Autoimmune diseases stem from a complicated interplay of immune dysregulation, characterized by chronic inflammation and the loss of self-tolerance.
Cytokines are central to these processes. These soluble signaling proteins coordinate immune responses by mediating communication between cells.
Cytokine networks demonstrate dual functionality. Cytokines are key regulators of immune homeostasis, but paradoxically, cytokines also function as pathogenic drivers in diseases such as systemic lupus erythematosus (SLE), psoriasis, rheumatoid arthritis (RA), and inflammatory myopathies.
Research has successfully highlighted the therapeutic potential of cytokine-targeted biologics, as well as Janus kinase (JAK) inhibitors and evolving strategies like engineered cytokines and miRNA modulation.
Mechanism of autoimmunity
Autoimmunity is the result of complex disruptions in immune signaling pathways. These disruptions collectively damage normal tissues, with genetic predisposition and environmental factors triggering excessive cytokine production, the activation of self-reactive lymphocytes, and autoantibody release.
Resolution of autoimmune processes is sometimes possible by restoring regulatory mechanisms via regulatory T cells (Tregs) and regulatory B cells (Bregs).
These cells produce immunosuppressive cytokines such as interleukin-10 (IL-10) and transforming growth factor-beta (TGF-β), which help repair tissue damage and suppress the inflammatory pathways activated during the initiation and propagation phases.
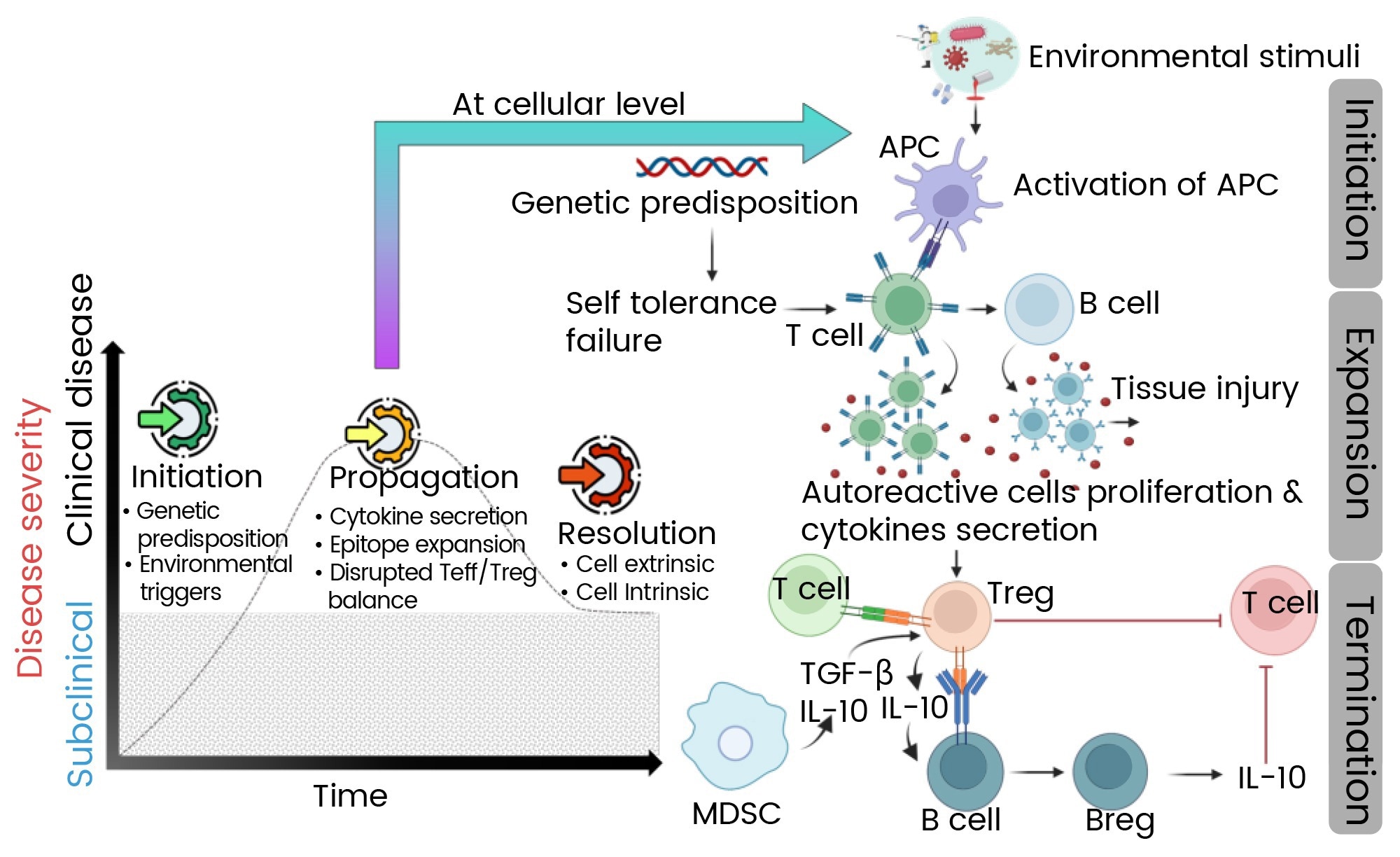
Figure 1. Mechanism of autoimmune disease progression.1 Image Credit: https://doi.org/10.3390/ijms25147666
Cytokine networks in autoimmune pathogenesis
Pro-inflammatory cytokine dominance
Autoimmune diseases’ pathogenesis is often driven by a hyperactive pro-inflammatory cytokine environment. For example, IL-6, IL-17, and tumor necrosis factor-alpha (TNF-α) are core mediators in conditions like psoriasis and RA.
The IL-1 family, including IL-1β and IL-18, exacerbates tissue damage even further by activating the NLRP3 inflammasome, a mechanism that is also implicated in SLE.2,3
Not only do these cytokines drive local inflammation, but they also contribute to systemic manifestations like fatigue and fever via their actions on the hypothalamic-pituitary-adrenal axis.4
Table 1. Disease-specific cytokines. Source: Sino Biological Inc.
| Disease |
Cytokines Involved |
References |
Rheumatoid
Arthritis |
TNF-α, IL-6, GM-CSF, IL-23/IL-17 axis,
IL-7 and IL-21 |
Kondo, N., et al, 2021:
