Sponsored Content by LonzaReviewed by Ify IsiborMay 1 2026
Pharmaceutical products must be produced in a way that guarantees their efficacy and safety. For parenteral drug products, visual inspection (VI) is an essential quality control measure used to detect packaging defects and particulate contamination.
Whether carried out manually or using (semi-) automated techniques, effective VI depends on well-trained operators and appropriately qualified equipment. VI test sets employed for such purposes must, therefore, be of the highest quality.
To meet regulatory expectations and maintain high-quality standards, biopharmaceutical companies must choose reliable VI test set providers that offer end-to-end services and have a proven track record of quickly delivering high-quality test sets across geographies, modalities, and applications. Lonza is one such company.
Introduction
Maintaining the safety and efficacy of pharmaceutical products is a crucial parameter in drug manufacturing. This includes rigorous control of sterility and quality for parenteral drug products.
Microbial, viral, and particulate (visible and non-visible) contaminants, discoloring, and other negative impacts on appearance must be avoided. Similarly, there must be no flaws in primary packaging (vials, prefilled syringes, bags, etc.).
Visible particulates, generally composed of glass, metals, polymers, or fibers, are of particular concern as they can cause clinical complications, such as obstruction of pulmonary arteries, reactions at the injection site, and sub-acute and chronic immunogenic responses.1
As a result, parenteral drug products must be ‘essentially’ or ‘practically’ free of visible particles according to regulations in Europe and the United States.2
To address discoloration, visible particles, incorrect fill levels, and primary packaging defects, parenteral drug products must undergo visual inspection, which can be performed manually or with automated or semi-automated VI test systems. Effectively implementing VI procedures is crucial to supporting patient safety and minimizing business risks, as visible particulates are a common cause of drug recalls.3
From 2010-2021, the number of drug products recalled by the U.S. Food and Drug Administration (FDA) due to visible particulates being present ranged from a low of six to a high of 25.4
Between 2017 and 2021, one-third of FDA product recalls were due to visible particulates (Figure 1). The percentage has decreased in recent years, but from January 2022 through the end of March 2024, approximately 11 % of FDA recalls were still due to visible particulate contaminants.5
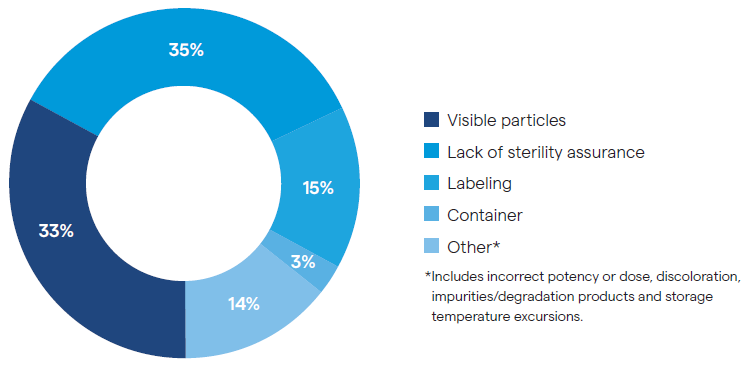
Figure 1. Distribution of FDA Sterile Injectable Drug Recall Notices (2017–2021). Image Credit: FDA Drug Recalls5
The quality of VI test sets directly determines the robustness of visual inspection systems for parenteral drug products; therefore, using the highest-quality test sets is crucial. They support regulatory compliance and operational efficiency, and ensure more robust qualification of operators and equipment, reducing the risk of issues during VI testing.
As an added benefit, high-quality VI test sets have a longer shelf life, thus helping to reduce the long-term costs of VI testing.
This white paper outlines best practices for designing and manufacturing VI test sets, and highlights how Lonza’s comprehensive end-to-end solutions can support quality assurance strategies.
References and further reading
- Doessegger, L., et al. (2012). The Potential Clinical Relevance of Visible Particles in Parenteral Drugs. Journal of Pharmaceutical Sciences, (online) 101(8), pp.2635–2644. DOI: 10.1002/jps.23217. https://jpharmsci.org/article/S0022-3549(15)31495-7/abstract.
- Izi Bruker (2023). The Importance of Visual Inspection in Injection of Parenteral Drug Products. (online) Available at: https://pda.org/pda-letter-portal/home/full-article/the-importance-of-visual-inspection-in-injection-of-parenteral-drug-products.
- Mazaheri, M., et al. (2024). Monitoring of Visible Particles in Parenteral Products by Manual Visual Inspection - Reassessing Size Threshold and Other Particle Characteristics that Define Particle Visibility. Journal of pharmaceutical sciences, 113(3), pp.616–624. DOI: 10.1016/j.xphs.2023.10.002. https://jpharmsci.org/article/S0022-3549(23)00411-2/fulltext.
- John G. Shabushnig (2022). Visual Inspection of Injectable Products: More than Sorting Good from Bad …. (online) Available at: https://www.fda.gov/media/162175/download.
- Center for Drug Evaluation and Research (2019). Drug Recalls. (online) U.S. Food and Drug Administration. Available at: https://www.fda.gov/drugs/drug-safety-and-availability/drug-recalls.
Acknowledgments
Produced using materials originally written by Michael Jahn and Nils Krause from Lonza.
About Lonza
Lonza is one of the world’s largest contract development and manufacturing organizations (CDMOs) dedicated to serving the healthcare industry. Working across five continents, our global team of approximately 20,000 colleagues works alongside pharma and biotech companies to turn their breakthrough innovations into viable therapies. We support our customers in bringing life-saving and life-enhancing treatments to patients worldwide with a combination of cutting-edge science, smart technology, and lean manufacturing.
Our company generated sales of CHF 6.5 billion with a CORE EBITDA of CHF 2.1 billion in Full-Year 2025.
Find out more at www.lonza.com.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.