Research has highlighted that many patients complain about disturbed memory, concentration, fatigue, or insomnia during recovery from acute coronavirus disease 2019 (COVID-19). Critically ill patients such as those with acute respiratory distress syndrome (ARDS) also face similar problems that persist even after one year. This has led to a fear of long-term neurocognitive deficits post-COVID-19, primarily due to cerebral hypoxia, viral encephalitis, metabolic dysfunctions, immune activation, or other mechanisms.
A recent review that involved neuropsychological test data of people who had recent severe acute respiratory syndrome coronavirus 2 (SARS‑CoV‑2) infection reported cognitive impairments as well as impairment of verbal fluency, attention and executive function, and memory to be quite common. Many patients were expected to show improvement during the first 6 to 12 months. However, whether the early assessment of cognitive function is a good indicator of long-term implications is unknown.
Furthermore, there is little information on the cognitive function of patients who recovered without hospitalization, even though they comprise most of the population infected with SARS-CoV-2. In addition, it is uncertain whether neurocognitive impairment is related to COVID-19 severity. Two recent studies with patients 2 to 8 months post-COVID-19 reported worse performance on cognitive tests as compared to controls who had no previous infection.
Now, a new study published in the PLOS ONE journal aims to analyze cognitive deficits in non-hospitalized patients 8 to 13 months post-COVID-19. It also aimed to investigate the variables that are associated with neurocognitive deficits, especially focusing on initial symptoms that suggest an association with the central nervous system (CNS) during the acute phase of infection.
 Study: Cognitive function in non-hospitalized patients 8–13 months after acute COVID-19 infection: A cohort study in Norway. Image Credit: DOERS / Shutterstock
Study: Cognitive function in non-hospitalized patients 8–13 months after acute COVID-19 infection: A cohort study in Norway. Image Credit: DOERS / Shutterstock
About the study
The study involved participants who were 18 years of age and above and had a positive polymerase chain reaction (PCR) for severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) but were not hospitalized. They were recruited during the first wave of the pandemic till 1st June 2020.
Initially, the participants had to respond to a paper or a similar web-based questionnaire to obtain information on background variables (demography, comorbidity, education, weight, height, smoking status, etc.), symptoms experienced during the acute phase of COVID-19, symptoms present during the time of the survey, and some other standard questionnaires such as Chalder fatigue questionnaire (CFQ) and EQ-5D-5L health status questionnaire.
During the follow-up period between December 2020 and April 2021, participants provided blood samples and answered questionnaires. Participants were then divided into three groups based on their previous reports of fatigue, dyspnea, and fatigue and dyspnea. A control group with participants who had no dyspnea or fatigue was also included. The dyspnea and fatigue, fatigue, and no dyspnea or fatigue groups then underwent cognitive testing.
The assessment of cognitive function was done 11 months post-COVID-19 using a battery of tasks that were obtained from the Cambridge Neuropsychological Test Automated Battery (CANTAB) with the help of an iPad. One warm-up task, four tests, and a motor screening test (MOT) were selected. The four tests included Delayed matching to sample (DMS), which tested short-term memory, attention, and learning, One-touch Stockings of Cambridge (OTS), which tested executive functions, Rapid visual information processing (RVP), which tested sustained attention, and Spatial working memory (SWM) which tested strategy and memory. The participants were advised to first complete the MOT followed by RVP, DMS, OTS, and SWM tests.