Leung, S. et al, 2010 |
Systemic Lupus
Erythematosus |
IFN-α /ẞ, IL-2, IL-1ẞ,
IL-18 and IL-12 |
Leung, S., et al, 2010;
Kotyla, P., et al, 2022 |
Psoriasis and
Psoriatic Arthritis |
IL-23/IL-17 axis (IL-17A, IL-17F),
IL-22, TNF-α and IL-6 |
Wojas-pelc, A., et al, 2006 |
Inflammatory
Myopathies |
IFN-α/ẞ, IFN-y, CXCL9,
CXCL10, and IL-1ẞ |
De Paepe, B., et al, 2015 |
GM-CSF: granulocyte-macrophage colony-stimulating factor
IFN-α/β: type I interferons
Regulatory cytokine deficiencies
Immunosuppressive cytokines such as IL-10 and TGF-β. IL-10, produced by Bregs and Tregs, can counterbalance pro-inflammatory signals. This inhibits antigen presentation and suppresses Th1/Th17 responses.5,6
It was observed that reduced IL-10 production by Bregs correlated with disease flares in SLE, serving to highlight its protective role.6 TGF-β, in contrast, maintains peripheral tolerance by suppressing effector T cell proliferation and inducing Treg differentiation.3.
Dysregulation of these regulatory pathways creates a permissive environment for autoimmunity. This is observable in RA and multiple sclerosis (MS), where defective TGF-β signaling can be seen to permit unchecked Th17 activity.3,7
Cytokine imbalance and feedback loops
Autoimmune diseases typically exhibit self-reinforcing cytokine feedback loops. For example, IL-6 enhances Th17 differentiation while simultaneously inhibiting Treg development. This creates a pathogenic cycle that sustains inflammation.7
In the case of psoriasis, Th17 survival is perpetuated by IL-23 produced by dendritic cells. This in turn results in the secretion of IL-17 and IL-22, further activating stromal cells and keratinocytes.8
These loops are further exacerbated by tissue-resident cells. For example, synovial fibroblasts in RA produce chemokines like CXCL13 in order to recruit B cells and plasma cells, resulting in the fostering of ectopic lymphoid structure formation.7
Therapeutic targeting of cytokine networks
Biologic therapies
Anti-TNF-α agents: TNF inhibitors (for example, adalimumab and infliximab) have transformed RA treatment through the reduction of synovitis and radiographic progression.7 TNF blockade may induce psoriasiform lesions in some patients, however, underscoring cytokine pleiotropy.8
IL-6 inhibition: The IL-6 receptor antagonist Tocilizumab has been shown to ameliorate systemic inflammation in juvenile idiopathic arthritis (JIA) and RA.7 IL-6 blockade also shows the potential to reduce relapse frequency in neuromyelitis optica spectrum disorder (NMOSD).
