Researchers in the UK have conducted a study showing that infection with the B.1.1.7 (Alpha) lineage of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) may be associated with an increased risk of death or intensive care unit (ITU) admission among women, compared with non-B.1.1.7 infection.
The SARS-CoV-2 virus is the agent responsible for the ongoing coronavirus disease 2019 (COVID-19) pandemic that has now claimed the lives of more than 3.93 million people globally.
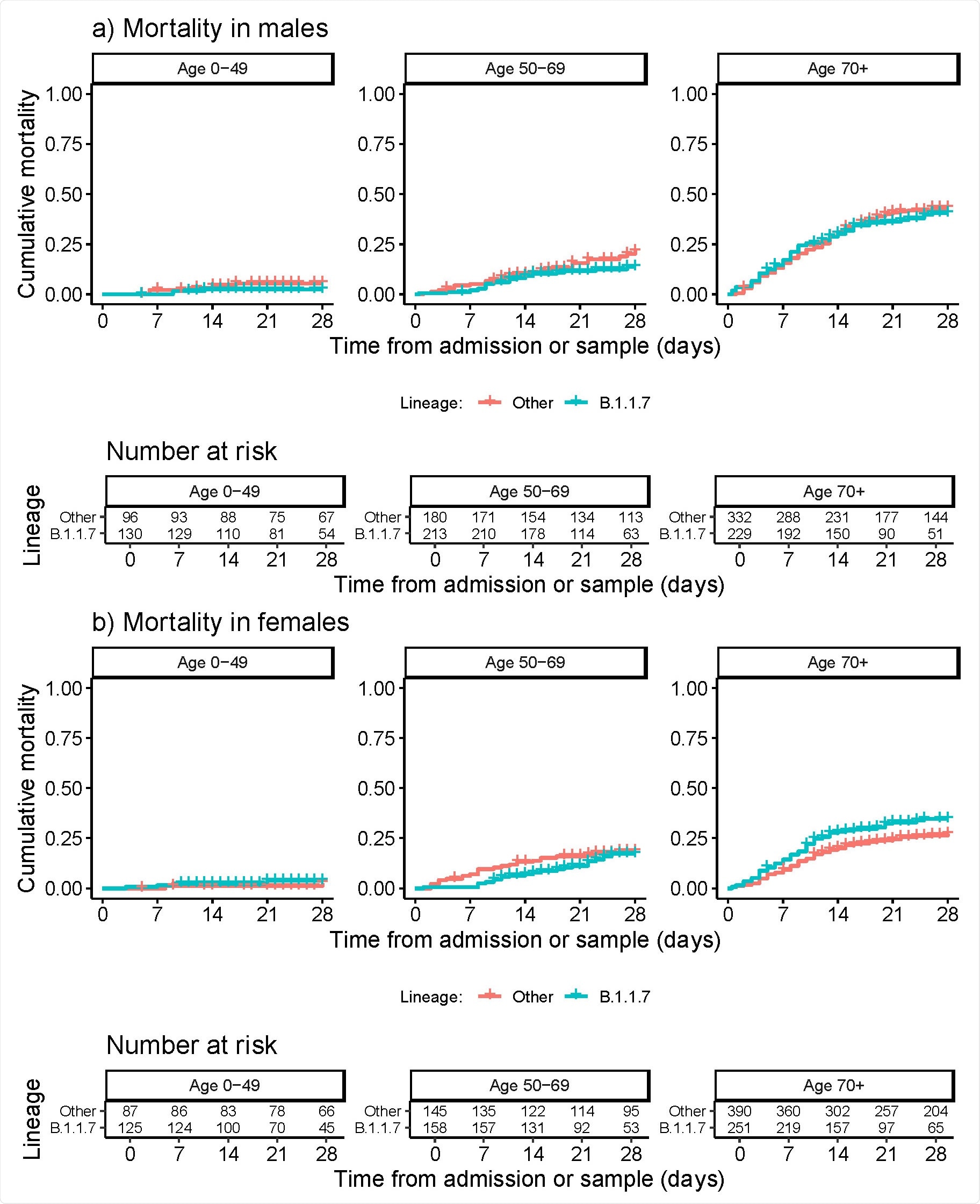
The study of more than 2,300 patients hospitalized with SARS-CoV-2 infection found that the overall risk of mortality and ITU admission was unchanged for patients infected with B.1.1.7 versus previously circulating viral variants.
However, the sex-specific analysis revealed an increased risk of both mortality and ITU admission associated with the variant among females but not males.
