The efforts of many researchers have uncovered multiple aspects of coronavirus disease 2019 (COVID-19) immunity. However, most of this information has been derived from individuals with severe disease, whereas most infections are asymptomatic and mild.
It is necessary to understand whether prior infection with SARS-CoV-2 contributes to protection against reinfection. It is known that even after asymptomatic or mild infection, an antibody response occurs in most cases. Notably, antibody titers do appear to wane noticeably over time.
The cell-mediated aspect of adaptive immunity is known to be more durable. For instance, after the SARS outbreak in 2003, T-cell responses to the virus were detectable up to 17 years later.
Some research indicates a lower T-cell response to mild infection as compared to more severe COVID-19. The CD4+ T-cell response is more prominent than the CD8+ T-cells in mild-to-moderate infection; however, in critical illness, the CD4+ T-cell responses are often weak.
In children, who typically show mild or asymptomatic infection, not much is known about cellular immunity. The T-cell response is vital for long-term protection against reinfection with the same agent, as these cells promote a quick and robust triggering of the immune response when the host is exposed again to the same agent.
The current study provides more information as to whether T-cell responses in children are strong enough to be useful in preventing the occurrence and spread of COVID-19 following mild SARS-CoV-2 infection.
About the study
The researchers of the current study compared the specific T-cell responses against SARS-CoV-2 in 27 adults and 24 children with the infection, versus unexposed matched individuals. Blood samples were taken at a median of 12.5 days and 8 days from symptom onset in adults and children, respectively.
The second set of blood samples was collected at T2, which was defined as 10-14 days from the first sample (T1). A third set of blood samples was collected from adults only at 4-6 weeks from T1 (T3).
The researchers found that 83% of children exhibited gamma-interferon (IFN-ɣ)-producing T-cells reactive to the SARS-CoV-2 spike protein, while all infected adults showed this response. In contrast, none of the unexposed children, and only 1/12 unexposed adults, showed IFN-ɣ+ T-cells.
Adults with moderate symptoms had a 2-6-fold higher proportion of SARS-CoV-2-specific IFN-ɣ+ T-cells as compared to those with mild symptoms at T1. At T3, such a difference in the response was found only after both the nucleocapsid protein and the spike protein were used to stimulate the T-cells, or if the inactivated whole virus was used to stimulate the T-cells at both later time points.
In the infected cohort, children exhibited lower frequencies of these cells than the adults at 18 v. 62 spot-forming units (SFU)/2.105 peripheral blood mononuclear cells (PBMCs), respectively at T1. The higher IFN-ɣ+ T-cell frequencies in adults were driven mainly by the moderately symptomatic adults, though mild COVID-19 in adults was also linked to a small increase in frequencies compared to children.
The T-cell response elicited by prior seasonal endemic coronavirus (HCoV-OC43) spike antigen in children was low, with only a small difference between exposed and unexposed children at T2. In the corresponding group of adults, there was no difference in T-cells reactive to this virus in either group,
Among the SARS-CoV-2-infected cohort, IFN-ɣ+ T-cells were 4- to 60-fold lower in frequency when stimulated with CoV-OC43 spike than when the SARS-CoV-2 antigens were used. With the SARS-CoV-2 spike, however, CD4+ T-cell responses predominated over CD8+ T-cells, with both infected children and adults showing higher frequencies of activated CD4+ T-cells.
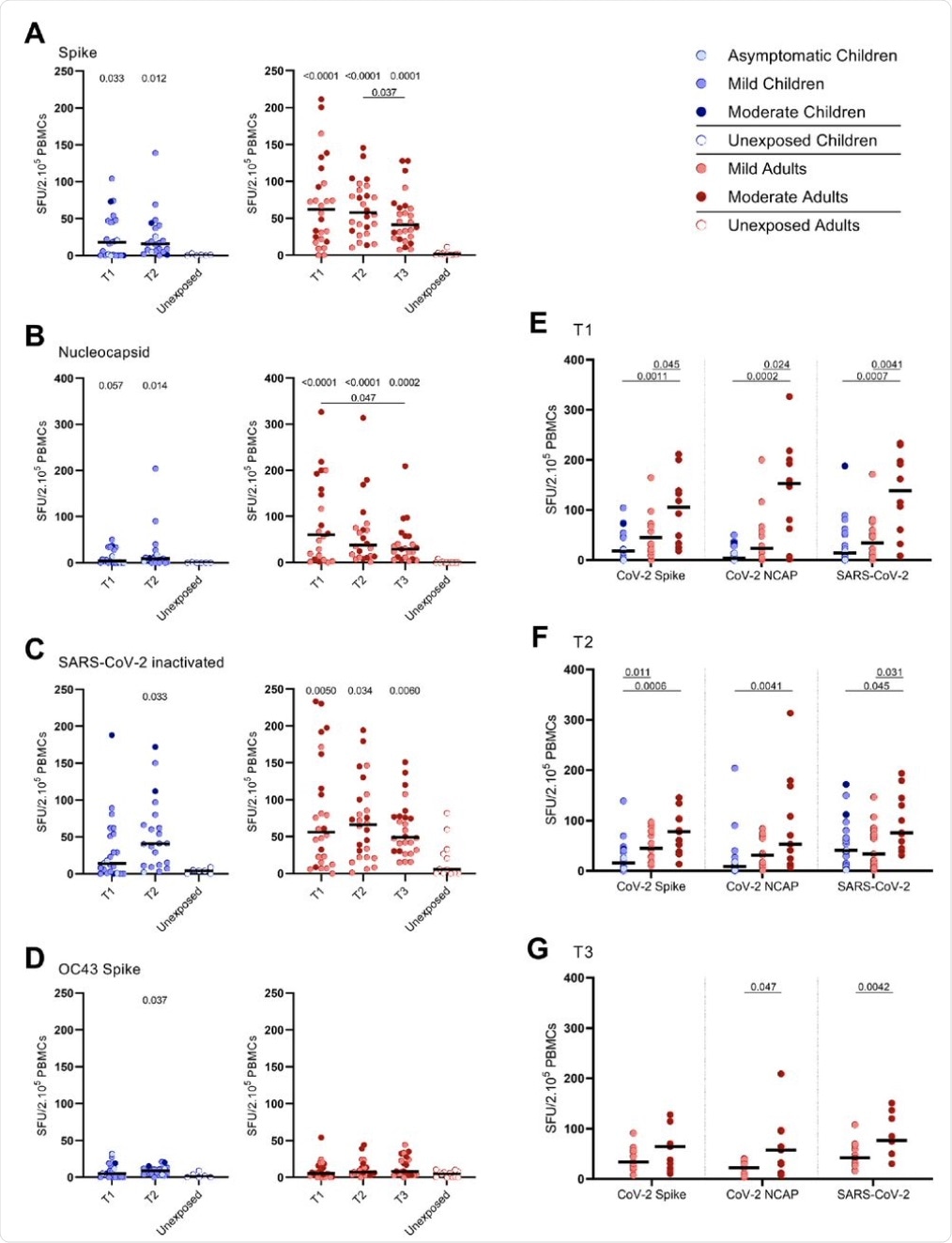 SARS-CoV-2-specific IFN-γ+ T cell response in infected children and infected adults (mild and moderate cases) versus unexposed healthy controls over time after infection. Dot plots summarizing the frequencies of IFN-γ-producing cells responding to SARS-CoV-2 and HCoV-OC43 antigens for (A-D) children (left panel), adults (right panel), over time after infection, and compared to unexposed adults/children (ELISPOT assay). Frequencies of IFN-γ-producing cells responding to (A) set of overlapping of peptides of SARS-CoV-2 spike protein, (B) set of overlapping peptides of SARS-CoV-2 nucleocapsid protein, (C) inactivated SARS-CoV-2, and (D) set of overlapping peptides of HCoV-OC43 spike protein. (E) Comparison of IFN-γ-producing cells derived from infected children, mild symptomatic SARS-CoV-2-infected adults versus adult COVID-19 patients with moderate symptoms at T1, (F) at T2, and (G) mild symptomatic SARS-CoV-2-infected adults versus adult COVID-19 patients with moderate symptoms at T3. Each dot represents one subject. Bars indicate the median of spot-forming units per 200,000 PBMCs. SFU, spot-forming unit. (A-D) P values related to comparisons with the unexposed controls are listed at the top of the graph, above the corresponding group for comparison. For unpaired comparisons, Mann-Whitney U test (two-group comparisons) (mild adults versus moderate adults) or Kruskal-Wallis rank-sum test with Dunn’s posthoc test for multiple comparisons were used (children versus mild adults versus moderate adults; unexposed versus infected children or adults at T1 versus T2 versus T3). Differences between paired data were compared using the Wilcoxon signed-rank test (for comparison of two paired groups) (infected children at T1 versus T2) or the Friedman test with Dunn’s multiple comparison tests (infected adults at T1 versus T2 versus T3). Statistically significant comparisons are indicated, with P values < 0.05 considered significant. T1, first timepoint of sampling for adults median 12.5 days and children median 8 days post-symptom onset; T2, 10-14 days after T1; T3, 4-6 weeks after T1.
SARS-CoV-2-specific IFN-γ+ T cell response in infected children and infected adults (mild and moderate cases) versus unexposed healthy controls over time after infection. Dot plots summarizing the frequencies of IFN-γ-producing cells responding to SARS-CoV-2 and HCoV-OC43 antigens for (A-D) children (left panel), adults (right panel), over time after infection, and compared to unexposed adults/children (ELISPOT assay). Frequencies of IFN-γ-producing cells responding to (A) set of overlapping of peptides of SARS-CoV-2 spike protein, (B) set of overlapping peptides of SARS-CoV-2 nucleocapsid protein, (C) inactivated SARS-CoV-2, and (D) set of overlapping peptides of HCoV-OC43 spike protein. (E) Comparison of IFN-γ-producing cells derived from infected children, mild symptomatic SARS-CoV-2-infected adults versus adult COVID-19 patients with moderate symptoms at T1, (F) at T2, and (G) mild symptomatic SARS-CoV-2-infected adults versus adult COVID-19 patients with moderate symptoms at T3. Each dot represents one subject. Bars indicate the median of spot-forming units per 200,000 PBMCs. SFU, spot-forming unit. (A-D) P values related to comparisons with the unexposed controls are listed at the top of the graph, above the corresponding group for comparison. For unpaired comparisons, Mann-Whitney U test (two-group comparisons) (mild adults versus moderate adults) or Kruskal-Wallis rank-sum test with Dunn’s posthoc test for multiple comparisons were used (children versus mild adults versus moderate adults; unexposed versus infected children or adults at T1 versus T2 versus T3). Differences between paired data were compared using the Wilcoxon signed-rank test (for comparison of two paired groups) (infected children at T1 versus T2) or the Friedman test with Dunn’s multiple comparison tests (infected adults at T1 versus T2 versus T3). Statistically significant comparisons are indicated, with P values < 0.05 considered significant. T1, first timepoint of sampling for adults median 12.5 days and children median 8 days post-symptom onset; T2, 10-14 days after T1; T3, 4-6 weeks after T1.
SARS-CoV-2 infection in children was linked to a lower activated specific CD4+ T-cell response than in adults at 0.04% and 0.21%, respectively, of the total CD4+ T-cells. Moderate symptoms were associated with a higher frequency of activated CD4+ T-cells specific to this virus, regardless of whether spike or nucleoprotein antigen was used for stimulation as compared to mild symptoms.
In children, the activated CD4+ T-cells carried the effector memory cell phenotype. Moreover, the frequency of activated CD4+ T-cells and IFN- ɣ+ T-cell frequency showed moderate associations upon stimulation with the SARS-CoV-2 spike and nucleoprotein at T2. However, in the first samples, this correlation was seen only if the whole virus was used for stimulation.
 Frequencies of activated CD4+ T cells of infected children and infected adults (mild and moderate cases) versus unexposed healthy controls over time after infection. Dot plots summarizing the percentages of CD25+/CD137+ activated CD4+ T cells responding to SARS-CoV-2 and HCoV-OC43 antigens for (A-D) children (left panel), adults (right panel), over time after infection, and compared to unexposed adults/children. Percentages of CD25+/CD137+ activated CD4+ T cells responding to (A) set of overlapping peptides of SARS-CoV-2 spike protein, (B) set of overlapping peptides of SARS-CoV-2 nucleocapsid, (C) inactivated SARS-CoV-2, and (D) set of overlapping peptides of HCoV-OC43 spike protein. (E) Comparison of IFN-γ-producing cells derived from infected children, mild symptomatic SARS-CoV-2-infected adults versus adult COVID-19 patients with moderate symptoms at T1, (F) at T2, and (G) mild symptomatic SARS-CoV-2-infected adults versus adult COVID-19 patients with moderate symptoms at T3. (H) Immunophenotyping at the single-cell level showed the different memory subsets within the SARS-CoV-2-specific activated CD4+ T cells from infected adults.
Frequencies of activated CD4+ T cells of infected children and infected adults (mild and moderate cases) versus unexposed healthy controls over time after infection. Dot plots summarizing the percentages of CD25+/CD137+ activated CD4+ T cells responding to SARS-CoV-2 and HCoV-OC43 antigens for (A-D) children (left panel), adults (right panel), over time after infection, and compared to unexposed adults/children. Percentages of CD25+/CD137+ activated CD4+ T cells responding to (A) set of overlapping peptides of SARS-CoV-2 spike protein, (B) set of overlapping peptides of SARS-CoV-2 nucleocapsid, (C) inactivated SARS-CoV-2, and (D) set of overlapping peptides of HCoV-OC43 spike protein. (E) Comparison of IFN-γ-producing cells derived from infected children, mild symptomatic SARS-CoV-2-infected adults versus adult COVID-19 patients with moderate symptoms at T1, (F) at T2, and (G) mild symptomatic SARS-CoV-2-infected adults versus adult COVID-19 patients with moderate symptoms at T3. (H) Immunophenotyping at the single-cell level showed the different memory subsets within the SARS-CoV-2-specific activated CD4+ T cells from infected adults.
Such a correlation was observed in adults at all three time points and with both antigens. Alternatively, stimulation with the whole virus led to correlated increases in the frequency of both these cell types at T1 and T2.
Cytokine release was marginally higher after spike stimulation in SARS-CoV-2 infected children, including interleukin 2 (IL-2) and IL-10, but somewhat higher in adults. Significantly increased levels were found only in moderately ill adults.
Antibodies to the spike protein were found in over 75% of infected children, even in 2/4 of children without detectable T-cell immunity. The presence of immunoglobulin M (IgM) was found in 88%, IgG in 79%, and IgA in 75%. Conversely, all adults showed IgM, 96% IgG, and 89% IgA antibodies.
In the early phase of infection, all children showed correlations between the antibody titers and the frequency of IFN-ɣ+ T-cells against the spike antigen. In adults, this correlation occurred with IgM and IgG during the second and third time points, while for IgA, the two were related only at T3.
Implications
This report shows for the first time that children and adults with mild COVID-19 show higher IFN-ɣ+ T-cell responses on stimulation with the SARS-CoV-2 spike, nucleoprotein, or other viral antigens. Children had lower frequencies of these cells compared to adults, perhaps because they had mostly asymptomatic or mild infections.
This may indicate that viral clearance in children involves other components of the immune response, especially involving the innate immune system, thus accelerating the process.
Earlier studies have shown the importance of T-cell responses in protecting against disease progression, as critical COVID-19 is linked to lower specific CD4+ T-cell responses. These cells continue to be detected for more than eight months after infection, regardless of whether the infection caused mild or severe symptoms.
The current study supports sustained T-cell immunity after a mild infection, though activated specific CD4+ T-cells are lower in infected children than in adults, both acute and memory cells. The correlation between the activated cells and the SARS-CoV-2-specific IFN-ɣ+ T-cells indicates that the IFN-ɣ originated from virus-specific activated CD4+ cells.
The additional presence of IL-2 also suggests a beneficial type 1 T helper (Th1) effector memory cell response. The low CD8+ T-cells could be due to their migration into the infected tissue to destroy the infected cells. This reduced response may also be due to the fact that these cells do not respond to the specific antigens used in this study, but rather to smaller peptides or the live virus.
The authors point out that this study did not provide proof that children were protected against COVID-19 by prior immunity to the seasonal endemic coronaviruses. The effector memory T-cell response after mild infection with SARS-CoV-2 may help provide partial protection, however, as well as limit the viral load and therefore the spread of the virus.
The authors of the current study plan to continue monitoring how long and in what manner the T-cell response will persist in an effort to provide insights into protective immunity against reinfection.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Kaajik, P., Pimentel, V. O., Emmelot, M. E., et al. (2021). Children And Adults with Mild COVID-19 Symptoms Develop Memory T Cell Immunity to SARS-Cov-2. medRxiv. doi:10.1101/2021.09.10.21263333. https://www.medrxiv.org/content/10.1101/2021.09.10.21263333v1.
- Peer reviewed and published scientific report.
Kaaijk, Patricia, Verónica Olivo Pimentel, Maarten E. Emmelot, Martien C. M. Poelen, Alper Cevirgel, Rutger M. Schepp, Gerco den Hartog, et al. 2022. “Children and Adults with Mild COVID-19: Dynamics of the Memory T Cell Response up to 10 Months.” Frontiers in Immunology 13 (February). https://doi.org/10.3389/fimmu.2022.817876. https://www.frontiersin.org/articles/10.3389/fimmu.2022.817876/full.