Exosomes are extracellular vesicles ranging between 30 and 150 nm in diameter that are released from different kinds of cells, including tumor cells. They have the potential to induce apoptosis, modulate the immune system, and work as biomarkers for diagnosis. As a key component of cell-to-cell communication, exosomes can control the tumor microenvironment and contribute immensely to the development, progression, and metastasis processes of numerous cancers.1,2
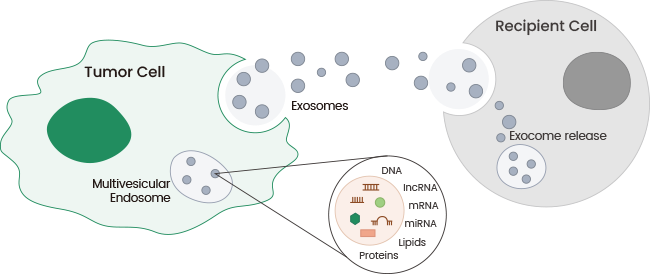
Image Credit: Sino Biological Inc.
Tumor-derived exosomes include nucleic acids, proteins, metabolites, and lipids that can serve as efficient messengers in the communication among tumor cells and different kinds of cells, such as vascular endothelial cells, immune cells, and mesenchymal cells.
Communication between such cells has vital impacts on tumor biological activities, like angiogenesis, immune regulation, and epithelial-mesenchymal transition (EMT), which further direct tumor cell growth, proliferation, and metastasis.3
Exosomes and tumor immune response
Tumor exosomes tend to play a dual role in immune regulation. On the one hand, exosomes play an important role in promoting antitumor immune responses through their immune activity. A few exosomes released by tumor cells carry tumor-specific antigens, like HER2, mesothelin, CEACAM5, CD24, and EpCAM, inducing protective antitumor immune responses and activating cytotoxic T-cell responses.
Exosomes derived from tumors can also directly activate NK cells by expressing the stress protein HSP70, promoting tumor regression and eliciting antitumor immune responses.
On the other hand, there is some evidence to suggest that tumor-derived exosomes can encourage cancer progression by suppressing the host immune response. The mechanisms include activating suppressor immune cells and inhibiting immune cell effectors. Some tumor-derived exosomes carry PD-L1, with IFN-γ increasing the quantity of PD-L1 on such exosomes.
PD-L1 binds to PD-1 via its extracellular structural domain, inactivating CD8 T cells, in turn hindering CD8 T cell function and encouraging tumor growth. Exosomes may also suppress T cell receptor activity or regulate effector T cell transcriptome.
Moreover, TGF-β from tumor-derived exosomes exerts an inductive effect on Treg cells, hinders the activation of IL-2-mediated NK cells, and decreases the receptor expression of NK cell activation.3-5 Altogether, the tumor-derived exosomes’ role in the immune response is complicated and associated with the local microenvironment produced by the tumor itself.
As a leading global supplier of CRO services and bioreagents for the biopharmaceutical field, Sino Biological provides a complete assembly of high-quality recombinant proteins and equivalent antibodies to aid in research on exosomes and tumor immune response.
CEA, HER2, mesothelin, CD24, and EpCAM: Tumor-specific antigens
Source: Sino Biological Inc.
| Cat# |
Proteins |
Purity |
Expression Host |
| 11077-H02H |
CEACAM5 (Human) |
≥95% (SEC-HPLC) |
HEK293 Cells |
| 10004-H08H |
HER2 (Human) |
>90% (SEC-HPLC) |
HEK293 Cells |
| 13128-HNCH |
Mesothelin (Human) |
≥95% (SEC-HPLC) |
HEK293 Cells |
| 11030-H02H |
CD24 (Human) |
>92% |
HEK293 Cells |
| 10694-H08H |
EpCAM (Human) |
≥90% (SEC-HPLC) |
HEK293 Cells |
TGF-β, IFN-γ, PD-L1: Associated with immunosuppression
Human TGF-β1 protein
(Cat#: 10804-H08H)
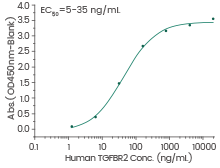
Immobilized human TGF beta 1 protein can bind human TGFBR2 protein. Image Credit: Sino Biological Inc.
Human IFN-γ protein
(Cat#: 11725-HNAE)
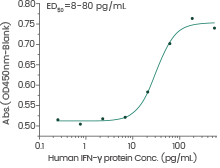
Anti-viral assays using WISH cells infected with vesicular stomatitis virus (VSV). Image Credit: Sino Biological Inc.
Human PD-L1 Protein
(Cat#: 10084-H08H-B)
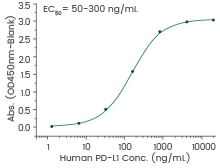
Immobilized human PD-1 (Cat#: 10377-H03H) can bind human PD-L1. Image Credit: Sino Biological Inc.
