Aug 30 2018
Chemists at Hong Kong Baptist University (HKBU) have discovered the use of a metal compound that inhibits the enzyme closely associated with triple-negative breast cancer (TNBC), one of the most difficult forms of breast cancer to treat.
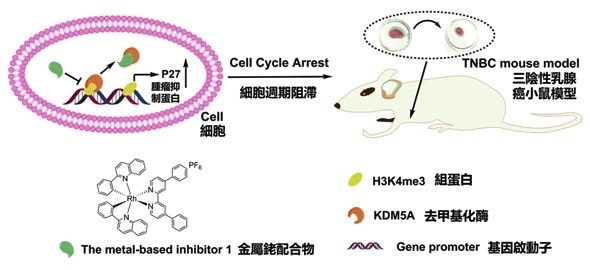
The diagram shows the use of the new inhibitor for the treatment of triple-negative breast cancer.
The metal compound is found to inhibit the TNBC tumors with less toxicity in mice, thus their work has further unmasked the role of the enzyme, lysine-specific demythylase 5A (KDM5A), in TNBC. The reported compound shows tremendous potential for the development of drugs for TNBC therapy. The results of this study were published in the leading chemistry journal Angewandte Chemie International Edition.
The team was led by Dr Edmond Ma Dik-lung, Associate Professor of HKBU’s Department of Chemistry. The team developed the compound using a metal named rhodium as a direct, potent and selective inhibitor of KDM5A, in other words the compound can effectively target KDM5A which is a key limitation of existing KDM5A inhibitors.
Dr Edmond Ma said that KDM5A is overexpressed in breast cancer and other human cancers, including brain, lung and gastric cancers, thus promoting carcinogenesis and drug resistance. Up to now, only a few KDM5A inhibitors have been reported, but most of them cannot effectively detect KDM5A, limiting their ability to inhibit it.
He said:
The compound acts by disrupting the interaction between KDM5A and trimethylated histone H3, a protein that regulates the folding of DNA. This leads to the accumulation of methylated marks on histone H3, which in turn increases the expression of p27, a protein that suppresses tumor growth. The compound was found to be capable of suppressing cells’ growth process in TNBC cell lines, and also showed potent anti-tumor activity against TNBC in an in vivo mouse model. Importantly, the new compound was less toxic in vivo compared with two well-known clinical anti-cancer drugs, cisplatin and doxorubicin.”
Dr Ma said:
The traditional TNBC treatments are limited by drug resistance and serious side effects, including organ damage. Therefore, there is an urgent need to develop new targeted approaches for the treatment of TNBC. We therefore believe that our metal compound shows great promise for the development of targeted drugs for TNBC therapy.”