Sponsored Content by LonzaReviewed by Ify IsiborMay 4 2026
Robust and reliable potency assays are essential to the drug development process and constitute a major quality factor. By utilizing DoE, it is possible to efficiently optimize assay conditions, reduce variability, and accelerate research while maintaining scientific confidence.
Primer: The importance of potency assays for biologics
Potency assays are essential for product quality and patient safety. These tests enable a direct measurement of a biologic's functional activity, verifying that the therapy works as intended.
Potency is considered a critical quality attribute (CQA) because it reflects the biological mechanism of action and informs essential decisions throughout the product's lifecycle. Whether you are working with monoclonal antibodies, bispecifics, or fusion proteins, well-designed potency test development is critical for:
- Assessing biological activity and efficacy
- Demonstrating the Mechanism of Action (MoA)
- Ensuring consistency among batches
- Monitoring stability and shelf life
- Meeting regulatory requirements and obtaining permission
- Supporting early development and process modifications
In summary, potency assays are critical instruments for guiding development, ensuring consistency, and protecting therapeutic performance. They provide patients confidence that complicated biologics will work as intended - safely, effectively, and consistently - every time.
Application of Quality by Design principles
For potency test development research, Lonza uses a Quality by Design (QbD) methodology, providing a structured, science- and risk-based framework to ensure assays are robust at launch and reliable throughout their lifecycle.
Guided by ICH guidelines (Q8-Q10 and Q14), QbD promotes a thorough understanding of assay performance and the factors that influence it from the start. At its core, QbD for potency testing entails several steps:
Defining the Analytical Target Profile (ATP)
This is the "start with the end in mind" phase. The ATP defines the assay's intended purpose and establishes unambiguous performance requirements, including accuracy, precision, specificity, linearity, robustness, and stability-indicating capacity.
Identifying Critical Method Parameters (CMPs)
While potency is a critical quality attribute (CQA) of the therapeutic product, the assay's Critical Method Attributes (CMAs), variables such as cell seeding density, incubation duration, and temperature, can have a substantial impact on performance.
Establishing the Method Operable Design Region (MODR)
By analyzing these CMPs using a Design of Experiments (DoE) technique, it is possible to identify the MODR, a multidimensional "safe zone" in which the assay has been proven to meet all ATP requirements, ensuring that minor alterations in lab conditions do not compromise results.
The use of QbD principles shifts research from a reactive, trial-and-error approach to a proactive, knowledge-driven one. This increases assay robustness and reproducibility and facilitates method lifecycle management by allowing for smoother validation, transfer, and adaptation as goods or processes evolve.
Quality by Design (QbD) enables quality to be incorporated from the start, ensuring that potency assays consistently produce fit-for-purpose data throughout development, commercialization, and beyond.
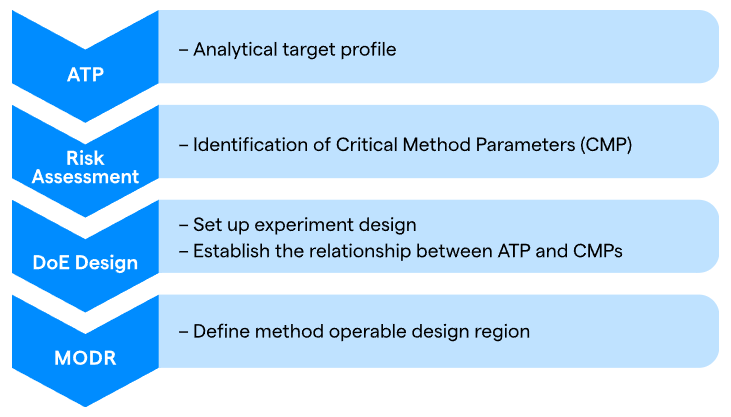
Figure 1. Quality by Design Approach. Image Credit: Lonza
Design of experiments for robust assay development
Assay development is a vital step in pharmaceutical and biological research, typically determining the pace and reliability of drug discovery and manufacturing.
Traditionally, this procedure uses a "one-factor-at-a-time" (OFAT) approach, in which one variable is examined while all others remain constant. While simple in concept, OFAT is time-consuming and resource-intensive, and often fails to uncover the complex connections among many parameters that can affect an assay's performance.
Design of Experiments (DoE) provides a wiser, more efficient approach. DoE enables systematic, data-driven exploration of the experimental design space by adjusting multiple parameters simultaneously, such as incubation duration, temperature, and reagent concentrations.
This strategy reduces the number of tests required, while also providing a better understanding of how crucial method parameters affect key assay properties such as accuracy, precision, and robustness.
