Sponsored Content by LonzaReviewed by Ify IsiborMay 4 2026
The IND Race: Rapid tox material supply as a key to success
The current biotech landscape is defined by rapid pharmaceutical development and constant pressure to move molecules from the laboratory to clinical trials faster than ever before.
For pharmaceutical developers, achieving Investigational New Drug Designation (IND) and/or Clinical Trial Application (CTA) status is both a critical turning point for initiating clinical trials and a key financial milestone that enables companies to secure additional funding for their clinical development journey.
As pharmaceutical developers partner with Contract Development and Manufacturing Organizations (CDMOs) to expedite this process, the rapid toxicology (tox) material supply has emerged as a novel critical path to IND.
Faster tox solutions maximize the impact of rapid DNA-to-IND timelines for accelerated pharmaceutical development
Across the biopharma sector, companies have reported a significant reduction in DNA-to-IND timelines for monoclonal antibodies (mAbs), with health authority applications for first-in-human (FIH) trials completed in months rather than years. Lonza’s Ibex® Design for mAbs is a clear example of such continuous efforts to expedite cutting-edge molecules to the clinic. This program is sufficiently fast to enable clients to achieve DNA-to-IND readiness in as little as 11 months.
Progressing to health authority applications with increasingly faster timelines presents new challenges. Accelerated IND applications require toxicology animal testing, including data analysis and report generation, to be completed within four to five months, a substantial time constraint that leaves no room for delays. With pressure to rapidly advance into FIH trials, the timely provision of toxicology study data is emerging as the next bottleneck.
To address this, Lonza recently introduced two new rapid-tox offerings designed to accelerate the production of toxicology materials - essential for initiating toxicology research - by up to 50 % compared to standard IND timelines for mAbs.
These solutions reduce tox material generation from 5 to 2.5 months without impacting Chemistry, Manufacturing, and Controls (CMC) activities. In this way, Lonza exceeds both client expectations and changing market demands.
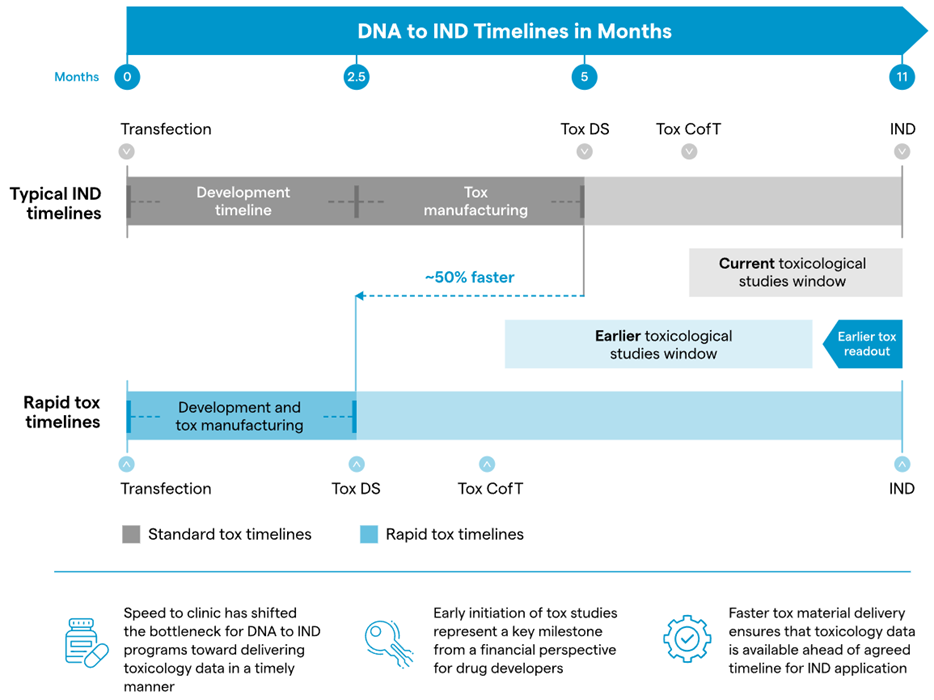
Figure 1. Estimated timelines for our accelerated tox offering for mAbs, which can reduce timelines for tox material supply by 50 %. Image Credit: Lonza
Leveraging industry expertise to improve pharmaceutical development with purpose
As a Principal Scientist of Global Process Development, Alejandro Fernandez-Martell is dedicated to identifying better solutions for both Lonza’s clients and operations. He is passionate about science and technology and eager to find ways to enhance operational excellence.
Recently, Alejandro has led a team of experts at Lonza focused on modernizing their drug material supply timeline for therapeutic proteins. Together, the team is pushing boundaries to accelerate toxicology material delivery, the new critical path for FIH trials, for a wide variety of protein modalities, while reducing production risk and satisfying market demands.
Developing innovative offerings is a complex task that requires close collaboration across several departments, including Process Development, Analytics, Operations, Business and Commercial Development, and Marketing. Every step demands careful coordination, drawing on specialized knowledge to ensure solutions are scientifically robust, in line with business objectives, and effectively meet market demands.
The impact of collaboration in supporting clients on their path to the clinic
With great dedication, Alejandro and the team strike a balance between collective and individual contributions to foster an environment where the expertise and unfaltering commitment of every team member are combined to deliver solutions that meet current client demands.
These efforts have not only shortened development timelines and reduced costs but also deepened Alejandro’s understanding of the complexities clients face when bringing life-changing therapies to clinical trials.
As a scientist at Lonza, Alejandro works closely with diverse teams of experts across the global network. He has channeled their expertise and passion to deliver integrated solutions that accelerate timelines while ensuring the production of high-quality products for clinical trial use.
Each stage, from accelerating toxicology material generation to minimizing risk, has been a collaborative effort. The experience has enhanced Alejandro’s understanding of the business and the critical role Lonza plays in the industry.
Gaining extra time for comprehensive toxicology research
Clients can start toxicology research earlier than before thanks to rapid-tox material delivery. In an industry where time is in short supply, these novel rapid-tox offerings are highly valuable and transformative, providing up to 2.5 months of additional time capacity for clients to complete tox research and meet expedited IND timelines.
Acknowledgements
Produced from materials originally authored by Alejandro Fernandez-Martell, Ph.D. (Principal Scientist, Global Process Development).
About Lonza
Lonza is one of the world’s largest contract development and manufacturing organizations (CDMOs) dedicated to serving the healthcare industry. Working across five continents, our global team of approximately 20,000 colleagues works alongside pharma and biotech companies to turn their breakthrough innovations into viable therapies. We support our customers in bringing life-saving and life-enhancing treatments to patients worldwide with a combination of cutting-edge science, smart technology, and lean manufacturing.
Our company generated sales of CHF 6.5 billion with a CORE EBITDA of CHF 2.1 billion in Full-Year 2025.
Find out more at www.lonza.com.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.