Oliver Stirrup from University College London and colleagues say the findings provide the most extensive dataset so far on disease severity among hospitalized patients infected with B.1.1.7.
“The findings may have implications for hospital practice and public health policy, both in the UK and in other countries where lineage B.1.1.7 is now dominant or spreading,” they write.
The team says continued large-scale sequencing of SARS-CoV-2 cases linked to data on patient characteristics and outcomes is needed to generate timely information regarding the associations between different viral lineages and disease severity.
A pre-print version of the research paper is available on the medRxiv* server, while the article undergoes peer review.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Little is known about the impact of B.1.1.7 among hospitalized patients
The emergence of the B.1.1.7 (Alpha) lineage of SARS-CoV-2 in the Southeast of England has been associated with an estimated 70% increase in community transmission, compared with previously circulating variants. The lineage is the dominant viral strain in the UK and has also been detected in more than 120 countries.
The B.1.1.7 lineage has acquired an exceptionally large number of mutations in a relatively short period of time.
However, the potential impact this large number of mutations may have on the virulence of SARS-CoV-2 has mainly been investigated using data from samples collected in the community, says Stirrup and colleagues.
“There are few data on the impact of B.1.1.7 infection as compared with other variants on disease outcomes in hospitalized patients,” says the team.
What did the researchers do?
The researchers investigated potential associations between infection with B.1.1.7 and the outcomes of mortality and ITU admission among patients admitted to hospital with COVID-19 and patients with hospital-onset COVID-19 infections (HOCIs) from eight hospitals participating in the COG-UK-HOCI study.
The first samples collected from all inpatients between November 16th, 2020, and January 10th, 2021, were sequenced, and data on age, sex, co-morbidities, care home residence, pregnancy, and ethnicity were obtained.
Sequences were obtained for 2,341 inpatients (HOCI cases = 786), and analysis of clinical outcomes was carried out for 2,147 inpatients for whom all data were available.
What did the study find?
The overall risk of mortality and ITU admission was no greater among patients infected with B.1.1.7 than among those infected with previously circulating viral variants.
However, multivariable Cox regression analysis revealed that female patients infected with B.1.17 were at a 30% increased risk of mortality and an 82% increased risk of ITU admission compared with non-B.1.1.7 infection.
No such increase in relative risk for either outcome was observed among male patients infected with B.1.1.7.
SARS-CoV-2 lineage B.1.1.7 is associated with greater disease severity among hospitalised women but not men
The sex-specific difference could be explained by physiological differences
Stirrup and colleagues suggest that this impact of B.1.1.7 on females, but not males, may explain physiological differences.
For example, females express higher levels of the host cell receptor angiotensin-converting enzyme (ACE2) that SARS-CoV-2 binds to as the initial stage of the infection process.
“Lineage B.1.1.7 has mutations that increase binding of the viral spike protein to ACE2, thereby providing a plausible mechanism by which the new variant might have a differential effect on disease severity in males and females,” writes the team.
What do the authors advise?
The researchers say the findings may have implications for hospital practice and public health policy, both in the UK and other countries where lineage B.1.1.7 has become dominant or is spreading.
“The dominance of lineage B.1.1.7 in the UK precludes ongoing comparison with earlier non-B.1.1.7 variants, and there is now concern regarding the spread of other lineages in the UK and elsewhere,” says Stirrup and colleagues.
“There is a need for ongoing large-scale sequencing of SARS-CoV-2 cases linked to data on patient characteristics and outcomes in order to generate timely information regarding the associations between viral lineages and disease severity,” concludes the team.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Stirrup O, et al. SARS-CoV-2 lineage B.1.1.7 is associated with greater disease severity among hospitalized women but not men. medRxiv, 2021. doi: https://doi.org/10.1101/2021.06.24.21259107, https://www.medrxiv.org/content/10.1101/2021.06.24.21259107v1
- Peer reviewed and published scientific report.
Stirrup, Oliver, Florencia Boshier, Cristina Venturini, José Afonso Guerra-Assunção, Adela Alcolea-Medina, Angela Beckett, Themoula Charalampous, et al. 2021. “SARS-CoV-2 Lineage B.1.1.7 Is Associated with Greater Disease Severity among Hospitalised Women but Not Men: Multicentre Cohort Study.” BMJ Open Respiratory Research 8 (1): e001029. https://doi.org/10.1136/bmjresp-2021-001029. https://bmjopenrespres.bmj.com/content/8/1/e001029.