Post-acute sequelae of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection is estimated to affect about 2% of the population in the United Kingdom (UK). These long-term symptoms of coronavirus disease 2019 (COVID-19), also called post-acute COVID-19 syndrome, post-COVID-19 syndrome, long COVID, or post-COVID condition, cause functional impairment in the majority of those affected.
COVID-19 vaccines have been successful in reducing the rate of incidence of long COVID by lowering the rate of occurrence of SARS-CoV-2 infection. However, the risk for these sequelae post-breakthrough infection remains obscure.
Until the end of January 2022, nearly 16% of the UK population were not fully vaccinated despite being eligible for the second vaccination dose. In addition, most ethnic minorities and deprived communities show lower vaccination coverage; these groups also record the highest infection rates.

Study: Risk of Long Covid in people infected with SARS-CoV-2 after two doses of a COVID-19 vaccine: community-based, matched cohort study. Image Credit: DesignPrax/ Shutterstock

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
The study
A new study posted on the medRxiv* preprint server investigated the difference between the likelihood of symptoms, 12 weeks after SARS-CoV-2 infection, among individuals who received two COVID-19 vaccination doses and unvaccinated individuals.
For this study, data were extracted from the UK COVID-19 Infection Survey (CIS)—which contains samples of more than half a million individuals from the UK community.
CIS participants (age range - 18-69 years) with a positive COVID-19 test report either via polymerase chain reaction (PCR)—using swabs, or any other swab test in national testing programs, between 2020 and 2021.
Findings
Overall, 3,333 participants were selected. Among these, samples of 92.7% of individuals who were double-vaccinated before their first SARS-CoV-2 positive test report, were matched in the ratio of 1:1 with those who were unvaccinated (controls) during the infection.
