Ethyl glucuronide (EtG) and ethyl sulfate (EtS) are direct metabolites of ethanol. Despite the fact that just a small portion of the ethanol consumed by humans is transformed into this pair of compounds, they are employed as biomarkers for consumption or abstinence from alcohol, for example, in tests determining fitness to drive.

Figure 1. EVOQ triple quadrupole MS with Elute UHPLC
It is still possible to detect these compounds in urine, even once ethanol concentration can no longer be measured, since their window of detection in this form is around 3-5 days. The typical cut-off value for EtG is approximately 100 ng/mL.
With EtG, there is a risk of both false-negative results, which can happen as a result of bacterial degradation, for example, with a urinary tract infection, and false-positive results, which can happen following the use of ethanol-based mouthwash or hand-sanitizers.
This is why the inclusion of EtS in the assay is vital. A specific detection process capable of picking up both EtG and EtS can be achieved through High Performance Liquid chromatography (HPLC) along with a triple quadrupole mass spectrometer.
Experimental
Samples
Recipe (Munich) provided urine calibrators at six diverse concentrations (Table 2), as well as a quality control sample. Analysis was carried out on two samples of a round robin test arranged by the “Society of Toxicological and Forensic Chemistry” (GTFCh).
Additionally, urine samples of two people (both social drinkers; individual 1 female, 30 y, 61 kg; individual 2 male, 37 y, 64 kg) were gathered 3 and 24 hours after each had ingested one glass of wine (200 mL) and one glass of spirits (20 mL).
Sample Preparation
Urine samples were placed in a centrifuge at 4000 rpm for 10 min. 100 µL urine, 20 µL internal standard solutions (5 µg/ mL EtG-d5 and EtS-d5) and 250 µL methanol were added to an Eppendorf tube.
Following vortexing, the tubes were placed in the centrifuge and 270 µL supernatant were transmitted into an HPLC vial. Evaporation of the supernatant took place in a vacuum concentrator at 45 °C and the residue re-formed in 600 µL 0.1% formic acid.
| Instrumentation |
|
| UHPLC: |
Bruker Elute UHPLC |
| Column: |
Restek, Ultra Biphenyl, 3 µm, 2.1 x 100 mm with guard column 2.1 x 10 mm |
| Mobile phase A: |
Water, 2 mM ammonium formate, 0.1% formic acid |
| Mobile phase B: |
Methanol, 2 mM ammonium formate, 0.1% formic acid |
| Gradient: |
0.0 min 2% B 1.0 min 80% B 2.0 min 80% B 2.01 min 2% B 3.0 min 2% B |
| Flow rate: |
400 µL/min |
| Injection volume: |
10 µL |
| Column oven: |
40 °C |
| Mass spectrometer: |
EVOQ Elite triple quadrupole mass spectrometer |
| Ion source: |
VIP H-ESI negative, 4000 V |
| Probe gas: |
50 units at 300 °C |
| Cone gas: |
25 units at 350 °C |
| Nebulizing gas: |
50 units |
| Active exhaust: |
on |
| Collision gas: |
Argon, 1.5 mTorr |
| MRM transitions: |
see Table 1 |
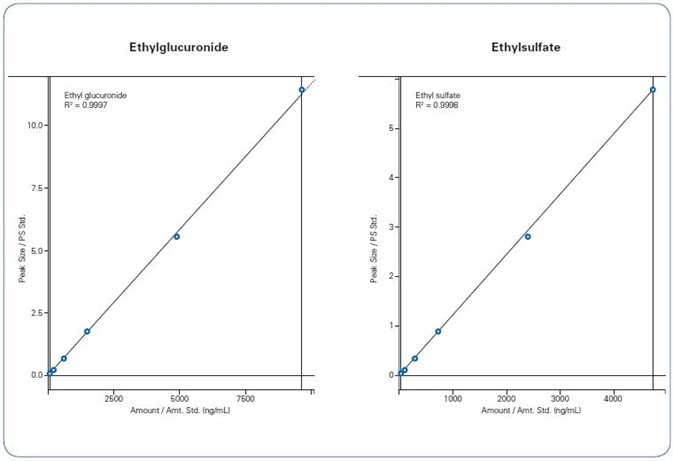
Figure 2. Calibration curves of EtG (left) and EtS (right) over the range 80 - 9621 ng/mL (EtG) and 27 - 4732 ng/mL (EtS)
Results and Discussion
Calibration curves demonstrated exceptional linearity (r² ≥ 0.999) and a high standard of accuracy with bias < ±8% for both EtG and EtS (Figure 2, Table 2). Strong accuracy was also found in the quality control sample, with a deviation of 9.4% for both analytes.
Replicate injections (n=5) of the two round robin test samples revealed outstanding accuracy, with RSD ≤ 2.8% for each compound. The quantitative results all fell inside the accepted range (Table 3).
EtG and EtS were each distinguished in the two genuine samples. The concentrations actually surpassed the calibration range in the samples collected 3 hours after the ingestion of alcoholic drinks. 24 hours after the ingestion, both analytes could still be detected in the two samples (Table 4).
The innovative Elute UHPLC system delivered outstanding chromatographic peak shape and a FWHM (full width half maximum) of just 3-4 seconds, as presented in Fig. 3.
