Sponsored Content by LonzaReviewed by Ify IsiborMay 4 2026
Accelerated drug development expertise
For small startups and academic spinoffs bringing their first molecules to market, reaching the clinic represents a major milestone. Reducing the time to investigate a new drug (IND) filing can help to improve the value of their pharmaceutical development programs.
Since 2021, Lonza has been offering an accelerated pharmaceutical development solution for monoclonal antibodies (mAbs). This offering spans a range of development, production, and testing activities – from the creation of expression vectors to the clinical production of therapeutic substances, along with the many activities in between.
The ability to perform activities in an integrated manner enables Lonza to deliver services from DNA to IND (Figure 1) within a shorter timeframe.
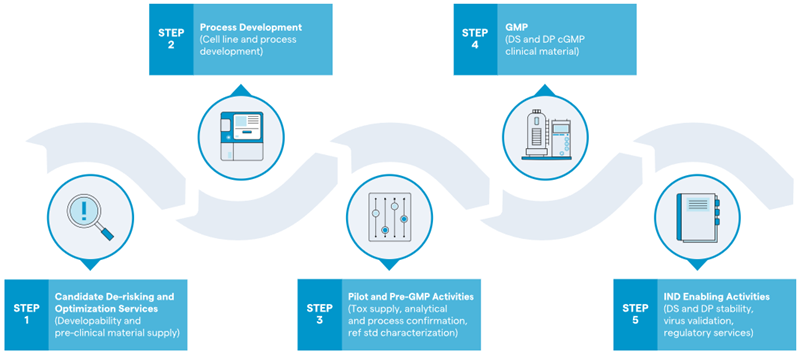
Figure 1. Biologics development pathway, from bench to clinic. Image Credit: Lonza
As the Manager of Global Process Development Support, Andrew Brown leads a team of experts at Lonza with deep knowledge of the process science of upstream and downstream operations for therapeutic protein manufacturing at all production scales.
Recently, Andrew has worked closely with a cross-functional team of SMEs to develop similar integrated offerings for a wide variety of antibody- and protein-based modalities currently under development at many biopharma companies.
Within Lonza, Andrew has gained first-hand experience working with teams across the global network. These teams deliver pharmaceutical development and production activities that accelerate the clinical entry of these molecules. He has also worked with a wide range of expression systems and generated the data required to support successful IND filings. Andrew actively applies this experience to develop improved solutions for Lonza’s clients.
A customized process for IND filings
Molecular diversity poses a challenge for pharmaceutical development and production, as platform approaches may not be suitable. This creates the need for a customized approach to meet product-specific standards.
The first task of the cross-functional team was to establish categories that spanned the range of modalities (Figure 2), enabling the development of tailored offerings. Four categories were chosen:
- Recombinant non-Fc proteins
- Bispecific
- Fc Fusion
- Fab
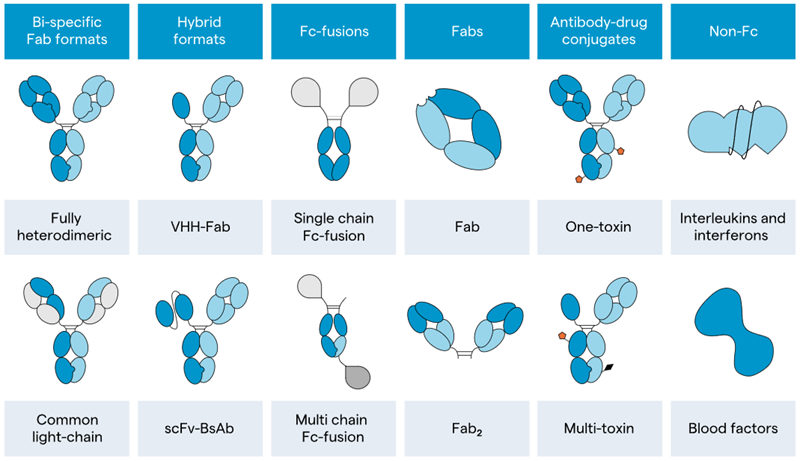
Figure 2. Examples of molecular modalities in which Lonza has experience. Image Credit: Lonza
Lonza closely evaluated the development pathways for these molecules. Through multiple collaborative workshop meetings, they analyzed the interdependencies across pharmaceutical development, production, and testing activities.
