Cancer remains a major global public health concern, and examining the tumor microenvironment helps to improve knowledge associated of cancer.
The tumor microenvironment is often referred to as the internal environment for the production and survival of tumor cells.
This includes thymus-dependent lymphocytes, burse-dependent lymphocytes, dendritic cells, tumor-associated macrophages, natural killer lymphocytes, myeloid-derived suppressor cells (MDSCs), cytokines, chemokines and growth factors in nearby areas.1
MDSCs are the primary immunosuppressive cells maintaining cancer progression in the tumor microenvironment. They can be utilized as potential therapeutic targets and prognostic markers in cancer patients to eradicate immunosuppressive activities and improve antitumor immune responses.
MDSCs inhibit the host’s antitumor immune response and play a central role in tumor promotion and development. Due to this, MDSCs in the tumor microenvironment are the main target for improving the efficiency of tumor immunotherapy.
MDSC-targeting immunotherapy strategies
Immunotherapy is one of the strategies that can be used to target MDSCs, and these include therapies that inhibit the infiltration of MDSC populations, anticytokine therapy, transport inhibition and MDSC recruitment. Recent studies on MDSCs have provided novel directions for cancer treatment.2
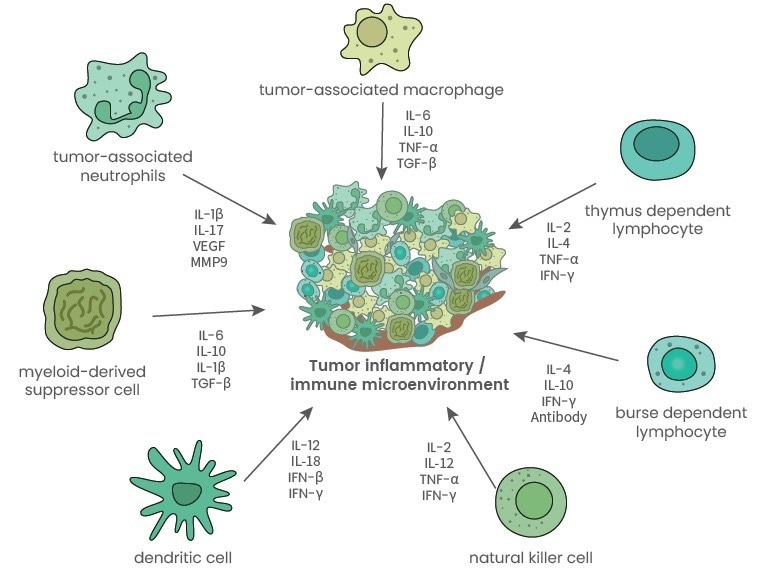
Figure 1. Major immune cells and related cytokines secreted in the tumor microenvironment1. Image Credit: Sino Biological Inc.
Depletion of MDSC populations and infiltration
VEGF – Vascular endothelial growth factor – is the key mediator of tumor angiogenesis and the primary target of anti-angiogenesis therapy for various malignant tumors.3 In the tumor microenvironment, VEGF secretion plays several roles in tumor progression.
This includes MDSC infiltration, tumor angiogenesis promotion, tumor progression acceleration, and immunosuppressive enhancement.4 Studies have revealed that VEGF-induced MDSCs possess greater immunosuppressive properties than other MDSCs.
Down-regulation of VEGF in tumor cells results in reduced MDSC infiltration, and targeting VEGF-induced MDSCs is an effective tumor therapy.5 Sunitinib and sorafenib, which are anti-VEGF or VEGF receptor (VEGFR) drugs, have been successfully used to block the VEGF pathway.
These drugs produce antiangiogenic effects and can effectively decrease the MDSC populations in cancer patients. Furthermore, chemotherapy treatments, including cisplatin, 5-fluorouracil (5FU), paclitaxel, and gemcitabine can deplete MDSCs and enhance antitumor immune activity.4
Thus, the company has developed high-purity and high-activity receptor proteins and VEGFs from varying species to help in this area of research.
The majority of said recombinant proteins were expressed in eukaryotic cells, which ensure the correct modification of proteins and higher activity. All proteins are produced at home and available with customized services.
