I am Gilberto Gonzalez, MD, Ph.D., a neuroradiologist at the Massachusetts General Hospital and Professor of Radiology at Harvard Medical School. We began our research into stroke in 1995.
What are large vessel occlusion strokes and ischemic cores?
Large vessel occlusions (LVOs) are occlusions of the major arteries to the brain typically caused by a blood clot coming from outside the head, usually the heart. After occlusion, a part of the brain dies, and this is called the ischemic core.
The core will grow if the clot is not removed. Around a third of all ischemic strokes are due to LVOs, but they cause the majority of deaths and severe disability due to stroke.
Image Credit: Kateryna Kon/Shutterstock.com
Please can you tell us about the current treatments available for large vessel occlusions and their limitations?
We have witnessed a recent revolution in the treatment of these types of strokes. The treatment is called thrombectomy, which is the removal of the clot using a device that is typically passed into the head starting from a large artery in the groin using X-rays for guidance. It is a noninvasive procedure. It is highly effective with the successful removal of clots over 90% of the time.
If the core is small at the time of clot removal, the patient will do very well, they will be either completely normal or have only minor deficits that do not impair their ability to live independently. A major limitation is treatment that occurs when the ischemic core is too large. Identifying patients with small cores is a major goal of imaging.
What has research shown about how ischemic core growth differs between individuals and how thrombectomy success depends on the speed of this growth?
It turns out that the growth of the ischemic core varies widely amongst patients. Our research, confirmed by work at other centers, shows that nearly half of all patients have a slow growth pattern and may be treated 24 hours after stroke onset, and perhaps even later.
What is CT angiography and how can it be used in stroke patients?
CT angiography is an imaging technique that visualizes the major arteries of the head and brain. In patients with ischemic stroke, it is used to identify the presence of LVOs. CT angiograms are created by widely available CT scanners.
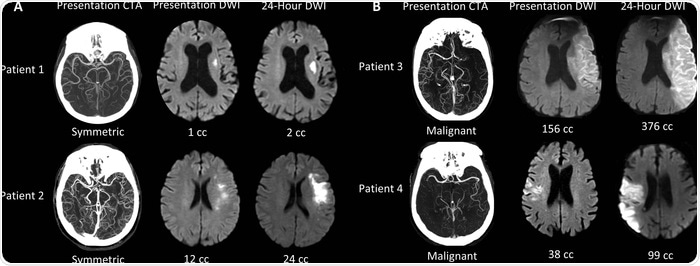
Image Credit: Radiological Society of North America
What link did you find between ischemic core growth and blood flow in collateral blood vessels?
The great variation in the growth of the ischemic core in patients with LVO is due to differences in the collateral circulation. The collateral circulation are arteries that provide blood to parts of the brain that are endangered.
The collaterals come from other large arteries that are not occluded. The variation in collateral blood flow is extraordinarily large, and because of that, the growth of ischemic cores is also very different from patient to patient.
What is the artificial intelligence algorithm you are currently working on and how could this help further examine stroke progression?
We are creating a machine learning algorithm that can identify LVO patients with symmetric collateral circulation that is a marker of small cores that are growing slowly. This is one of several algorithms that we are combining into a suite of algorithms that are capable of capturing all of the key information for the evaluation of the acute stroke patient.
How could this technology be made accessible to everyone and how could these practices be rolled out in hospitals?
We propose the creation of an intelligent network of CT scanners that deduces advanced imaging-type information from routine CT scanners, which are widely available in underserved communities throughout the world. All that is needed is an internet connection.
Advanced imaging-type information will be derived from routine CT scans using the artificial intelligence algorithms that we are creating. The CT scanners would be nodes in a secure, scalable cloud-based network managed by physicians, physicists, and data scientists from a central facility. This center would:
- Import CT imaging data
- Apply the appropriate AI algorithms to derive information on the status of the stroke
- Communicate critical information to all relevant caregivers and stakeholders
Will your research improve stroke patient outcomes?
Yes.
What is the next step for your study?
The next step is investigating whether the pattern of collateral flow on CTA predicts outcomes after thrombectomy. The answer appears to be yes. This information is from a retrospective study that looked at outcomes after thrombectomy over the course of 2 years at our hospital.
This paper has not yet been peer-reviewed, so I must be cautious about drawing definitive conclusions. Also, we are now conducting a prospective study asking the same question.
Where can readers find more information?
The following paper has more information and additional relevant references:
González RG, Silva GS, He J, Sadaghiani S, Wu O, Singhal AB. Identifying
Severe Stroke Patients Likely to Benefit From Thrombectomy Despite Delays of up to a Day. Sci Rep 2020;10(1):4008.
About Professor Gilberto Gonzalez
I am currently Associate Chief of Imaging Sciences and Director of Translational AI Research at the Massachusetts General Hospital. Prior to my current position, I was Chief of Neuroradiology at MGH for 25 years.
During my tenure, the Neuroradiology Division had major research projects on stroke, most notably on using imaging to select patients for thrombectomy.