Research led by Jimmy Gollihar from the CCDC Army Research Laboratory-South in Austin, Texas found the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) variant B.1.1.7 —first detected last fall in England — increased in Houston, Texas. The B.1.1.7 variant is rapidly spreading with a case doubling time of 6.9 days.
Previous work found all variants of concern in the Houston area after genomic surveillance of SARS-CoV-2 samples from March 2020 to February 2021. In early March 2021, Governor Greg Abbott issued an executive order lifting the mask mandate in Texas.
The state currently has the third global highest coronavirus cases by state with over 2.78 million. They also have the third-highest total coronavirus deaths in the United States, reporting over 48,000.
The study “Rapid, widespread, and preferential increase of SARS-CoV-2 B.1.1.7 variant in Houston, TX, revealed by 8,857 genome sequences” is available as a preprint on the medRxiv* server, while the article undergoes peer review.
Tracking genetic variants
The researchers performed genomic surveillance on 8,857 SARS-CoV-2 samples acquired from patients who tested positive for COVID-19. The samples were collected from January 1, 2021, to March 7, 2021. The sample was 94% of patients diagnosed in the Houston Methodist system. Patient information such as place of residence was obtained from electronic medical records to identify the geospatial distribution of the B.1.1.7 variant of concern.
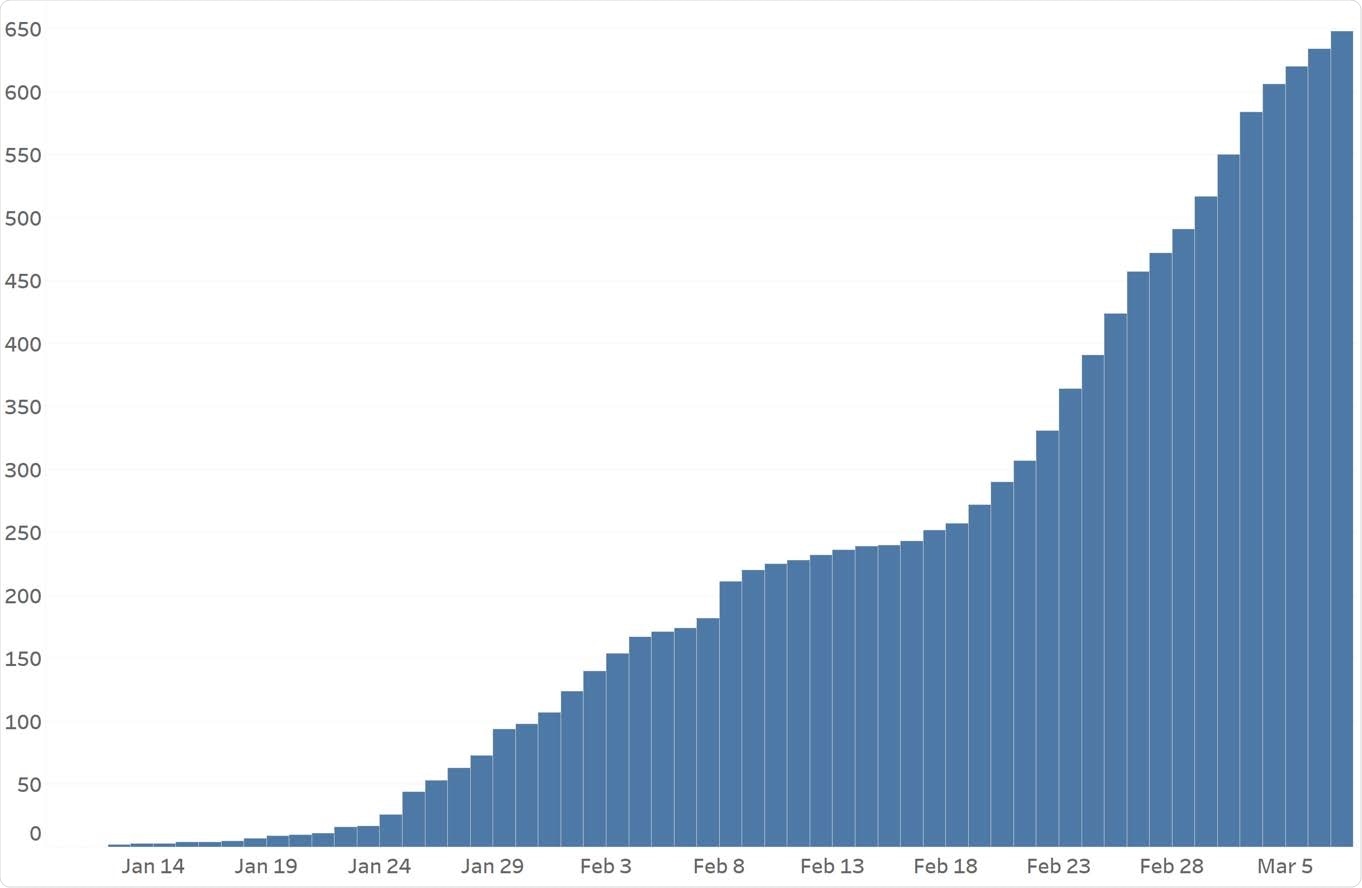
