a higher maximum antibody level, but not with changes in antibody half-lives.
No associations were observed between gender and maximum antibody levels or half-lives.
What are the implications of the study?
“We demonstrate that the half-life of SARS-CoV-2 anti-nucleocapsid IgG antibody responses in a cohort of adult HCWs [healthcare workers] is 85 days and varies between individuals by age, ethnicity and prior symptom history,” say Lumley and team.
“The extent and duration of immunity to SARS-CoV-2 infection following COVID-19 and its association with antibody titers remains a key question to be answered.”
The researchers point out that the durability of immunity following infection or vaccination will dictate the overall course of the current COVID-19 pandemic.
“Longitudinal cohorts with baseline immunology are required to determine immune correlates of protection, to determine whether measurement of the current antibody status is enough to infer whether an individual have functional immunity or not, whether waning IgG titers are representative of waning immune protection, or whether protection remains even after an individual seroreverts,” concludes the team.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Lumley S, et al. The duration, dynamics and determinants of SARS-CoV-2 antibody responses in individual healthcare workers. medRxiv, 2020. doi: https://doi.org/10.1101/2020.11.02.20224824, https://www.medrxiv.org/content/10.1101/2020.11.02.20224824v1
- Peer reviewed and published scientific report.
Lumley, Sheila F, Jia Wei, Denise O’Donnell, Nicole E Stoesser, Philippa C Matthews, Alison Howarth, Stephanie B Hatch, et al. 2021. “The Duration, Dynamics, and Determinants of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Antibody Responses in Individual Healthcare Workers.” Clinical Infectious Diseases 73 (3): e699–709. https://doi.org/10.1093/cid/ciab004. https://academic.oup.com/cid/article/73/3/e699/6064824.