Exosomes and angiogenesis
Cancer progression is closely related to angiogenesis. Nascent capillaries provide oxygen, nutrients, and growth factors to the tumor and serve a key role in tumor cell metastasis and proliferation.
Exosomes that are released from tumor cells, which are involved in tumor angiogenesis, can be taken up by vascular endothelial cells, thereby encouraging angiogenesis. Exosome production is improved under hypoxic conditions.
This process is associated with the pro-angiogenic secretory group of endothelial cells.3
There is also evidence to support the claim that exosomes play an important role in angiogenesis. Myeloid leukemia-derived exosomes regulate angiogenesis by enriching vascular cell adhesion molecule-1 (VCAM-1) and intercellular adhesion molecule-1 (ICAM-1).
Exosomes secreted by skin cancer cells deliver EGFR to endothelial cells and encourage angiogenesis. Furthermore, particular tumor-derived exosomes can influence the pro-angiogenic cytokines’ secretion, along with TNF-α, G-CSF, VEGF, FGF, etc.6
Among them, VEGF-A, which is a part of the VEGF family, serves an essential role in tumor angiogenesis. Other members of this family, including PlGF, VEGF-B, VEGF-C, VEGF-D, and VEGF-E, show attraction to VEGFR.
Except for PlGF, other members play important roles in the process of angiogenesis, thereby promoting cancer progression.7,8 Sino Biological has developed a comprehensive collection of cytokines and corresponding receptors associated with angiogenesis, which have been validated to be of high purity and activity.
Human VEGF165 protein
(Cat#: 11066-H27H-B)
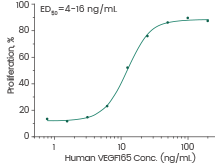
Cell proliferation assay using human umbilical vein endothelial cells (HUVEC). Image Credit: Sino Biological Inc.
Human FGF2 protein
(Cat#: 10014-HNAE)
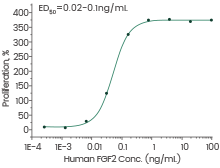
Cell proliferation assay using BALB/c 3T3 mouse embryonic fibroblasts. Image Credit: Sino Biological Inc.
Human EGFR protein
(Cat#: 10001-H08H)
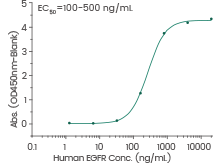
Immobilized human EGF protein (Cat#: 10605-H01H) can bind human EGFR protein. Image Credit: Sino Biological Inc.
Exosomes and epithelial–mesenchymal transition (EMT)
EMT is a cellular process in which epithelial cells lose cell polarity and intercellular adhesion and transform into loosely structured mesenchymal cells. During EMT, the expression of epithelial cell markers, like β-catenin and E-cadherin, is downregulated, while that of hypodermal cell markers, like vimentin and N-cadherin, is upregulated.
EMT is related to cancer progression and promotes metastasis and tumor invasion, which induce poor patient prognosis. Tumor-derived exosomes play an important role in the induction of tumor-associated EMT, from the initial activation of the invasive phenotype to metastasis.9,10
Tumor-derived exosomes comprise EMT-related inducers, like TNF-α, TGF-β, IL-6, caveolin-1, β-catenin, and HIF-1α, contributing to the stimulation of EMT, improving cell invasion, and inducing tumor cell metastasis and development. In hypoxic settings, the release of exosomes from varied tumor cells, like prostate and breast cancer, glioma, and leukemia cells, is enhanced and can further improve the incidence of metastasis via EMT.
Additionally, exosomes contain nucleic acids, like RNA, DNA, miRNA, and non-coding RNA, which can be involved in EMT regulation in cancer. The common EMT-related regulatory pathways comprise the Hippo, β-catenin, and MAPK/ERK pathways. Sino Biological offers EMT-associated recombinant proteins and equivalent antibody products to direct exosome and EMT research.10,11
Human IL-6 protein
(Cat#: 10395-HNAE)
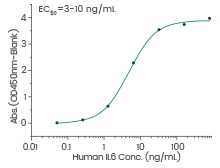
Immobilized human IL-6 can bind human IL-6R hFc (Cat#: 10398-H02H). Image Credit: Sino Biological Inc.
Anti-β-Catenin antibody
(Cat#: 11279-R021)
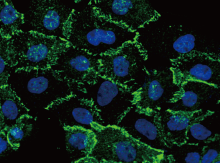
Immunofluorescence staining of Human CTNNB1 in A431 cells. Image Credit: Sino Biological Inc.
Anti-Vimentin antibody
(Cat#: 100254-R001)
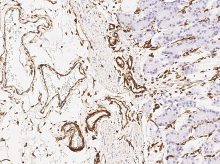
Immunochemical staining of human Vimentin in human stomach. Image Credit: Sino Biological Inc.