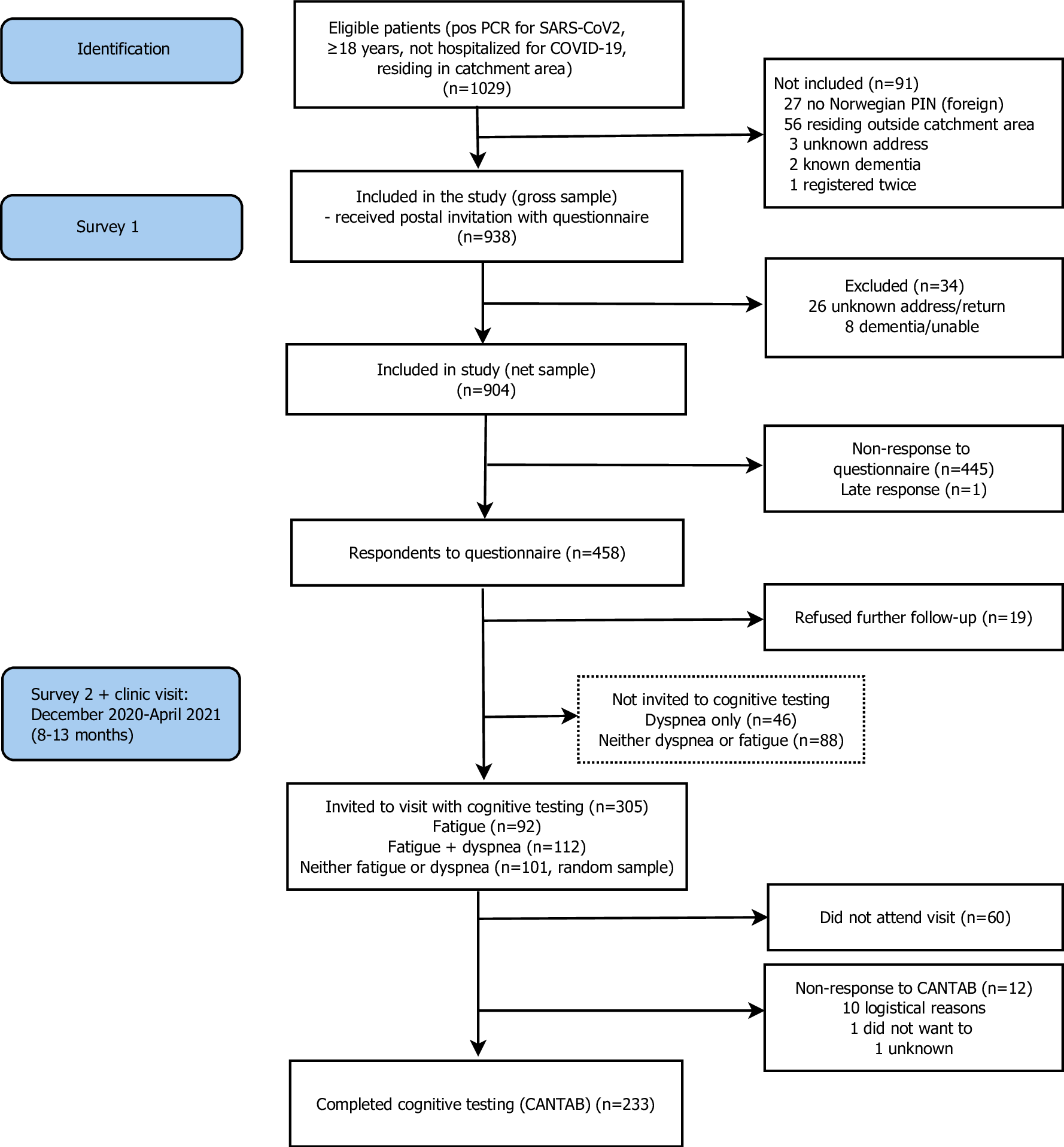 Flow chart of study recruitment and attrition.
Flow chart of study recruitment and attrition.
Study findings
The results indicated that 233 participants completed the CANTAB cognitive test battery. Most COVID-19 symptoms reported were headaches, fevers, dyspnea, and loss of smell. Moreover, those who completed the CANTAB test were found to experience more symptoms during the acute phase, along with a poorer quality of life.
Small reductions in cognitive scores were observed in participants 11 months post-COVID-19 as compared to those without any prior infection. However, no reduction in executive function (OTS) was observed in the entire sample. 29% of the participants reported an impaired cognitive function in at least 1 of 4 tests, while 6% reported in at least 2 of 4 tests at follow-up. Moreover, only the SWM test result was found to be associated with the initial number of COVID-19 symptoms, while no test showed associated with CNS-related symptoms. Additionally, only RVP was found to show an age-related decline, while both RVP and OTS were found to show better scores with a higher degree of education.
Therefore, the current study demonstrated only a slight impairment in cognitive function among non-hospitalized patients 8 to 13 months post-COVID-19. The results suggest that they have little cognitive outcomes after infection compared to experiencing many symptoms in the acute stage.
Limitations
The study has certain limitations. First, the sample size of the study was small. Second, information on pre-infection cognitive function was not available. Third, the study lacked a practical comparison group. Finally, the study might comprise selection bias which limits its generalization.