We are learning more about COVID-19 disease every day. Adults of any age with certain underlying medical conditions are at increased risk for severe illness from the virus that causes COVID-19.
COVID-19, caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, has a wide range of clinical manifestations and severity. Some factors contributing to an increased risk of severe COVID-19 include cardiovascular disease, obesity, chronic lung disease, and diabetes.
In low and medium-income countries (LMICs) infectious diseases are highly prevalent. Most notably, parasitic infections affect more than 2 billion people throughout the world. The effect of co-infection with parasites on the clinical features of COVID-19 is unknown.
Now, new research out of Mekelle, Ethiopia, examines the role of parasitic infestation on the prognosis of COVID-19. The research paper, which appears on the preprint server medRxiv*, shows that intestinal parasitosis is protective against severe COVID-19.
Study details
The researchers enrolled over 500 patients confirmed to have SARS-CoV-2 infection by polymerase chain reaction (PCR) testing. About 63% were male. The severity of COVID-19 was classified according to the criteria laid down by the World Health Organization (WHO), as asymptomatic, mild/moderate, severe, and critical.
The median age was 32 years. About 87% of the group had mild, asymptomatic or mild/moderate disease. Patients with severe disease were older and more symptomatic.
Intestinal parasitosis was detected by fresh stool examination for ova and parasites. They found that over half of them had parasite infestation of the intestine. The intensity of infection was reported as eggs per gram of feces, based on which the participants were classified as light, moderate and heavy.
About 52% of the cohort had one or more intestinal parasites, with a quarter of them having protozoal and 34% helminthic infestations, respectively. Symptomatic COVID-19 was significantly more common in the proportion of patients without parasitic infestation.
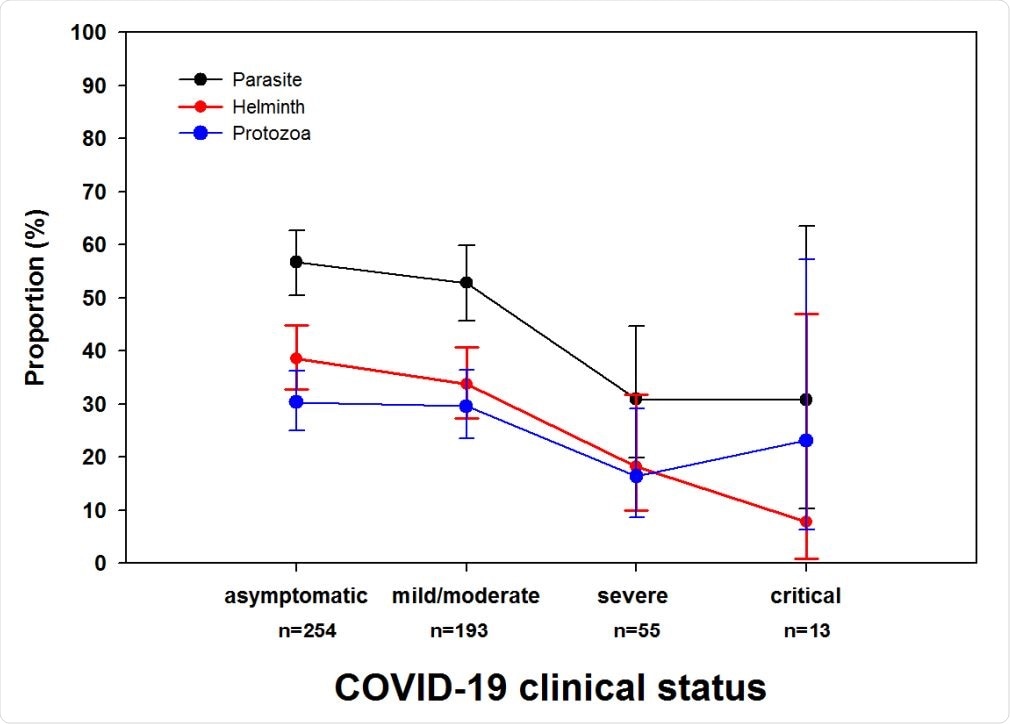
Proportion of parasites, protozoa and helminths among COVID-19 patients with asymptomatic, mild/moderate, severe and critical clinical presentation. P-values for trend (p=0.002, p=0.006 and p=0.196 for any parasite, helminth and protozoa, respectively).