IL-17/IL-23 axis targeting: Secukinumab (anti-IL-17A) and ustekinumab (anti-IL-12/23p40) have been shown to achieve rapid skin clearance in psoriasis through the disruption of Th17 signaling.8 Brodalumab also demonstrates efficacy in psoriatic arthritis by targeting the IL-17 receptor.8
JAK/STAT inhibition
JAK inhibitors (jakinibs) block downstream STAT phosphorylation, effectively modulating cytokine signaling. Tofacitinib (JAK1/3 inhibitor) and baricitinib (JAK1/2 inhibitor) are approved for the treatment of RA, working by suppressing IFN-γ, IL-6, and GM-CSF pathways.9
JAK inhibitors also reduce IFN-α signature and ameliorate nephritis in SLE, offering a useful alternative to broad immunosuppression.9
IL-2-based immunotherapies
Low-dose IL-2 has been shown to expand Tregs, restoring immune tolerance in type 1 diabetes and SLE.10 Engineered IL-2 variants with improved Treg specificity, for example, IL-2-anti-IL-2 complexes, have shown promise in preclinical models, though it has been noted that clinical trials report varying efficacy.10,11
miRNA modulation
miRNAs are able to regulate cytokine production post-transcriptionally. For example, miR-155 has been shown to promote TNF-α and IL-6 in RA synovium, while miR-146a feedback has been shown to inhibit NF-κB signaling.12
Antagomirs targeting miR-155 have the capacity to reduce disease severity in experimental autoimmune encephalomyelitis (EAE), showcasing the potential applicability of miRNA-based therapies.12
Engineered cytokines
Cytokine engineering seeks to improve therapeutic specificity. For example, PEGylated IL-10 (AM0010) has been shown to prolong half-life and suppress colitis in preclinical models,11 while IL-4 fusion proteins have been shown to bias macrophage polarization toward an anti-inflammatory phenotype, offering promising new strategies for fibrosis-prone diseases such as systemic sclerosis.11
Table 2. Representative FDA-approved drugs for autoimmune diseases targeting cytokines. Source: Sino Biological Inc.
| Drug Name |
Target Cytokine |
Brand Name |
FDA Approval |
Indications |
| Etanercept |
TNF-α |
Enbrel |
1998 |
Rheumatoid arthritis (RA), juvenile idiopathic arthritis (JIA), ankylosing spondylitis (AS), psoriasis, psoriatic arthritis (PSA) |
| Infliximab |
TNF-α |
Remicade |
1998 |
RA, AS, psoriasis, PSA, ulcerative colitis (UC), Crohn's disease (CD) |
| Adalimumab |
TNF-α |
Humira |
2002 |
RA, JIA, AS, psoriasis, PsA, UC, CD, hidradenitis suppurativa, uveitis |
| Golimumab |
TNF-α |
Simponi |
2009 |
RA, AS, PSA, UC |
| Certolizumab |
TNF-α |
Cimzia |
2008 |
RA, AS, psoriasis, PsA, CD |
| Anakinra |
IL-1 |
Kineret |
2001 |
RA, Cryopyrin-associated periodic syndromes (CAPS) |
| Tocilizumab |
IL-6R |
Actemra |
2010 |
RA, juvenile idiopathic arthritis (JIA), adult-onset Still's disease (AOSD), giant cell arteritis, cytokine release syndrome |
| Sarilumab |
IL-6R |
Kevzara |
2017 |
RA |
| Siltuximab |
IL-6 |
Sylvant |
2014 |
Multicentric Castleman's disease |
| Satralizumab |
IL-6R |
Enspryng |
2020 |
Neuromyelitis optica spectrum disorder (NMOSD) |
| Secukinumab |
IL-17A |
Cosentyx |
2015 |
AS, psoriasis, PSA |
| Ixekizumab |
IL-17A |
Taltz |
2016 |
Psoriasis, PsA, AS |
| Brodalumab |
IL-17 receptor |
Siliq |
2017 |
Psoriasis |
| Ustekinumab |
IL-12, IL-23 |
Stelara |
2009 |
Psoriasis, PsA, CD, UC |
| Canakinumab |
IL-1ẞ |
Ilaris |
2009 |
CAPS, systemic juvenile idiopathic arthritis (SJIA), TRAPS, HIDS/MKD, familial Mediterranean fever (FMF) |
