Preclinical studies increasingly place circular RNAs at the intersection of thermogenic programming and organelle stress control, raising the possibility that these stable RNA loops could eventually inform how metabolic disease is diagnosed and treated.
Cellular changes and associated mechanisms of fat transformation: fat browning is regulated by the central nervous system, peripheral nervous system, transcription factors, environmental factors, pharmacological agents.
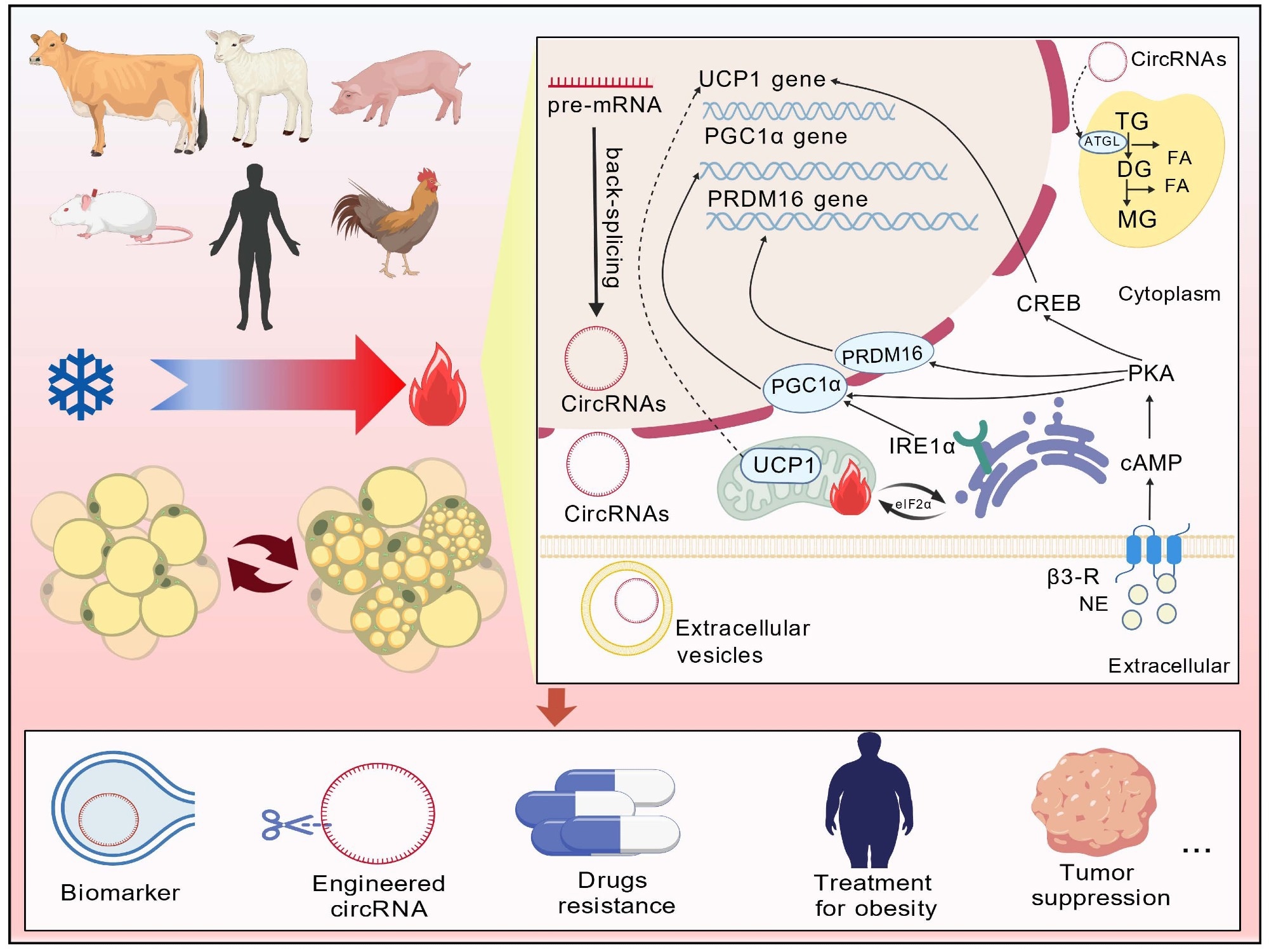
In a recent review published in the journal Cell Death & Disease, researchers describe circular ribonucleic acids (circRNAs) as emerging regulators of fat browning and metabolic health, with emphasis on mitochondria- and endoplasmic reticulum (ER)–linked mechanisms that shape cellular stress responses and energy homeostasis.
These stable, closed-loop RNA molecules can regulate gene expression by sponging microRNAs, binding proteins, and, in some cases, encoding functional peptides. Through effects on lipid metabolism, insulin sensitivity, and energy balance, circRNAs have been linked to obesity, type 2 diabetes (T2D), and metabolic syndrome.
The review discusses circRNAs as candidate diagnostic indicators and therapeutic targets, but notes that delivery efficiency and tissue specificity remain major barriers to clinical translation.
Obesity, Adipose Tissue Dysfunction, and the Rationale for Browning
Obesity is a major risk factor for chronic diseases such as cardiovascular disease, chronic kidney disease, and non-alcoholic fatty liver disease (NAFLD). These complications are associated with dysfunctional white adipose tissue (WAT), which stores excess energy, whereas brown adipose tissue (BAT) expends energy as heat.
