The Vi-CELL™ cell viability analyzer (Figure 1) from Beckman Coulter has been specifically designed to automate the trypan blue vital dye exclusion technique, which is otherwise performed manually. The main principle of this assay is the uptake of vital stain by cells that are porous and nonviable. The Vi-CELL™ is largely used for determining cells’ percent viability, such as CHO, used in the production of recombinant proteins.
Accurate, Reproducible Measurement
There are a number of laboratories which still employ the standard and manual hemacytometer technique to measure the concentration and percentage viability of blood samples. The Vi-CELL™ cell viability analyzer is capable of automating this procedure and consequently eliminates the biased nature of the standard white blood cell (WBC) counting process. The instrument thus makes it possible to obtain precise, reliable, and reproducible measurements.
Whenever a blood sample for WBC viability is examined, it is important to lyse the red blood cells (RBCs), without damaging the WBCs. However, there are various RBC lytic agents that result in porosity of the WBC membrane in reproducing and actively metabolizing cells. This results in incorrect data with regard to cellular viability.
Figure 1. Beckman Coulter Vi-CELL™. Image credt: Beckman Coulter
Methods and Discussion
When WBCs are examined in a whole blood sample, it is important to choose a suitable lysing solution so that accurate results are obtained. Care should be taken that the lysing mechanism does not damage the WBC membrane. As stated above, membrane porosity due to lysing mechanism promotes the uptake of trypan blue (vital dye) by viable and actively metabolizing cells. WBCs that are damaged will look nonviable.
The Zap-OGLOBIN® II Lytic Reagent from Beckman Coulter is generally used for lysing RBCs and proves suitable for WBC details using the Z™ Series models. A WBC image on the Vi-CELL™ cell viability analyzer using Zap-OGLOBIN II Lytic Reagent is shown in Figure 2.
The lysing mechanism removes the WBC membranes. Therefore, cells that are viable and non-viable now stain blue because of the trypan blue uptake. The Vi-CELL™ circles viable and non-viable cells in green and red, respectively. In addition, Immuno-Lyse and OptiLyse® C from Beckman Coulter were also examined which considerably reduced viability of cells through WBC membrane damage that occurs during the lyse process.
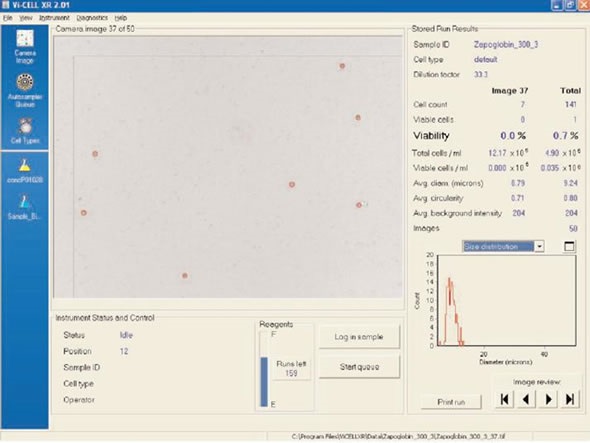
Figure 2. Zap-OGLOBIN® II. Image credt: Beckman Coulter
Ammonium chloride lysing solution is used by a number of laboratories. Beckman Coulter produces an IO Test® 3 lysing solution called PN IM3514, which is a reagent based on ammonium chloride. This reagent is available as a 10X concentrated product, although a 1X solution has to be prepared on a daily basis. The outcome of the same blood sample using the IO Test® 3 is shown in Figure 3. This lyse does not disrupt the remaining WBC population and allows a precise measurement of WBC viability.
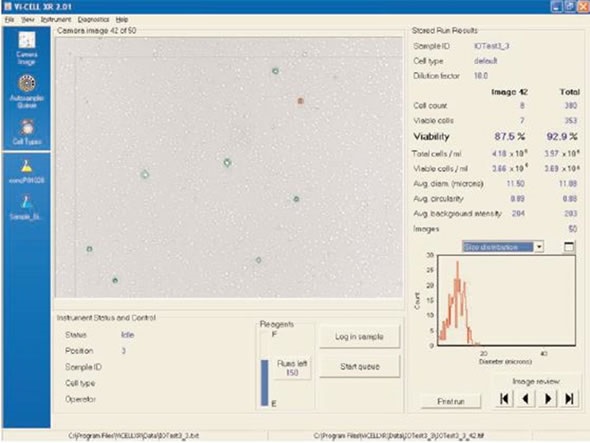
Figure 3. IO Test® 3. Image credt: Beckman Coulter
Beckman Coulter has also introduced a new lysing reagent called VersaLyse, PN IM3648. Figure 4 displays WBCs in a whole blood sample using VersaLyse reagent. It can be observed that the “background” of RBC “ghosts” (structures) is created with this reagent. When the IO Test® 3 reagent is used, “ghosts” can also be observed. These “ghosts” are the RBC “re-sealed” membranes that form following the release of RBC hemoglobin. The WBCs captured by the imaging algorithm of the Vi-CELL™ are quite evident.
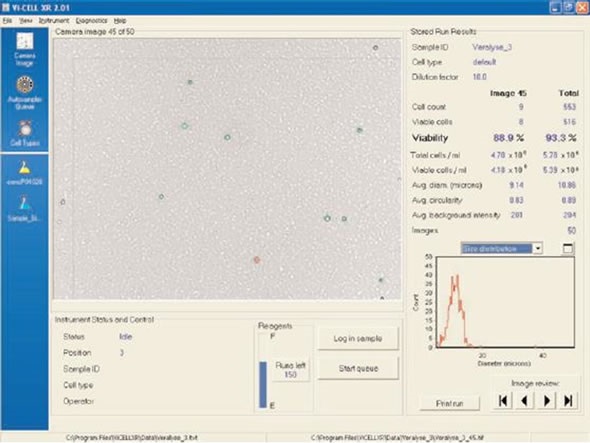
Figure 4. VersaLyse. Note RBC “ghosts” (white specs). Image credt: Beckman Coulter
With the help of VersaLyse, the sample can be easily prepared. First, 100µL of whole blood is added to a Vi-CELL cup or tube followed by 1ml of VersaLyse. The mixture is then vortexed for 5 seconds and incubated for a period of 10 minutes at ambient temperature. The sample is then loaded onto the Vi-CELL™ XR. Here, the default imaging cell type parameters should be used. By adding 2ml of lyse to 100 µL of blood, the same process can be employed with the IO Test® 3 solution.
Conclusion
Beckman Coulter’s Vi-CELL™ cell viability analyzer automates the WBC viability technique, which is rather tedious and cumbersome. An appropriate RBC lytic agent is important to obtain accurate measurement of WBC viability. However, OptiLyse® C, Immuno-Lyse, or Zap-OGLOBIN II are not suitable for WBC viability testing. Beckman Coulter’s VersaLyse and IO Test® 3 reagents allow accurate WBC viability measurements using the Vi-CELL™ cell viability analyzer.
 About Beckman Coulter
About Beckman Coulter
Beckman Coulter develops, manufactures and markets products that simplify, automate and innovate complex biomedical tests. More than a quarter of a million Beckman Coulter instruments operate in laboratories around the world, supplying critical information for improving patient health and reducing the cost of care.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.