| Satralizumab |
IL-6R |
Enspryng |
2020 |
Neuromyelitis optica spectrum disorder (NMOSD) |
| Tofacitinib |
JAKI, JAK3 |
Xeljanz |
2018 |
RA, PSA, UC, AS, JIA |
Abbreviations
- RA: Rheumatoid Arthritis
- AS: Ankylosing Spondylitis
- PsA: Psoriatic Arthritis
- UC: Ulcerative Colitis
- CD: Crohn's Disease
- JIA: Juvenile Idiopathic Arthritis
- pJIA: Polyarticular Juvenile Idiopathic Arthritis
- FMF: Familial Mediterranean Fever
- NMOSD: Neuromyelitis Optica Spectrum Disorder
Conclusion
Pioneering new developments in cytokine biology have enabled the determination of a range of disease-specific signatures, facilitating precision therapies able to target key nodes within these networks.
Challenges remain, however, including pleiotropy, interpatient heterogeneity, and cytokine redundancy. Ongoing research areas include personalized cytokine profiling, microbiome-directed interventions, and engineered biologics with enhanced cell-type specificity.
The integration of mechanistic insights with innovative therapeutics is set to open up the next frontier in autoimmunity, effectively harnessing the cytokine network to restore immune equilibrium.
Sino Biological provides a range of high-quality cytokine products designed to support targeted research and drug development for autoimmune diseases. The company’s products are subject to strict quality control in order to ensure high purity, stability, and bioactivity, along with low endotoxin levels.
Options are available across a range of species, including human, monkey, mouse, and rat. Sino Biological also offers a comprehensive portfolio of autoimmune disease solutions, including a wide range of research reagents for almost 50 diseases.
Its portfolio includes autoimmune disease target reagents such as cytokines, kinases, and target proteins, as well as research reagents for biomarker studies.
Sino Biological plays a key role in advancing early detection and targeted therapy development for autoimmune diseases by providing the high-quality tools required for biomarker analysis and drug discovery.
Featured Products for Autoimmune Research. Source: Sino Biological Inc.
| Cat# |
Molecule |
Species |
Expression Host |
SEC-HPLC Purity |
Activity |
| GMP-11848-HNAE |
IL-2 |
Human |
E. coli |
≥ 95% |
Active |
| GMP-11846-HNAE |
IL-4 |
Human |
E. coli |
≥ 95% |
Active |
| GMP-10360-HNAE |
IL-15 |
Human |
E. coli |
≥ 95% |
Active |
| GMP-CT011-H08H |
IL-12 |
Human |
HEK293 Cells |
≥ 95% |
Active |
| 51112-MNAH |
M-CSF/CSF1 |
Mouse |
HEK293 Cells |
≥ 95% |
Active |
| 10602-HNAE |
TNF-alpha |
Human |
E. coli |
≥ 95% |
Active |
| 10804-HNAC |
TGF beta 1 |
Human, Rhesus,
Cynomolgus, Canine |
CHO
Stable Cells |
> 95%
(SDS-PAGE) |
Active |
| 10395-HNAE |
IL-6 |
Human |
E. coli |
≥ 95%
(SDS-PAGE) |
Active |
| 11821-HNAE |
IL-7 |
Human |
E. coli |
≥ 95% |
Active |
| 50245-MNAE |
IL-10 |
Mouse |
E. coli |
≥ 90%
(SDS-PAGE) |
Active |
| 12047-HNAE |
IL-17 |
Human |
E. coli |
≥95%
(SDS-PAGE) |
Active |
| CT048-H08H |
IL-23 |
Human |
HEK293 Cells |
> 90%
(SDS-PAGE) |
Active |
| 10119-HNCE |
IL-18 |
Human |
E. coli |
≥ 95% |
Active |
| 17049-H07H2 |
TLIA/TNFSF15 |
Human |
HEK293 Cells |
≥ 95% |
Active |
References and further reading
- Farzana Yasmeen, et al. (2024). Understanding Autoimmunity: Mechanisms, Predisposing Factors, and Cytokine Therapies. International Journal of Molecular Sciences, 25(14), pp.7666–7666. https://doi.org/10.3390/ijms25147666.