Consequently, converting white adipocytes to brown or beige ones has been proposed as a therapeutic approach. The review summarizes evidence that circRNAs regulate adipogenesis and thermogenic programming, while noting open questions about mechanisms and safe, targeted clinical application, particularly given that much of the current evidence comes from cell and animal models.
Scope and Focus of the Review
The review examines circRNA function in WAT browning and lipid metabolism. It outlines proposed mechanisms, therapeutic applications for obesity-associated metabolic dysfunction, and strategies to improve translational relevance. A central theme is organelle-specific pathways involving mitochondria and ER stress responses, alongside the complexity and context-dependent nature of circRNA regulatory networks.
CircRNAs as Regulators of Metabolism and Fat Browning
Circular RNAs are covalently closed, non-coding RNA molecules generated through back-splicing, a structure that confers stability and resistance to degradation. Their persistence in tissues, serum, and exosomes, combined with cell-specific expression patterns, has made them of interest in metabolic homeostasis. CircRNAs influence gene expression through multiple mechanisms, most notably by acting as microRNA sponges, interacting with RNA-binding proteins (RBPs), and, in some cases, encoding functional peptides.
In metabolic disorders, studies have associated aberrant circRNA expression with glucose intolerance, lipid dysregulation, and chronic inflammation. Mechanistically, circRNAs may modulate fat browning and increase thermogenesis and energy expenditure by relieving microRNA-mediated repression of thermogenic genes. For instance, circOgdh regulates lipolysis in BAT through the miRNA-34a-5p/ATGL pathway, whereas circNrxn2 regulates WAT browning through the miR-103/FGF10 pathway. CircRNF111, identified in bovine adipocytes, sponges miR-27a-3p to increase PPARγ expression and is associated with enhanced UCP1 expression, promoting thermogenic differentiation in that model system.
In addition to acting as miRNA sponges, circRNAs can interact with metabolic proteins. CircACC1 promotes AMP-activated protein kinase (AMPK) complex assembly and activation by binding its regulatory subunits, which stimulates β-oxidation and glycolysis under metabolic stress, whereas circH19 and circMBOAT2 regulate lipid synthesis pathways. Some circRNAs, such as circ-SLC9A6 and circ-CUX1, encode peptides that modulate mitochondrial function, mitophagy, and lipid reprogramming.
Taken together, these reports suggest circRNAs contribute to adipocyte plasticity, organelle function, and broader metabolic remodeling, although the strength of evidence varies across experimental systems.
Diagnostic and Therapeutic Applications in Metabolic Disease
CircRNAs are being explored as diagnostic and therapeutic modalities in metabolic disorders. Their stability in blood and other body fluids supports potential use in long-term disease monitoring. For example, elevated hsa_circRNA_0039480 in early pregnancy may help predict gestational diabetes, while circGlis3 has been linked to obesity-related pancreatic β-cell dysfunction, highlighting tissue-specific diagnostic potential.
As therapeutic targets, circRNAs modulate major metabolic pathways, including adipogenesis, insulin secretion, and lipid metabolism. CircSAMD4A inhibits pathological adipogenesis by sponging miRNA-138-5p and relieving EZH2 repression, while circGlis3 stimulates insulin gene expression by regulating the miRNA-124-3p/NeuroD1/Creb1 axis. In T2D, ciRS-7 and circHIPK3 promote beta-cell expansion and insulin release.
In NAFLD, circ_0057558 has been shown to mitigate hepatic lipid accumulation via the ROCK1/AMPK pathway, while decreased expression of circRNA SCAR has been implicated in metabolic inflammation.
New therapeutic strategies are increasingly centered on circRNAs themselves. These include the development of synthetic circRNA mimics or inhibitors, clustered regularly interspaced short palindromic repeats (CRISPR)-based gene editing to modulate circRNA expression, and targeted RNA therapies delivered through lipid nanoparticles, viral vectors, or engineered exosomes. Their intrinsic stability and capacity to encode functional peptides also support circRNAs as experimental vaccine platforms.
The review also notes that circRNAs may influence neurodegenerative, immune, and oncologic conditions, particularly through regulatory effects on fat browning, inflammation, and mitochondrial function.
Challenges and Future Directions
According to the authors, despite strong preclinical evidence, several challenges must be addressed before circRNA-based strategies can be implemented in clinical practice. Key knowledge gaps remain regarding organelle-specific functions, interconnected signaling pathways, and long-term biological effects.
In addition, most findings derive from animal studies, and limited cross-species conservation complicates translation to humans, highlighting the need for validation in large, well-characterized patient cohorts.
The review also emphasizes technical constraints, including low endogenous abundance, transient expression, delivery efficiency, tissue specificity, and potential off-target effects, as well as challenges in precise detection, quantification, and functional validation of circRNAs in human tissues.
Advances in lipid nanoparticle systems, engineered exosomes, CRISPR-based editing, and synthetic circRNA design, together with multi-omics integration and artificial intelligence-assisted functional prediction, may support the development of more precise and clinically applicable circRNA-based interventions.
Journal reference:
- Huang, Y., He, T., Zheng, J., Chen, J., & Yang, Z. (2026). Circular RNAs in metabolic health: Bridging the gap between molecular biology and therapy. Cell Death & Disease. DOI: 10.1038/s41419-026-08450-5, https://www.nature.com/articles/s41419-026-08450-5