Epidemiologic curve of COVID- 19 cases in the Houston Methodist hospital system. The curve shows the increased frequency of cases caused by B.1.1.7 from January 1, 2021 to March 7, 2021.

 *Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
Prevalence of B.1.1.7 variant in Houston
The researchers found 648 B.1.1.7 cases which became more recurrent than other variants of concern throughout the study period. Altogether, the B.1.1.7 variant was widespread in the entire Houston metropolitan area. They estimate the Houston region doubled their B.1.1.7 cases every 6.9 days.
Fortunately, the team did not find the E484K mutation in the spike protein of any of the 648 B.1.1.7 variants identified. The E484K mutation has been associated with decreasing the immune response and from vaccine-induced neutralizing antibodies. It has been found in other variants of concern, including P.1 and B.1.351.
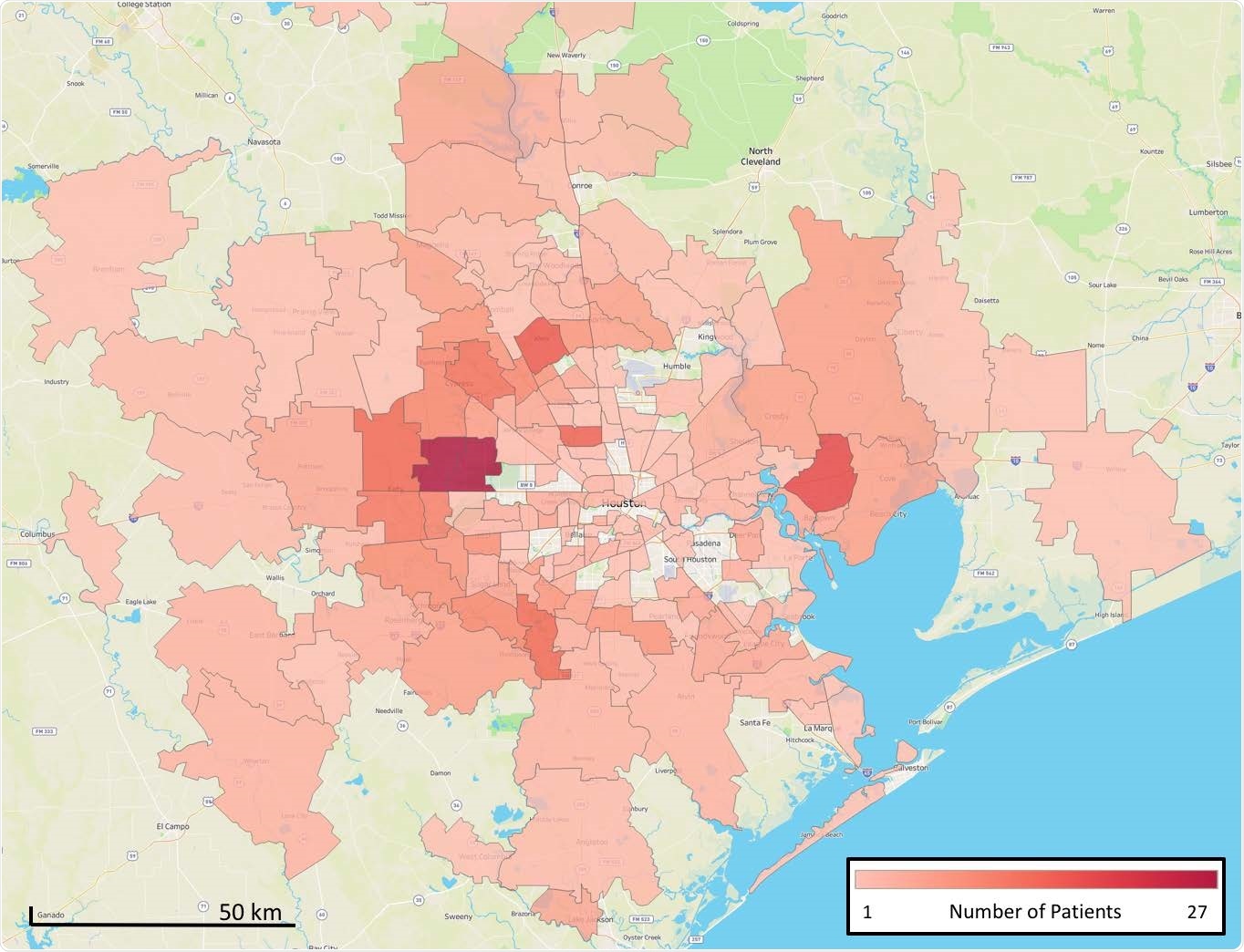
Geospatial distribution of B.1.1.7 cases identified in the study. The home address zip code for each patient was used and figures were generated using Tableau version 2020.3.4.
There was no significant association between infection by the B.1.1.7 variant and a person’s age, gender, length of hospitalization stay, or mortality risk. However, the researchers note patients with the B.1.1.7 variant were more likely to be admitted to a hospital.
Nasopharyngeal samples also found a lower viral load as indicated through lower Ct values, suggesting patients infected with the B.1.1.7 variant had a higher viral load upon initial diagnosis. They suggest the findings are consistent with the increased transmissibility associated with B.1.1.7.
