CBD may help in epilepsy and some psychiatric conditions, but this review shows why weak evidence, inconsistent products, and safety risks still stand in the way of wider medical use.

Circumspective: Cannabidiol at the crossroads: panacea, placebo, or problem? Image Credit: Creativan / Shutterstock
In a recent Circumspectives article published in the journal Neuropsychopharmacology, researchers presented two perspectives on the diverse pharmacological (e.g., neuropsychiatric) applications of cannabidiol (CBD) amid ongoing clinical risks associated with its unprecedented worldwide consumption.
The review reveals that while CBD has already shown promise for treating conditions like pediatric epilepsy and anxiety, significant concerns regarding evidence quality and product safety persist. It highlights issues with labeling accuracy, potential hepatotoxicity, and drug interactions, thereby underscoring the need for standardized research and rigorous regulatory oversight.
CBD Use, Popularity, and Evidence Gaps
Cannabidiol (CBD) is a major phytocannabinoid derived from the Cannabis sativa plant. Unlike its more famous sibling, delta-9-tetrahydrocannabinol (THC; more commonly called “THC”), CBD is non-intoxicating and has demonstrated low abuse liability in human laboratory studies.
CBD’s current therapeutic popularity stems from its perceived safety and its pleiotropic (affects several biological targets simultaneously) nature. Hemp, CBD’s primary source, has recently been removed from the controlled substance lists in many jurisdictions, resulting in a “boom” in the phytocannabinoid’s distribution and consumption. An unprecedented number of consumers now report using CBD for general wellness, pain management, and anxiety.
Unfortunately, this “CBD boom" has rapidly outpaced the rigorous scientific validation required for medical use, with most of the evidence supporting the phytocannabinoid’s use being categorized as "immature". Studies on the topic emphasize that this gap between commercial availability and scientific validation has created a landscape where product labeling is often inaccurate and therapeutic claims remain speculative.
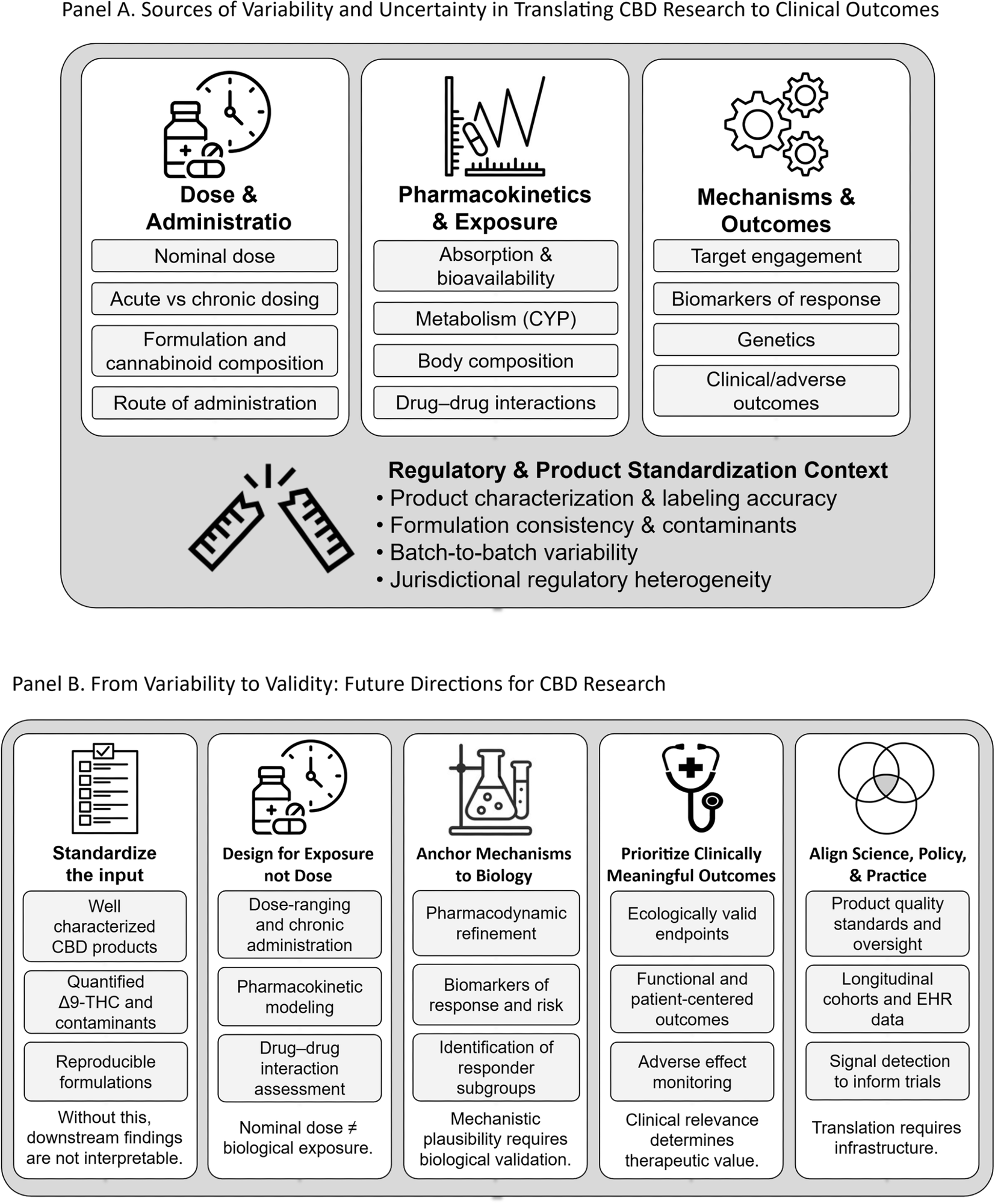
A Sources of Variability and Uncertainty in Translating CBD Research to Clinical Outcomes. Key concepts demonstrating how variability at the levels of dosing, pharmacokinetics, and mechanistic translation contributes to inconsistent findings in the CBD literature. The model highlights the need for integrated study designs that link administered dose to biological exposure and clinically meaningful outcomes. B Figure 1, Panel B depicts a roadmap illustrating strategies to reduce variability and improve clinical translation in CBD research. Aligning standardized and well-characterized products, exposure-informed trial design, biomarker-anchored mechanisms, clinically meaningful outcomes, and regulatory oversight is essential for defining CBD’s therapeutic utility.