This included identifying where information is generated that is necessary for decision-making in other functional areas. These efforts resulted in the identification of a project delivery critical path for each offering.
Shortening DNA-to-IND timelines
Effort was then directed towards optimizing the critical pathways and understanding the basis and assumptions that underpin the approaches. The team’s extensive experience with these diverse modality types, combined with deep process and analytical expertise, was used to identify which operations could be conducted in parallel and to inform the development of highly robust upstream and downstream processes.
Historically, timelines for these more intricate molecules ranged from 22 to 24 months to go from DNA to IND filing. This timeline has now been shortened to 13 months for some bispecific types (Figure 3), representing substantial time savings.
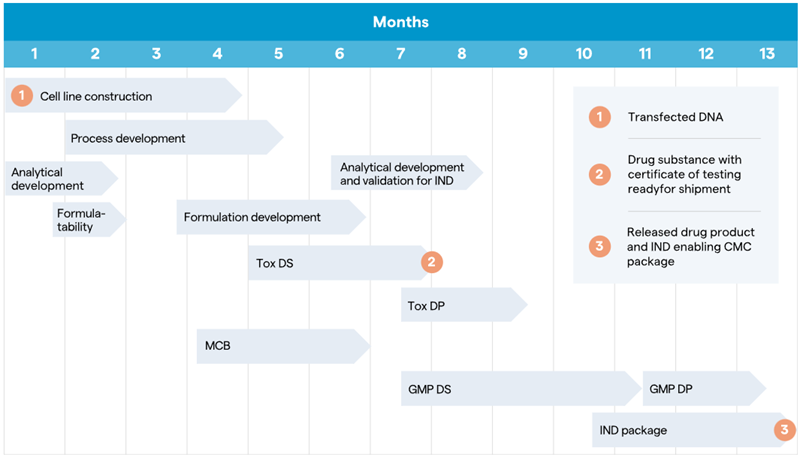
Figure 3. Key stages of the DNA to IND timeline for bispecific antibodies. Image Credit: Lonza
A collaborative approach for faster, integrated drug development solutions for clients
Andrew Brown has worked at Lonza for five years. During this time, he has benefited from opportunities to work across functions, learning about the capabilities of each facility and team across the global network, while collaborating with highly talented and skilled experts.
While he initially viewed this opportunity primarily as a technical challenge, Andrew has come to realize that delivering novel solutions to clients requires close collaboration and open communication across teams that share a common goal.
Andrew has enjoyed working on the development of integrated offerings that support Lonza’s clients, helping them identify opportunities to accelerate timelines while maintaining confidence in the production of high-quality products suitable for clinical trials.
He is proud to see the impact of these integrated offerings and believes that they will benefit patients. Talking to current and potential clients about the integrated offerings and explaining the team’s approach has been enjoyable and insightful, helping to validate and challenge their strategy.
The integrated offerings developed across modalities reflect the team’s current interpretation of the best approach, informed by their knowledge and skills. While not all offerings can benefit from these approaches yet, Andrew and Lonza aim to change this by continuing to learn from how their process and analytical instruments work with the molecules and by working with clients.
Andrew and Lonza look forward to using this feedback to identify further opportunities for growth, enabling them to drive transformative solutions in the future.
Acknowledgements
Produced from materials originally authored by Andrew Brown, Eng.D. (Director, Global Process Development).
About Lonza
Lonza is one of the world’s largest contract development and manufacturing organizations (CDMOs) dedicated to serving the healthcare industry. Working across five continents, our global team of approximately 20,000 colleagues works alongside pharma and biotech companies to turn their breakthrough innovations into viable therapies. We support our customers in bringing life-saving and life-enhancing treatments to patients worldwide with a combination of cutting-edge science, smart technology, and lean manufacturing.
Our company generated sales of CHF 6.5 billion with a CORE EBITDA of CHF 2.1 billion in Full-Year 2025.
Find out more at www.lonza.com.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.