Exosomes in cancer therapies
Targeting tumor-derived exosomes can have a positive effect on cancer therapy. Exosomes secreted by malignant cells have a tumor-promoting effect. In general, tumor cells secrete more exosomes than normal cells. These secretomes carry lncRNAs, miRNAs, and proteins that have the potential to serve as biomarkers for cancer prognostic monitoring and diagnosis.4
Additionally, exosomes play an important role in resistance to therapy. Exosomes carry a specific set of miRs that can transfer resistant phenotypes to sensitive cancer cells, conferring resistance to chemotherapy and radiation, which is achieved by altering the cell cycle and inducing anti-apoptotic processes.
Exosomes are also able to hinder the passing of chemotherapeutic agents into target cancer cells, subsequently impacting the efficiency of chemotherapy.3 Thus, targeting exosomes hold great promise for cancer therapy, and relevant clinical trials are already underway.
Summary
Exosomes are biologically active extracellular vesicles that include nucleic acids, proteins, and lipids that facilitate cell-to-cell communication. Interaction between cells can shape the tumor microenvironment and is vital for tumor invasion, growth, and immune surveillance. Exosomes hold great promise as new biomarkers for therapeutic intervention, cancer diagnosis, and prognostic forecast, which has the potential to open up new opportunities in cancer therapy.
Future studies of exosomes may concentrate on the characterization, purification, and compositional assay of exosomes related to cancer progression.5
Sino Biological is a leading global supplier of bioreagents and CRO services for the biopharmaceutical field. As such, the company supports exosome and tumor progression research by equipping researchers with a vast range of high-quality reagents and services.
References and further reading
- Han, Chen, et al. "Exosome-mediated communication between tumor cells and tumor-associated macrophages: implications for tumor microenvironment." Oncoimmunology 10.1 (2021): 1887552. doi: 10.1080/2162402X.2021.1887552
- Nicolini, Andrea, Paola Ferrari, and Pier Mario Biava. "Exosomes and cell communication: from tumour-derived exosomes and their role in tumour progression to the use of exosomal cargo for cancer treatment." Cancers 13.4 (2021): 822. doi: 10.3390/cancers13040822
- Kalluri, Raghu. "The biology and function of exosomes in cancer." The Journal of clinical investigation 126.4 (2016): 1208-1215. doi: 10.1172/JCI81135
- Liu, Shang-Long, et al. "Exosomes as critical mediators of cell-to-cell communication in cancer pathogenesis and their potential clinical application." Translational Cancer Research 8.1 (2019): 298. doi: 10.21037/tcr.2019.01.03
- Li, Qiao. "Role of exosomes in cellular communication between tumor cells and the tumor microenvironment." Oncology Letters 24.1 (2022): 1-8. doi: 10.3892/ol.2022.13360
- Maia, Joana, et al. "Exosome-based cell-cell communication in the tumor microenvironment." Frontiers in cell and developmental biology 6 (2018): 18. doi: 10.3389/fcell.2018.00018
- Aslan, Cynthia, et al. "Tumor‐derived exosomes: Implication in angiogenesis and antiangiogenesis cancer therapy." Journal of cellular physiology 234.10 (2019): 16885-16903. doi: 10.1002/jcp.28374
- Olejarz, Wioletta, et al. "Exosomes in angiogenesis and anti-angiogenic therapy in cancers." International Journal of Molecular Sciences 21.16 (2020): 5840. doi 10.3390/ijms21165840
- Kim, Hyunwoo, et al. "The emerging roles of exosomes as EMT regulators in cancer." Cells 9.4 (2020): 861. doi: 10.3390/cells9040861
- Wu, Miaowei, et al. "Emerging roles and therapeutic value of exosomes in cancer metastasis." Molecular cancer 18.1 (2019): 1-11. doi: 10.1186/s12943-019-0964-8
- Vella, Laura Jayne. "The emerging role of exosomes in epithelial–mesenchymal-transition in cancer." Frontiers in oncology 4 (2014): 361. doi: 10.3389/fonc.2014.00361
About Sino Biological Inc.

Sino Biological is an international reagent supplier and service provider. The company specializes in recombinant protein production and antibody development. All of Sino Biological's products are independently developed and produced, including recombinant proteins, antibodies and cDNA clones. Sino Biological is the researchers' one-stop technical services shop for the advanced technology platforms they need to make advancements. In addition, Sino Biological offers pharmaceutical companies and biotechnology firms pre-clinical production technology services for hundreds of monoclonal antibody drug candidates.
Sino Biological's core business
Sino Biological is committed to providing high-quality recombinant protein and antibody reagents and to being a one-stop technical services shop for life science researchers around the world. All of our products are independently developed and produced. In addition, we offer pharmaceutical companies and biotechnology firms pre-clinical production technology services for hundreds of monoclonal antibody drug candidates. Our product quality control indicators meet rigorous requirements for clinical use samples. It takes only a few weeks for us to produce 1 to 30 grams of purified monoclonal antibody from gene sequencing.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.