CBD Mechanisms and Review Scope
The present Circumspectives article aimed to address this knowledge gap by discussing findings from preclinical models, acute human laboratory paradigms, and early-phase clinical trials to present a balanced view of CBD's therapeutic potential and current limitations.
The review mapped CBD’s pharmacodynamic interactions with a focus on four main subtopics:
- Serotonergic (5-HT1A) agonism, which has been linked to potential anxiolytic (anti-anxiety) and antidepressant effects.
- Transient Receptor Potential (TRPV1) channels, which may contribute to CBD’s analgesia (pain relief) and temperature regulation benefits.
- Endocannabinoid System: Studies have found that CBD acts as a negative allosteric modulator of CB1 receptors and increases anandamide (an endogenous cannabinoid) levels by inhibiting fatty acid amide hydrolase (FAAH).
- Peroxisome Proliferator-Activated Receptors (PPARs): Emergent evidence suggests that CBD-activated PPARs may help regulate neuroinflammatory cascades and could be relevant to age-associated neurodegeneration.
When investigating the scientific and safety risks of the ongoing CBD boom, the review addressed the "heterogeneity" of the market, examining how different product formats (oils, vapes, topicals) and chemical compositions (isolates vs. full-spectrum) affect how the body absorbs the compound and its resultant downstream physiological impacts.
CBD Efficacy Evidence and Safety Concerns
The review emphasizes a stark dichotomy between clinically validated pharmaceutical successes (predominantly in therapeutic interventions against epilepsy) and inconsistent retail data. It found that the most robust evidence that currently exists for CBD (Epidiolex) lies in its validated ability to treat rare disorders like Dravet and Lennox-Gastaut syndromes (pediatric epilepsy).
Emergent evidence suggests that CBD use may reduce cue-induced craving and anxiety in individuals with opioid use disorder. Furthermore, a comparative study found that CBD had antipsychotic effects similar to the drug amisulpride, but with a superior side-effect profile, underscoring its potential in psychiatry.
Most notably, a systematic review of clinical studies showed moderate therapeutic benefit for pure CBD in social or generalized anxiety disorders. However, the same analysis found no effect when CBD was combined with THC.
In contrast to these limited clinical successes, the review identified several safety and quality concerns regarding CBD use. Research has shown that chronic exposure to high-dose CBD can negatively impact hepatic (liver) function (dose-dependent).
Furthermore, CBD was found to be a potent inhibitor of multiple CYP450 enzymes (the known targets of several commonly used drugs). CBD use can potentially slow the metabolism of these medications, significantly increasing the risks of suboptimal drug-drug interactions and adverse events.
Most alarmingly, the review highlights that current standardization and public health supervision protocols are often inadequate in the retail CBD market. This regulatory oversight is confirmed by product surveillance studies, which have repeatedly shown poor dose label accuracy and the presence of contaminants (predominantly THC and, less commonly, harmful chemicals) in retail products.
Future CBD Research and Regulation Needs
This review establishes CBD as a compound of significant pharmacological potential, but it is currently not a “miracle cure”. The authors conclude that the current evidence base is insufficient for broad clinical adoption.
Furthermore, the observed variability in "interindividual" pharmacokinetics means that the administered dose often poorly predicts actual systemic exposure, complicating attempts to establish consistent dose-response relationships and clinically meaningful exposure thresholds.
The review highlights the urgent need for well-powered, randomized clinical trials (RCTs) that utilize chronic dosing paradigms and ecologically valid outcomes before CBD’s true medical utility can be realized and the compound can be more appropriately integrated into evidence-based clinical and consumer settings.