Influenza viral nucleoprotein (NP) is an essential structural protein that is a major factor in virus replication and host adaptation. Antibodies designed to target NP proteins are frequently employed in the immunodetection of influenza viruses in various assays; for example, lateral flow assay (LFA), ELISA and direct fluorescent antibody tests.
These assays are suitable for use in research and clinical diagnostics. The high frequency of antigenic shift or drift exhibited by various influenza strains makes broad-spectrum influenza antibodies an especially useful tool for flu diagnostics.
Sino Biological has successfully identified six Pan antibody pairs able to act against the NP protein of Influenza A and Influenza B, respectively. These matched pairs have been validated as delivering high, pg-level sensitivity in the sandwich ELISA while offering broad reactivity against a sizable cohort of flu strains.
Antibody pairs for Pan Influenza A NP detection
Table 1. Source: Sino Biological Inc.
| Antibody Pairs |
Capture Ab |
Detection Ab |
| PanA-1 |
40205-MM16 |
40208-R014 |
| PanA-2 |
40205-R063 |
40205-MM18 |
| PanA-3 |
40208-R117 |
40205-MM18 |
Antibody pairs for Pan Influenza B NP detection
Table 2. Source: Sino Biological Inc.
| Antibody Pairs |
Capture Ab |
Detection Ab |
| PanB-1 |
40438-R004 |
40438-MM10 |
| PanB-2 |
40438-R036 |
40438-MM10 |
| PanB-3 |
40438-MM05 |
40438-R036 |
High sensitivity (pg level)
Image Credit: Sino Biological Inc.
Broad spectrum and high specificity
Antibody pairs from Sino Biological can be used to detect a wide range of strains within the target subtype, ensuring there is no cross-reactivity with other subtypes. A total 24 kinds of influenza antigens from A or B type flu have been tested via ELISA assay, and it has been confirmed that this combination of A and B antibody pairs can facilitate detection of both subtypes.
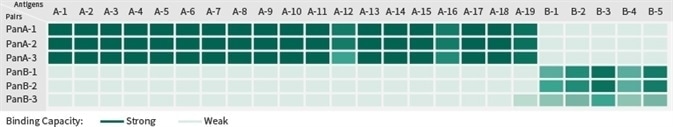
Image Credit: Sino Biological Inc.
High binding affinity
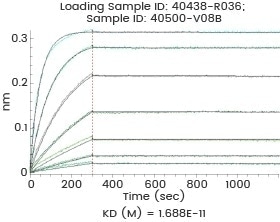
Image Credit: Sino Biological Inc.
Antibody cocktail can successfully detect both A and B type flu
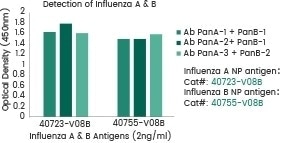
Image Credit: Sino Biological Inc.
High production quality
The company’s products and processes are ISO 13485 certified, and SEC-HPLC has been used to confirm that the purity of these antibodies is >95%.
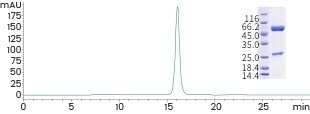
Image Credit: Sino Biological Inc.
Other Pan Influenza NP antibodies
A total of 16 Pan Influenza A or B Nucleoprotein antibodies have also been developed, and these have been successfully validated via ELISA and WB assays.
Pan Influenza A NP antibodies
Table 3. Source: Sino Biological Inc.
| . |
. |
. |
| 40208-MM03 |
40208-R014 |
40208-MM04 |
| 40208-R010 |
40208-R061 |
40208-R113 |
| 40208-R117 |
40205-MM16 |
40205-MM18 |
| 40205-R063 |
|
|
Pan Influenza B NP antibodies
Table 4. Source: Sino Biological Inc.
| . |
. |
. |
| 40438-MM05 |
40438-R016 |
40438-MM09 |
| 40438-R036 |
40438-R004 |
40438-MM10 |
Learn more antibodies for influenza antigen test.
About Sino Biological Inc.

Sino Biological is an international reagent supplier and service provider. The company specializes in recombinant protein production and antibody development. All of Sino Biological's products are independently developed and produced, including recombinant proteins, antibodies and cDNA clones. Sino Biological is the researchers' one-stop technical services shop for the advanced technology platforms they need to make advancements. In addition, Sino Biological offers pharmaceutical companies and biotechnology firms pre-clinical production technology services for hundreds of monoclonal antibody drug candidates.
Sino Biological's core business
Sino Biological is committed to providing high-quality recombinant protein and antibody reagents and to being a one-stop technical services shop for life science researchers around the world. All of our products are independently developed and produced. In addition, we offer pharmaceutical companies and biotechnology firms pre-clinical production technology services for hundreds of monoclonal antibody drug candidates. Our product quality control indicators meet rigorous requirements for clinical use samples. It takes only a few weeks for us to produce 1 to 30 grams of purified monoclonal antibody from gene sequencing.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.