Many of the non-pharmaceutical interventions that have been put in place since the beginning of the COVID-19 pandemic, such as travel restrictions and business closures, are being relaxed as vaccines and testing become more widely available. In doing so, experts have highlighted the risk that local outbreaks are likely to occur, and early reports have observed such spikes.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Developing the model
Non-pharmaceutical interventions have proved extremely effective when properly applied. New Zealand, Australia, and Sweden often are cited as good examples of well-implemented travel policies, lockdown procedures, and thorough contact tracing.
The distribution of vaccines, too, has proven highly effective in reducing the proliferation of and presentation of symptoms of SARS-CoV-2, and countries that have undertaken widespread vaccination programs have seen a significant drop in the per-day cases of COVID-19, allowing some travel restrictions to be lifted.
However, vaccines do not entirely prevent transmission, and the entirety of the population is yet to be immunized, starkly so in less-developed countries. Some studies suggest that mixed populations of immunized and non-immunized individuals are more likely to provide ideal conditions for the development of SARS-CoV-2 escape mutants that evade vaccine-induced immunity. The introduction of such mutants from abroad becomes an increasing threat as travel restrictions are lifted.
Branching process mathematical models are frequently used to describe and predict pathogen transmission in epidemiology, using the common formula:
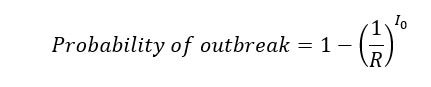
Where R is the reproduction number of the virus, and I0 is the number of newly introduced infectious cases. This formula assumes that the reproduction number is a fixed value. However, the introduction of vaccines alters this value by lowering transmissibility and pathogenicity. To account for this factor, the group developed a model that adjusts reproduction number based on the number of individuals vaccinated:
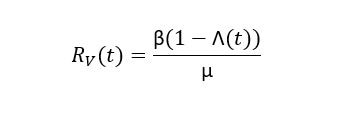
Where ß represents the number of new infections in a set period, 1/µ being the duration of infectiousness, and Λ the effective immunity rate by vaccination, accounting for the expected rate of immunity from first or second doses. Therefore Rv(t) is the predicted number of secondary infections generated by a single infection individual in the population at time t, representing “instantaneous transmission.” If there were no vaccinated members of the population (Λ(t)=0), then R would be set to the original wildtype reproduction number.
Analyzing the data
Utilizing data regarding vaccination and infection rates from the Isle of Man and Israel, the group plotted the outbreak risk of each location, accounting for immunity developed by vaccination. At the ordinary wildtype reproduction number of 3, being gradually reduced by vaccination as discussed, the risk of outbreak reduced from over 0.6 in both locations in January 2021, lowering to around 0.5 and 0.25 in the Isle of Man and Israel, respectively, by May 2021, the end-point of data collection. Past this point, based on predicted vaccination rates, each location is expected to lower to a respective outbreak risk of 0.0 and 0.2 by August 2021. The massive population size discrepancy between these locations accounts for the much more rapid change seen in the Isle of Man, while Israel implemented the vaccination program earlier.
Setting the reproductive rate of the virus to 5 instead of 3, to represent a highly pathogenic escape mutant, instead saw these numbers at around 0.5 by this time, demonstrating the risk posed to even heavily vaccinated populations by the introduction of these lineages. As these calculations do not account for non-pharmaceutical interventions, they assume that they have been completely lifted. Thus the data indicates that the lifting of restrictions can go ahead safely in the absence of highly pathogenic escape mutants, for which new vaccines may need to be developed.
The group state that future outbreaks are likely to be smaller in scale than previously observed for COVID-19 before vaccines were widely administered, presuming that no escape mutant entirely avoids engineered immunity. However, background immunity was not considered by this model, and would likely contribute, particularly in countries that have experienced high case numbers.
Additionally, immunity acquired by any method will fade over time, and the risk of foreign variants sparking an outbreak will remain even once the population is fully vaccinated. A global approach that would reduce the number of mutant variants transported around the world, giving them a chance to develop further variation, is recommended by the authors. This should be combined with an emphasis on surveillance of inbound passengers and which virus lineages they may carry.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources