The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infects hosts through its interaction with the angiotensin-converting enzyme 2 (ACE-2) receptor, expressed by a number of cells, including cardiac myocytes.
As a result, the novel coronavirus disease 2019 (COVID19) impacts the heart and can lead to heterogenous cardiovascular manifestations that contribute to the high case fatality rate. Some of the cardiovascular events associated with COVID-19 include acute myocardial infarction (MI), stress cardiomyopathy, myocarditis, heart failure (HF), pulmonary embolism (PE), and cardiac arrhythmias.
Study: Risk of Cardiovascular Events after Covid-19: a double-cohort study. Image Credit: Irina Shatilova / Shutterstock.com

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Background
Myocardial injury can be caused by direct injury to cardiac myocytes due to COVID-19 and as a result of secondary effects from the systemic inflammation and hypercoagulable state seen in acute infection. Therefore, COVID-19 patients with known cardiovascular disease (CVD) and other risk factors like age, hypertension, diabetes, obesity, kidney disease, and respiratory system disorders are more likely to require critical care and have a higher mortality rate.
Long COVID, which is also known as post-acute COVID, is the persistence of symptoms or complications after the end of the acute phase of infection. However, the long-term impacts of COVID-19 on cardiovascular outcomes remain unknown. Furthermore, the causal role of asymptomatic and symptomatic SARS-CoV-2 infections in precipitating cardiovascular events has yet to be clarified.
The objective of a recent study published on the medRxiv* preprint server was to determine the absolute risk, relative conditional risk, and causal inference effect of symptomatic and asymptomatic SARS-CoV-2 infection on post-acute cardiovascular events and all-cause mortality.
About the study
The current retrospective double-cohort study was conducted at the Oregon Health & Science University (OHSU). The study was approved by the OHSU Institutional Review Board (IRB) and all results were reported using the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Herein, the electronic medical records (EMRs) of adults with a positive or negative COVID-19 test result were included. A random sample of records was extracted from March to September 2020.
The primary outcome was a composite of cardiovascular death, acute HF, acute coronary syndrome (ACS) including ST-Segment Elevation Myocardial Infarction (STEMI), non-STEMI (NSTEMI), or unstable angina, as well as incident stroke or transient ischemic attack, another acute or new cardiovascular outcome prompting healthcare utilization.
These new outcomes could include deep venous thrombosis (DVT), PE, pulmonary hypertension, myocarditis, endocarditis, hypertension emergency, or kidney injury. Critical care utilization due to primary/secondary cardiovascular condition or development of a life-threatening arrhythmia was also included.
Secondary outcomes included all-cause death and any documented cardiac arrhythmia.
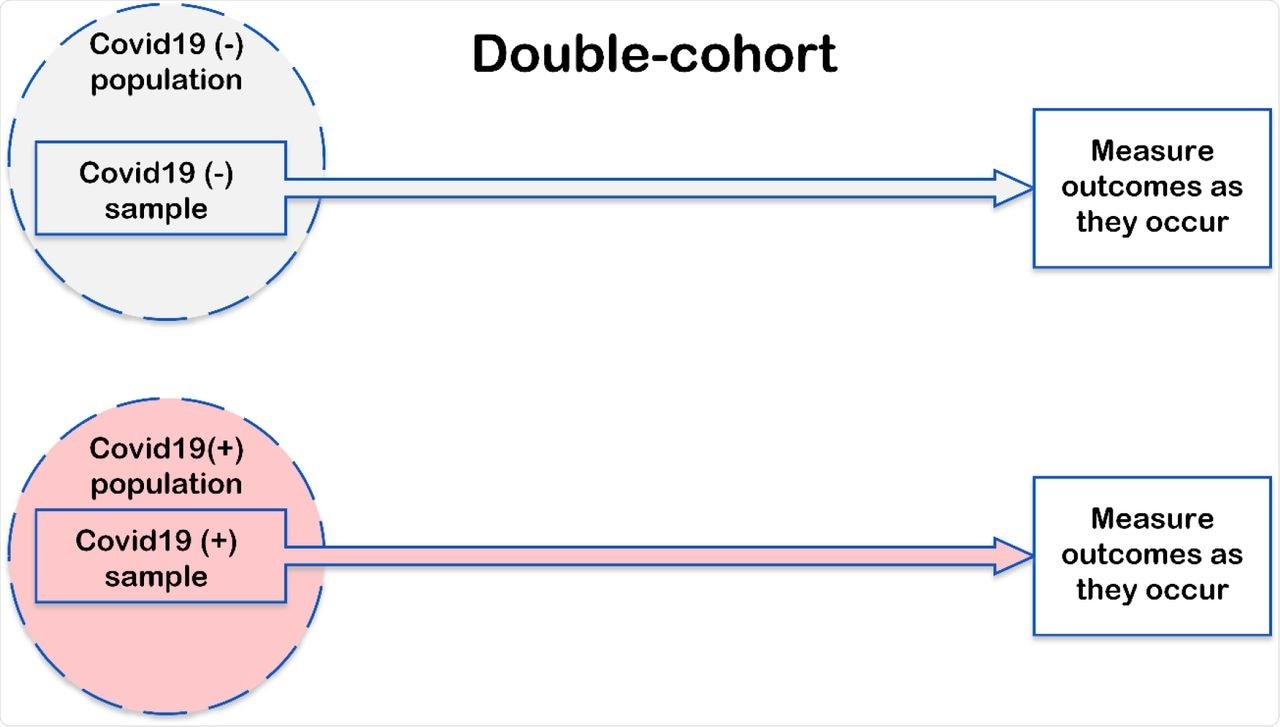 Study design and analysis.
Study design and analysis.
Study findings
During the study period, 99,711 COVID-19 tests were performed for 65,585 individuals. Notably, COVID-19 positive (+) patients were often younger and more likely to be non-white or Hispanic. Additionally, a higher number of COVID-19(+) patients had a history of liver disease and/or diabetes mellitus than those who tested COVID-19 negative (-), while there was no difference in CVD history between the two cohorts.
The most frequent COVID-19 symptoms were cough and fever, both of which were observed in more than 40% of patients. Comparatively, runny nose, headache, muscle and body aches, as well as shortness of breath were present in more than 20% of patients.
In addition, 17% of the patients reported a loss of taste (ageusia) or smell (anosmia), whereas 16% experienced fatigue. Nausea, vomiting, diarrhea, anorexia, and chest pain were less frequently reported.
Other uncommon symptoms included abdominal pain, ear pain, dizziness, vertigo, hemoptysis, and dark stool. Importantly, 20% of COVID-19(+) patients were asymptomatic.
Most patients had a single COVID-19 episode; however, four out of 319 patients had reinfection that occurred at an average of 58.5 days after the first COVID-19(+) episode. Overall, 35 patients tested negative for COVID-19 at an average of 145.5±90.9 days after the COVID-19(+) episode. Only a small number of patients experienced reinfection or two different types of COVID-19 episodes.
During a median of 178 days, the primary composite outcome was noted in 103 patients, of which 12% belonged to the COVID-19(+) cohort and 6% hailed from the COVID-19(-) cohort. Patients detected with the primary outcome were more likely to have greater severity of COVID-19, yet only 26% were hospitalized because of COVID-19, and only 13% required critical care.
In an unadjusted survival analysis and unadjusted Cox regression analysis, COVID-19(+) patients had a significantly higher probability of developing the primary composite outcome than COVID-19(-) patients. The estimated average time to the primary composite outcome in the COVID-19(+) cohort were tested COVID negative was 148.5 days or approximately 4.9 months.
Among the COVID-19(+) cohort, the incidence rate of all-cause death was 41.6 per 1,000 person-years of follow-up. Comparatively, this measure was 45.1 per 1,000 person-years of follow-up among the COVID-19(-) cohort. There was no statistically significant incidence rate difference in the all-cause death between the two cohorts.
For every COVID-19(+) patient, the estimated average time to all-cause death was 65.5 days less than when they were COVID-19(-). The estimated average time to all-cause death when all these patients were COVID-19(-) was 98.6 days.
Moreover, the all-cause death in the COVID-19(+) cohort occurred two months earlier than that which was projected in the absence of SARS-CoV-2 infection. Meanwhile, cardiac arrhythmia was documented in four COVID-19(+) patients and nine COVID-19(-) individuals.
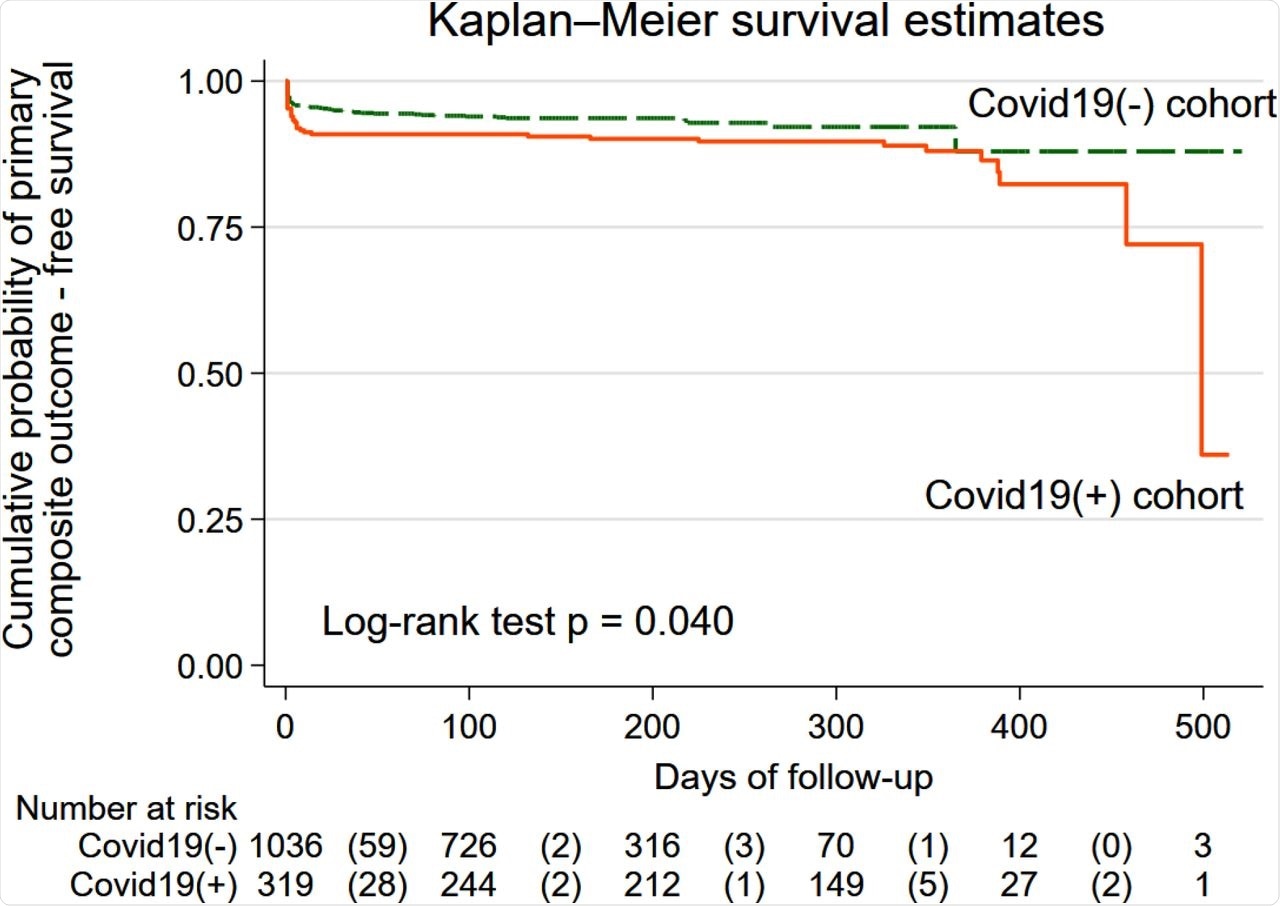 The estimated unadjusted Kaplan-Meier survivor functions for the primary composite outcome in COVID-19(+) (solid orange line) and COVID-19(-) (green dashed line) cohorts. The table below the graph shows the number at risk in each group at every 100 days of follow-up. The number of primary composite outcome events at every 100 days of follow-up is shown in parenthesis.
The estimated unadjusted Kaplan-Meier survivor functions for the primary composite outcome in COVID-19(+) (solid orange line) and COVID-19(-) (green dashed line) cohorts. The table below the graph shows the number at risk in each group at every 100 days of follow-up. The number of primary composite outcome events at every 100 days of follow-up is shown in parenthesis.
Conclusions
The results of the current study suggest that both symptomatic and asymptomatic SARS-CoV-2 infections are associated with an increased risk of late cardiovascular outcomes and have a causal effect on all-cause mortality.
These findings underscore the importance of COVID-19 prevention and emphasize careful follow-up for all patients who encounter SARS-CoV-2, regardless of whether they are symptomatic or asymptomatic, to monitor for late cardiovascular events. Furthermore, the identification and mitigation of future cardiovascular risks are warranted in COVID-19 patients with appropriate screening and preventative measures.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Tereshchenko, L., Bishop, A., Fisher-Campbell, N., et al. (2021). Risk of Cardiovascular Events after Covid-19: a double-cohort study. medRxiv. doi:10.1101/2021.12.27.21268448. https://www.medrxiv.org/content/10.1101/2021.12.27.21268448v1.
- Peer reviewed and published scientific report.
Tereshchenko, Larisa G., Adam Bishop, Nora Fisher-Campbell, Jacqueline Levene, Craig C. Morris, Hetal Patel, Erynn Beeson, et al. 2022. “Risk of Cardiovascular Events after COVID-19.” The American Journal of Cardiology 179 (September): 102–9. https://doi.org/10.1016/j.amjcard.2022.06.023. https://www.ajconline.org/article/S0002-9149(22)00655-5/fulltext.