![Growth model for the B.1.1.7 variant in metropolitan Houston. The relative growth of the B.1.1.7 variant was fitted to logistic growth model using the curve fit procedure with nonlinear least-squares estimation in SciPy (1.1.0). Growth rate: 0.1012 [95% confidence interval (CI): 0.0631 - 0.1393]; doubling time: 6.91 days (95% CI: 5.02 - 11.08). Growth model for the B.1.1.7 variant in metropolitan Houston. The relative growth of the B.1.1.7 variant was fitted to logistic growth model using the curve fit procedure with nonlinear least-squares estimation in SciPy (1.1.0). Growth rate: 0.1012 [95% confidence interval (CI): 0.0631 - 0.1393]; doubling time: 6.91 days (95% CI: 5.02 - 11.08).](https://www.news-medical.net/image-handler/picture/2021/3/dsdse-1.jpg)
Growth model for the B.1.1.7 variant in metropolitan Houston. The relative growth of the B.1.1.7 variant was fitted to logistic growth model using the curve fit procedure with nonlinear least-squares estimation in SciPy (1.1.0). Growth rate: 0.1012 [95% confidence interval (CI): 0.0631 - 0.1393]; doubling time: 6.91 days (95% CI: 5.02 - 11.08).
Study limitations to consider
The researchers note their data consists of 94% of Houston Methodist cases and 4.8% of all metropolitan area cases. About 197,817 COVID-19 cases were from Harris County and eight other counties.
The researchers also note their test population was diverse in ethnicity/race, but they may not be translatable to children and overlooked populations such as the homeless.

 *Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.