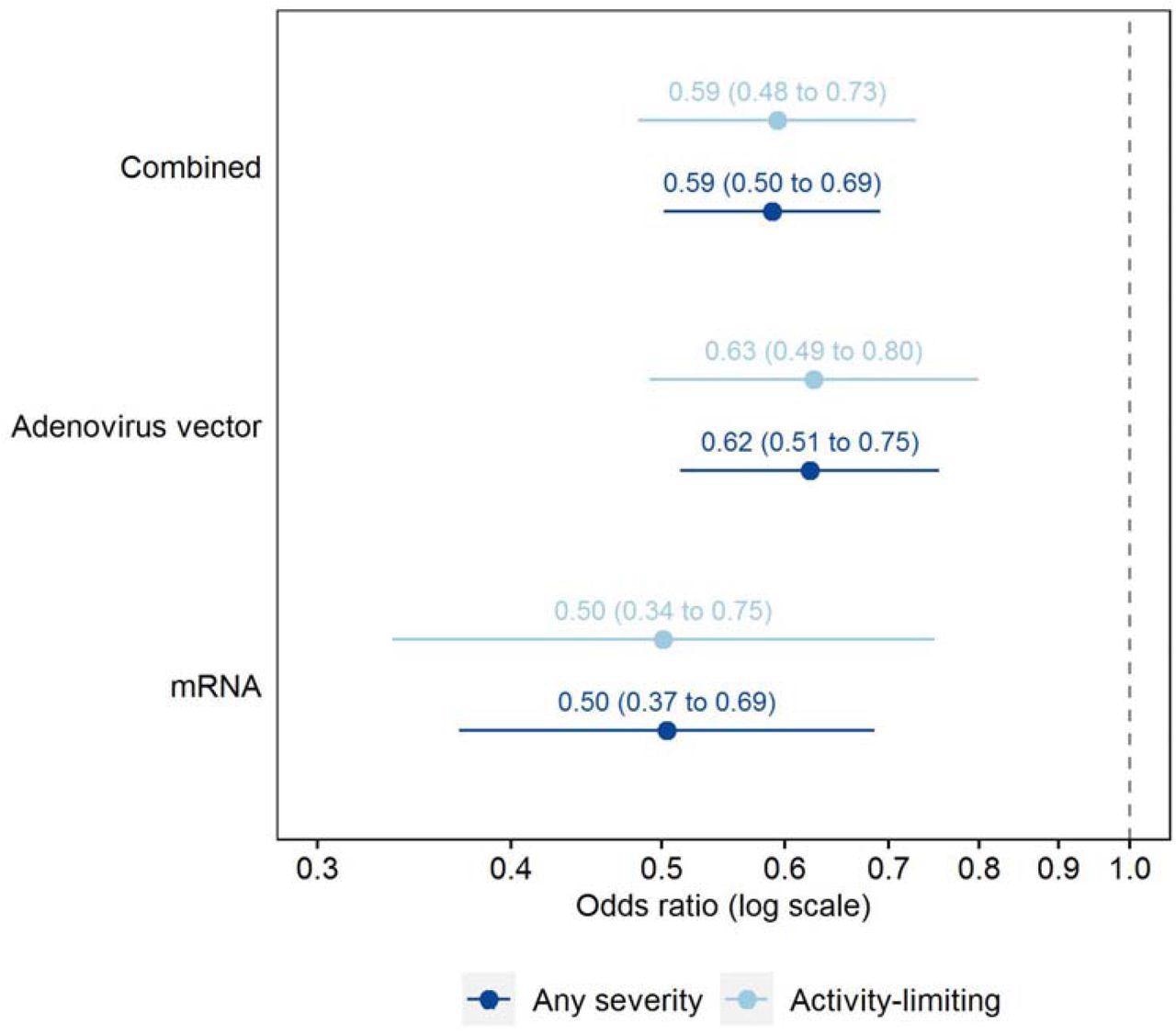
Adjusted odds ratios for Long Covid symptoms ≥12 weeks after first infection, comparing matched study participants who were double-vaccinated or unvaccinated (reference group) before infection Odds ratios adjusted for socio-demographic characteristics (age, sex, white or non-white ethnicity, country/region of residence, area deprivation quintile group, and self-reported, pre-existing health/disability status) and time from infection to follow-up for Long Covid. Confidence intervals are at the 95% level.
It was found that among those who had completed both doses, 2,287 individuals received Oxford/AstraZeneca; 788 Pfizer/BioNTech vaccine; and 15 were given Moderna vaccine. In this group, most were infected during the Delta wave in UK, whereas almost all controls were infected before this phase.
Median follow-ups for long COVID (12 weeks or more post-infection) in the double-vaccinated and control groups were conducted at 96 and 98 days, respectively. In the former group, long COVID symptoms were reported by 294 participants versus 452 controls. While 170 people who received two vaccination doses complained of functional impairments, compared to 268 controls.
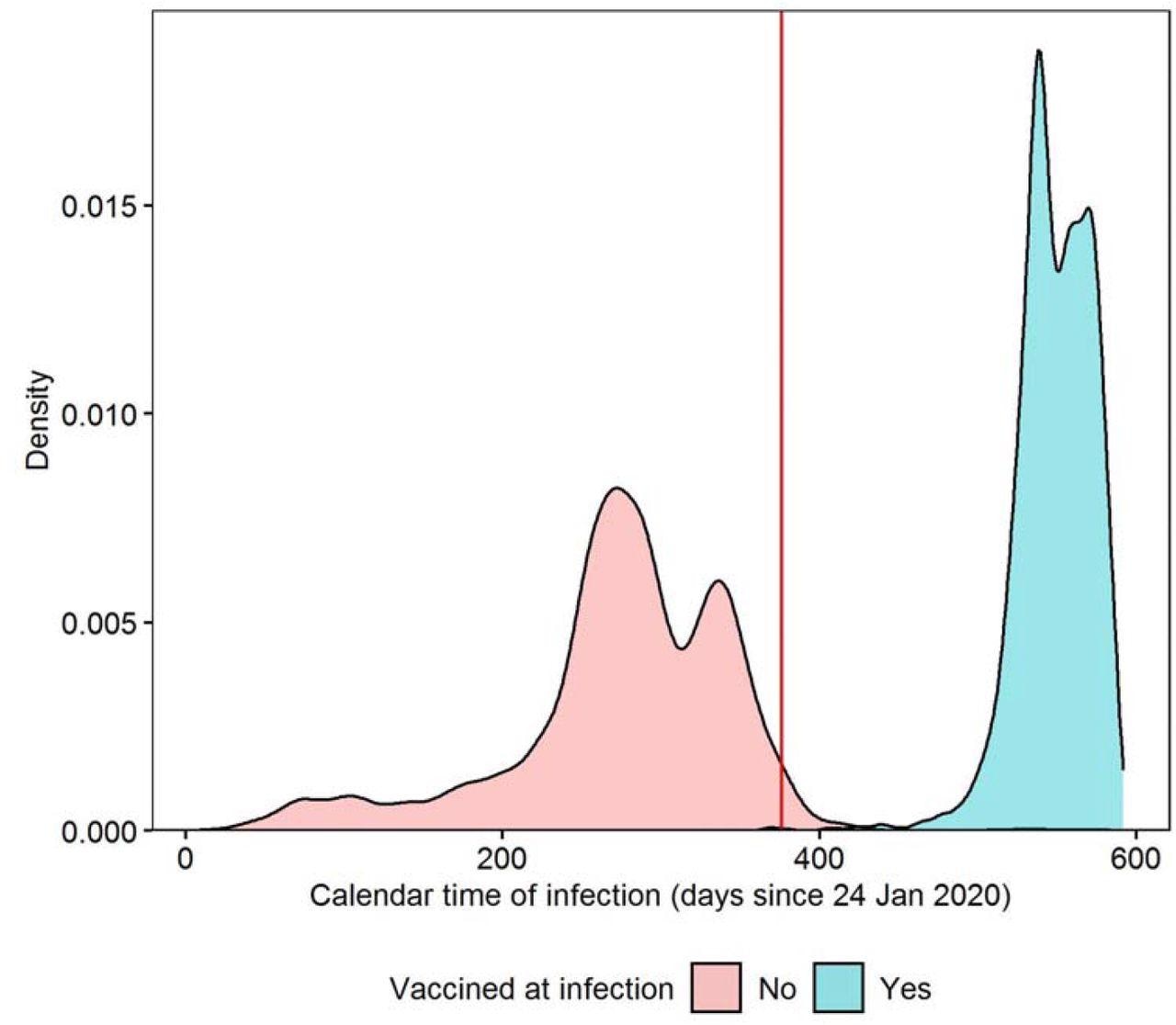
Density plot of calendar time of first infection, stratified by whether study participants were double-vaccinated ≥14 days before infection; the red line indicates the introduction of the survey question on Long Covid on 3 February 2021 Calendar time of infection calculated as the number of days from 24 January 2020, when the first COVID-19 case was reported in the UK. Density estimated from 3,333 double-vaccinated participants and 9,854 unvaccinated participants before matching.