- De Paepe, B. and Zschüntzsch, J. (2015). Scanning for Therapeutic Targets within the Cytokine Network of Idiopathic Inflammatory Myopathies. International Journal of Molecular Sciences, 16(8), pp.18683–18713. https://doi.org/10.3390/ijms160818683.
- Leung, S., et al. (2010). The cytokine milieu in the interplay of pathogenic Th1/Th17 cells and regulatory T cells in autoimmune disease. Cellular & Molecular Immunology, 7(3), pp.182–189. https://doi.org/10.1038/cmi.2010.22.
- Biscetti, L., et al. (2021). Headache and immunological/autoimmune disorders: a comprehensive review of available epidemiological evidence with insights on potential underlying mechanisms. Journal of Neuroinflammation, 18(1). https://doi.org/10.1186/s12974-021-02229-5.
- de Gruijter, N.M., Jebson, B. and Rosser, E.C. (2022). Cytokine production by human B cells: role in health and autoimmune disease. Clinical and Experimental Immunology, (online) 210(3), pp.253–262. https://doi.org/10.1093/cei/uxac090.
- Lino, A.C., et al. (2015). Cytokine-producing B cells: a translational view on their roles in human and mouse autoimmune diseases. Immunological Reviews, 269(1), pp.130–144. https://doi.org/10.1111/imr.12374.
- Kondo, N., Kuroda, T. and Kobayashi, D. (2021). Cytokine Networks in the Pathogenesis of Rheumatoid Arthritis. International Journal of Molecular Sciences, 22(20), p.10922. https://doi.org/10.3390/ijms222010922.
- Wojas-Pelc, A. et al. (2007) Review paperCytokine network in psoriasis. Cross-talk between keratinocytes and cells. https://www.termedia.pl/Review-paper-Cytokine-network-in-psoriasis-Cross-talk-between-keratinocytes-and-cells,10,7464,0,1.html.
- Kotyla, P., et al. (2022). Jak Inhibitors for Treatment of Autoimmune Diseases: Lessons from Systemic Sclerosis and Systemic Lupus Erythematosus. Pharmaceuticals, 15(8), p.936. https://doi.org/10.3390/ph15080936.
- Raeber, M.E., et al. (2023). A systematic review of interleukin-2-based immunotherapies in clinical trials for cancer and autoimmune diseases. eBioMedicine, (online) 90. https://doi.org/10.1016/j.ebiom.2023.104539.
- Deckers, J., et al. (2023). Engineering cytokine therapeutics. Nature Reviews Bioengineering, 1(4), pp.286–303. https://doi.org/10.1038/s44222-023-00030-y.
- Salvi, V., et al. (2019). Cytokine Targeting by miRNAs in Autoimmune Diseases. Frontiers in Immunology, (online) 10. https://doi.org/10.3389/fimmu.2019.00015.
- Jung, S.M. and Kim, W.-U. (2022). Targeted Immunotherapy for Autoimmune Disease. Immune Network, 22(1). https://doi.org/10.4110/in.2022.22.e9.
Acknowledgments
Produced from materials originally authored by Sino Biological.
About Sino Biological Inc.
Sino Biological is an international reagent supplier and service provider. The company specializes in recombinant protein production and antibody development. All of Sino Biological's products are independently developed and produced, including recombinant proteins, antibodies and cDNA clones. Sino Biological is the researchers' one-stop technical services shop for the advanced technology platforms they need to make advancements. In addition, Sino Biological offer pharmaceutical companies and biotechnology firms pre-clinical production technology services for hundreds of monoclonal antibody drug candidates.
Sino Biological's core business
Sino Biological is committed to providing high-quality recombinant protein and antibody reagents and to being a one-stop technical services shop for life science researchers around the world. All of our products are independently developed and produced. In addition, we offer pharmaceutical companies and biotechnology firms pre-clinical production technology services for hundreds of monoclonal antibody drug candidates. Our product quality control indicators meet rigorous requirements for clinical use samples. It takes only a few weeks for us to produce 1 to 30 grams of purified monoclonal antibody from gene sequencing.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.