DoE's true strength lies in its ability to identify an ideal design space - a robust working range within which the assay performs dependably, even in the face of slight fluctuations that the assay may encounter on a daily basis.
This proactive method incorporates quality into the assay from the beginning, decreasing the need for troubleshooting and redevelopment later on. As a result, DoE shortens development times, increases repeatability, and promotes long-term method sustainability for cell-based assays.
Design of Experiments (DoE) converts assay creation from a reactive process to a strategic enabler of quality and speed, making it a must-have tool for modern biopharmaceutical analytics.
Demonstrating DoE in action: Optimizing a T-cell activation bioassay
In the current era of advanced biologics, such as bispecific antibodies and fusion proteins, T-cell activation bioassays have become crucial tools. These assays confirm the desired treatment mechanism while also detecting any off-target immune responses, ensuring the efficacy and safety of these innovative immunotherapies.
In one recent example, Lonza used QbD and DoE techniques to quickly develop and test the robustness of a T-cell activation bioassay before certification and validation.
The team assessed the potency of two bispecific monoclonal antibodies used to treat B-cell malignancies, Blinatumomab (Blincyto) and Glofitamab (Columvi), using TCR/CD3 Effector Cells (NFAT) and Raji Target cells in culture, as well as Bio-Glo Luciferase assay output.
The team employed a split-plot DoE design to assess how five intra-assay operating parameters affected assay variability. The studies were carried out in a 384-well plate format utilizing a semi-automated pipetting robot.
Surprisingly, the team was able to demonstrate that in a single development run, this structured approach successfully identified the critical method attributes (CMAs) and defined a robust control space for the assay, demonstrating that when DoE principles are applied with a clear understanding of key operational variables, the desired outcomes can be achieved.
By combining DoE with a 384-well format and semi-automated pipetting, the development process was accelerated, made more reliable, and scalable.
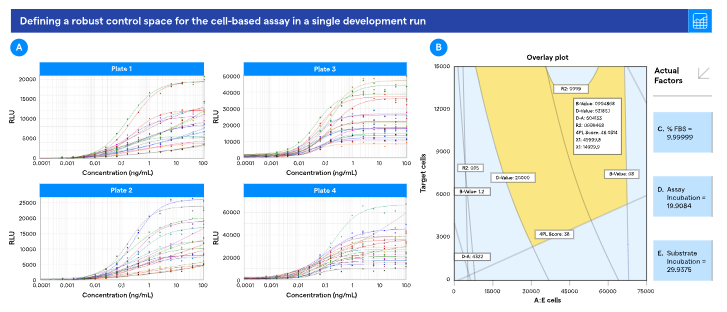
Figure 2. DoE on T-cell Activation Assay. A. DoE dose response curves; B. Defining a robust control space from DoE analysis. Image Credit: Lonza
Conclusion: A scalable framework for potency assay success
The combined use of Quality by Design (QbD) and Design of Experiments (DoE) provides a powerful, scalable framework for creating robust potency assays, whether for T-cell activation or other complex cell-based assay (CBA) formats.
This approach is particularly useful because it allows systematic evaluation of multiple interacting factors, which are frequently at the root of test variability and performance. This methodology is applicable to a wide range of CBA formats, such as reporter gene assays, cytotoxicity assays, proliferation assays, and receptor signaling assays.
DoE unveils the underlying 'why' behind assay results, transforming complex relationships into a clear, visual map.
Combining this with a 384-well format is very intriguing, as it allows researchers to quickly establish a strong MODR, ensuring the method is fail-proof from the start. For Lonza's customers, this means transitioning from trial and error to a process that is statistically assured to be dependable and scalable.
By clearly defining performance targets, identifying essential method parameters, and conducting organized experimentation, Lonza’s teams can accelerate development, reduce variability, and ensure long-term assay sustainability.
Finally, this enables faster, more confident decision-making throughout the biologics lifecycle, promoting both scientific quality and patient benefit for Lonza’s customers.
Acknowledgments
This article was produced using materials originally written by Paola Cecchini, Ph.D., Senior Principal Scientist, Analytical Development, Lonza.
About Lonza
Lonza is one of the world’s largest contract development and manufacturing organizations (CDMOs) dedicated to serving the healthcare industry. Working across five continents, our global team of approximately 20,000 colleagues works alongside pharma and biotech companies to turn their breakthrough innovations into viable therapies. We support our customers in bringing life-saving and life-enhancing treatments to patients worldwide with a combination of cutting-edge science, smart technology and lean manufacturing.
Our company generated sales of CHF 6.5 billion with a CORE EBITDA of CHF 2.1 billion in Full-Year 2025.
Find out more at www.lonza.com.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.