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Higher incidence of severe COVID-19
The researchers found a negative correlation between parasitic co-infection and COVID-19 severity. Of the ~270 patients who had an infestation, only ~8% developed severe COVID-19, in contrast to almost a fifth of those without intestinal parasites (47/248).
When subclassified by the type of parasite, those who had non-protozoal parasites numbered ~370, compared to ~150 with protozoal infection. About 15% and 8% of patients in these two groups developed severe COVID-19.
When it came to helminthic infection, there were 174 and 341 patients with and without pre-existing helminthiasis. Only 6% of the former developed severe COVID-19 compared to ~17% of the latter.
After compensating for the effect of age and other co-existing illnesses, the presence of any parasitic infestation along with COVID-19 had a 60% decline in the odds of severe COVID-19. For those with protozoal or helminthic infestation, the adjusted odds of severe COVD-19 fell by 55% and 63%, respectively.
Parasitic infestations and SARS-CoV-2
LMICs faced the onset of the COVID-19 pandemic at a far different standard of living and healthcare compared to high-income countries. For one thing, infectious diseases, including parasitic infestations, are far more common in the former setting.
Such human parasites include hookworm tapeworm, roundworm, and whipworm, among helminthic parasites, as well as protozoa such as Entamoeba, Giardia, Toxoplasma, and Cryptosporidia.
Possible mechanisms
The presence of chronic parasitic infestation has been suggested to be an immunomodulatory factor, affecting the host response to other infections and the outcomes of such conditions. Such pre-existing infections could alter the immune response to SARS-CoV-2 as well.
Parasites that cause chronic infections are known to enhance T helper cell responses and induce predominant regulatory (Treg) responses. This leads to a Th2-biased response, which may compensate for the Th1-response known to be responsible for the severe and exaggerated state of inflammation found in severe COVID-19.
Secondly, generalized immunomodulation may occur in response to the host's gut microbiome changes due to chronic parasitic infestations. Both factors may alter the outcome of SARS-CoV-2 infection in such patients.
An earlier animal study demonstrated the protective role of enteric helminthiasis in viral respiratory infections via the interaction of the parasites with gut microbiota.
Interestingly, recent studies indicate a lower incidence of COVID-19 in regions with significant helminthiasis, schistosomiasis or malaria. However, the lack of evidence on the actual effects of co-existing parasitoses on COVID-19 severity drove the current study.
What are the implications?
The presence of any parasitic co-infestation reduced the risk of severe COVID-19, while the presence of hypertension, chronic renal disease, and older age drove up the odds of severe disease. Even after these comorbidities were adjusted for, patients with parasitic infestation had significantly lower odds of severe COVID-19.
An interesting sidelight from this study is the dramatically lower chances of having a non-communicable disease (NCD) in COVID-19 patients with parasite co-infections. The odds of having NCDs were lowered by a respectable 48% and an impressive 74%, if the COVID-19 patient had parasites or helminths, respectively.
"Our results suggest that co-infection with parasitic co-infection appears to be associated with reduced COVID-19 severity. The results suggest that parasite-driven immunomodulatory responses may mute hyperinflammation associated with severe COVID-19."
This may account for the observation that COVID-19 has a low infection fatality rate in LMICs. Further study should follow up these findings in other low-income settings and explore the kind of alterations brought about in the gut microbiota due to these chronic parasitic infections. Such knowledge could contribute to new therapeutic and preventive measures to contain the pandemic.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Gebrecherkos, T. et al. (2021). Effect of co-infection with parasites on severity of COVID-19. medRxiv preprint. doi: https://doi.org/10.1101/2021.02.02.21250995, https://www.medrxiv.org/content/10.1101/2021.02.02.21250995v1
- Peer reviewed and published scientific report.
Wolday, Dawit, Teklay Gebrecherkos, Zekarias Gessesse Arefaine, Yazezew Kebede Kiros, Atsbeha Gebreegzabher, Geremew Tasew, Mahmud Abdulkader, et al. 2021. “Effect of Co-Infection with Intestinal Parasites on COVID-19 Severity: A Prospective Observational Cohort Study.” EClinicalMedicine 39 (September). https://doi.org/10.1016/j.eclinm.2021.101054. https://www.thelancet.com/journals/eclinm/article/PIIS2589-5370(21)00334-5/.