Researchers in South Africa have submitted evidence that suggests that vaccines modeled on the South African variant of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) would not only be effective on that strain but would also function well against other variants.
The team’s analysis found that antibodies from patients with the 501Y.V2 SARS-CoV-2 variant, also known as the B.1.351, first identified in South Africa in October 2020, exhibited good cross-reactivity with viruses from the first wave and effectively neutralized the Brazil variant.
In the last quarter of 2020, three SARS-CoV-2 variants were identified that were considered cause for concern. All three exhibited an immune escape from the current vaccines: a UK strain, a Brazil strain, and the South African strain, which appeared to be of the most concern. This study appears to have provided a new hope going forwards.
“The [South African] variant is the one which has been demonstrated to have the most immune evasion to date,” commented American scientist Eric Topol on the research paper. “That’s why this [preprint] is very good news.”
A pre-print version of the research paper is available to read in full on the bioRxiv* server.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
What did the study involve?
Between December 31st, 2020 and January 15th, 2021, Penny L Moore and colleagues took blood samples from 89 COVID-19 patients admitted to the Groote Schuur Hospital in Cape Town, South Africa. Of these, they sequenced the infections of 28 patients, confirming all to have the 501Y.V2 variant. Prior to the study, none of the patients reporting having COVID symptoms or infections from the first wave.
These samples were compared with samples taken from a cohort of 62 patients hospitalized in Pretoria, South Africa, between May and September of 2020 (all infected with and during the first wave). The team found no difference in the rate at which antibodies neutralized the two variants.
Previously, the team had reported that antibodies from plasma belonging to individuals infected with the original variant were less effective at neutralizing the South African variant.
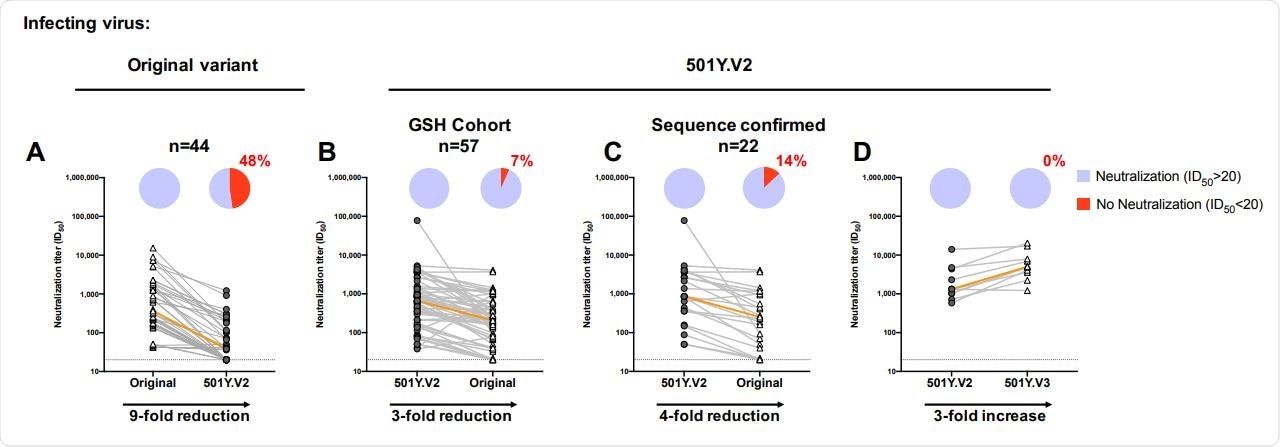
Neutralizing antibodies elicited by 501Y.V2 infection are more cross-reactive than those from patients infected with the original variant. (A) Plasma samples from patients infected with the original variant and (B-C) 501Y.V2-infected GSH cohort samples were compared for their neutralization cross-reactivity against other variants (n=57). In (C), the analysis was limited to those samples where sequencing confirmed infection by 501Y.V2 (n=22). (D) A subset of samples (n=10)
Here, they performed the same experiment but in reverse; the team assessed the cross-reactivity of antibodies for the South African against the original and Brazil variant.
They found that neutralization was far more successful against the original variant than vice versa, but also that the antibodies were even more potent when used against the Brazilian variant.
With this, the team has concluded from their data that vaccines based on the South African variant, rather than the original variant, would be far more effective in neutralizing the virus.
“If we design a vaccine based on the 501Y.V2 variant, it may be able to protect us from these different variants,” tweeted one of the researchers involved, Thandeka Moyo-Gwete. Moyo-Gwete is a Senior Medical Scientist at the National Institute for Communicable Diseases in South Africa and one of the lead authors of the paper. “We could also think along the lines of using it as a booster vaccine. Lots of possibilities and really exciting times for COVID vaccine.”
What happens next?
Some similar studies have found that antibodies based on the UK strain of the virus were not effective against the South African variant. The researchers suspect that it may be that vaccines based on the latter may be more effective as the viruses are, convergently, more structurally similar to each other than other variants.
The authors suggest that further study with other variants, such as the Brazil strain, might yield additional important information.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Penny L Moore, et al. SARS-CoV-2 501Y.V2 (B.1.351) elicits cross-reactive neutralizing antibodies. bioRxiv, 2021. doi: https://doi.org/10.1101/2021.03.06.434193, https://www.biorxiv.org/content/10.1101/2021.03.06.434193v1
- Peer reviewed and published scientific report.
Moyo-Gwete, Thandeka, Mashudu Madzivhandila, Zanele Makhado, Frances Ayres, Donald Mhlanga, Brent Oosthuysen, Bronwen E. Lambson, et al. 2021. “Cross-Reactive Neutralizing Antibody Responses Elicited by SARS-CoV-2 501Y.V2 (B.1.351).” New England Journal of Medicine 384 (22): 2161–63. https://doi.org/10.1056/nejmc2104192. https://www.nejm.org/doi/10.1056/NEJMc2104192.