The estimated adjusted odds ratios (aOR) were 0.50-0.69 for long COVID of any kind, and that for activity-limiting symptoms was 0.48-0.73, among the fully vaccinated and control groups. A difference by time from infection to follow-up could not be depicted, nor was there any heterogeneity found among adenovirus vector or mRNA vaccine recipients.
The results hold importance as understanding the benefits of vaccines in preventing long COVID symptoms may help propagate public health awareness and enhance vaccine uptake with appropriate information.
Two doses of COVID-19 vaccination reduce the risk of developing long COVID symptoms 12 weeks post-infection, compared to incomplete or no vaccination. The finding highlighted the need for public health initiatives to improve community awareness and population-level vaccine uptake.
It was also stated that studies including more extended follow-up periods are warranted to assess symptom trajectories after longer post-infection durations and the effects of booster doses and the Omicron variant. Also, investigating the pathophysiology of long COVID can guide future therapeutic strategies.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Ayoubkhani, D., Bosworth, M., King, S., et al. (2022). Risk of Long Covid in people infected with SARS-CoV-2 after two doses of a COVID-19 vaccine: community-based, matched cohort study. medRxiv* preprint. doi: 10.1101/2022.02.23.22271388. https://www.medrxiv.org/content/10.1101/2022.02.23.22271388v1
- Peer reviewed and published scientific report.
Ayoubkhani, Daniel, Matthew L. Bosworth, Sasha King, Koen B. Pouwels, Myer Glickman, Vahé Nafilyan, Francesco Zaccardi, Kamlesh Khunti, Nisreen A. Alwan, and A. Sarah Walker. 2022. “Risk of Long COVID in People Infected with Severe Acute Respiratory Syndrome Coronavirus 2 after 2 Doses of a Coronavirus Disease 2019 Vaccine: Community-Based, Matched Cohort Study.” Open Forum Infectious Diseases 9 (9): ofac464. https://doi.org/10.1093/ofid/ofac464. https://academic.oup.com/ofid/article/9/9/ofac464/6696170.