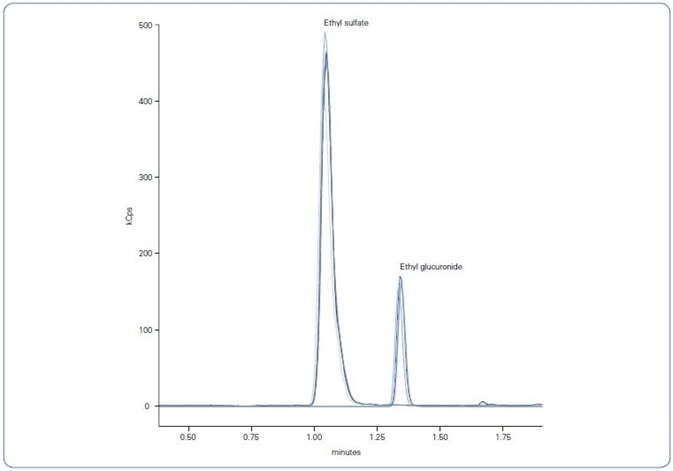
Figure 3. Overlaid chromatograms of the five replicates of round robin test sample 1; left peak EtS, right peak EtG
Table 1. MRM transitions
| Compound |
Rt
(min) |
Precursor
Ion |
Product
Ion 1 |
CID 1
(V) |
Product
Ion 2 |
CID 2
(V) |
| Ethyl glucuronide |
1.34 |
221.0 |
75.2 |
11 |
85.2 |
14 |
| Ethyl glucuronide-d5 |
1.33 |
226.1 |
75.0 |
11 |
|
|
| Ethyl sulfate |
1.08 |
125.0 |
97.1 |
12 |
80.1 |
26 |
| Ethyl sulfate-d5 |
1.07 |
130.0 |
98.0 |
12 |
|
|
Table 2. Calibration and QC of EtG and EtS
| |
Ethyl glucuronide |
Ethyl sulfate |
| Sample |
Target concentration [ng/mL] |
Calculated concentration [ng/mL] |
Bias [%] |
Target concentration [ng/mL] |
Calculated concentration [ng/mL] |
Bias [%] |
| Calibrator 1 |
80.1 |
80 |
-0.1 |
27.8 |
27 |
-2.5 |
| Calibrator 2 |
191 |
192 |
0.6 |
75.9 |
82 |
7.6 |
| Calibrator 3 |
582 |
570 |
-2.1 |
244 |
236 |
-3.2 |
| Calibrator 4 |
1464 |
1505 |
2.8 |
580 |
594 |
2.4 |
| Calibrator 5 |
4873 |
4754 |
-2.4 |
1933 |
1860 |
-3.8 |
| Calibrator 6 |
9621 |
9736 |
1.2 |
4732 |
4705 |
-0.6 |
| Quality control |
111 |
121 |
9.4 |
48 |
53 |
9.4 |
Table 3. Results of round robin test
| Sample |
Analyte |
Target concentration [ng/mL] |
Accepted Range [ng/mL] |
Calculated concentration [ng/mL] (mean, n = 5) |
Accuracy Bias [%] |
Precision RSD [%] (n = 5) |
| Round robin test sample 1 |
EtG |
609 |
399 – 819 |
613 |
0.7 |
2.3 |
| EtS |
886 |
596 – 1176 |
856 |
-3.4 |
2.4 |
| Round robin test sample 2 |
EtG |
1240 |
840 – 1640 |
1558 |
25.6 |
2.8 |
| EtS |
790 |
528 – 1052 |
807 |
2.2 |
2.6 |
Table 4. Results of real samples; * concentration above calibration range
| Sample |
Concentration EtG [ng/mL] |
Concentration EtS [ng/mL] |
| Individual 1: 3 h |
approx. 19,000* |
approx. 9,000* |
| Individual 1: 24 h |
93 |
96 |
| Individual 2: 3 h |
approx. 19,000* |
approx. 8,600* |
| Individual 2: 24 h |
179 |
124 |
Conclusion
A rapid, 3 minute method to detect and quantify EtG and EtS in urine with the new Bruker Elute UHPLC coupled to the EVOQ Elite triple quadrupole MS was demonstrated. The technique offers outstanding linearity and excellent accuracy. Both EtG and EtS were distinguished in the two genuine samples.
About Bruker Daltonics
Empowering Science – Improving Life
Bruker Daltonics delivers cutting-edge mass spectrometry solutions and workflows that help scientists and industry leaders tackle real-world challenges and make new discoveries. From life sciences and pharmaceutical research to food and contaminant analysis, environmental monitoring, forensics, and industrial quality control, our technologies and instruments provide the precision and reliability you need to make confident decisions.
Our innovative platforms - such as timsTOF, scimaX, neofleX, and DART-TQ - combined with advanced software like SCiLS™ Lab, MetaboScape®, and Biopharma Compass®, transform complex data into actionable insights. Breakthrough innovations like Trapped Ion Mobility (TIMS), Omnitrap®, and dual ionization GC-HRMS are redefining what’s possible in mass spectrometry.
Trusted by leading research institutes, universities, government agencies, and industrial partners worldwide, Bruker Daltonics is committed to driving scientific progress and delivering solutions that matter.
(For Research Use Only. Not for use in clinical diagnostic procedures).
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net, which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.