Using various methods to provide consistent and reproducible results, these high-quality reagents have been validated.
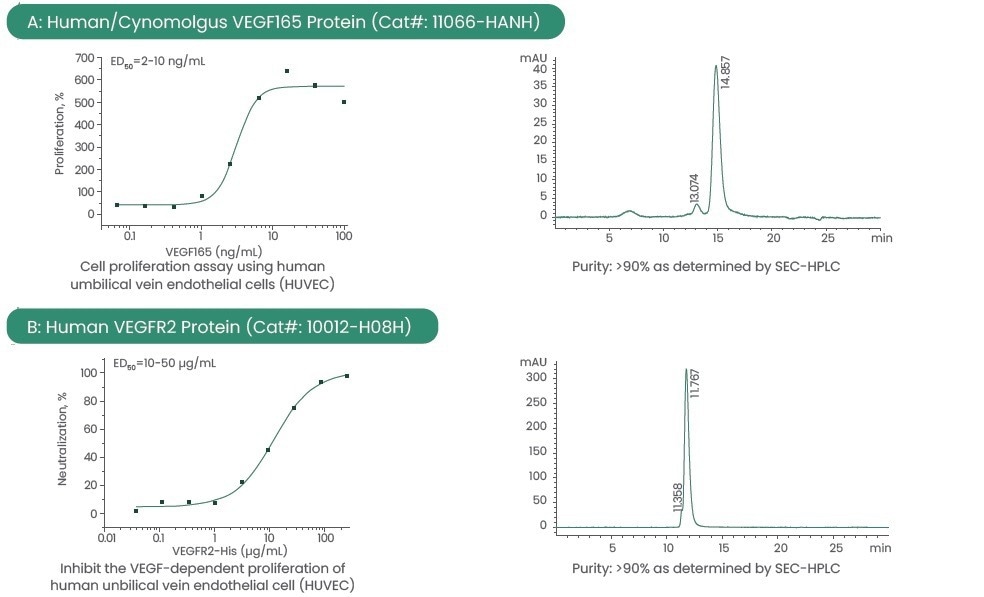
Figure 2. Examples of high-quality VEGF and VEGFR. Image Credit: Sino Biological Inc.
Inhibition of MDSC recruitment and transport
A large number of chemokines in the tumor microenvironment is a key factor in MDSCs migration to the tumor site. Therefore, targeting chemokines receptors on MDSCs is a potentially effective cancer therapy strategy.
Tyrosine kinase receptors, such as a colony stimulating factor 1 receptor (CSF-1R), can promote the migration of MDSCs to tumor sites, inhibiting antitumor immunity.
CSF-1R promotes both the myeloid cell migration to the tumor sites and myeloid cell differentiation to MDSCs and tumor-related macrophages after combining with ligand CSF-1.
Studies have found that CSF-1/CSF-1R signaling inhibition regulates the number and function of MDSCs in murine tumors, which can be reprogrammed into an antitumor phenotype. Additionally, CSF-1R inhibitor combined with CTLA-4 blockade therapy induces antitumor T-cell response and tumor regression in many tumor models.
Therefore, utilizing a CSF-1/CSF-1R approach, MDSCs are targeted to regulate the tumor microenvironment, and the CSF-1/CSF-1R signaling blockade is crucial to improve the checkpoint-based immunotherapy response.6
The nuclear factor-κ-gene binding (NF-κB) signaling pathway plays an important role in MDSC expansion during tumor initiation and progression. Amp-activated protein kinase (AMPK) is an important protein kinase that regulates energy metabolism and innate adaptive immunity by targeting major signal pathways.
AMPK activation reduces the MDSC expansion and activation by inhibiting multiple signal pathways, such as STATs and NF-κB pathways.2
Therefore, Sino Biological has developed high-activity and high-purity CSF ligand and receptor proteins and CTLA-4 proteins from various species to support this therapy research.
In addition, Sino Biological has also created a series of antibodies with high affinity and sensitivity for CSF growth factor and receptor, which can be used in flow cytometry (FCM), enzyme-linked immunoabsorbent assay (ELISA), and Western blotting (WB).
All proteins and antibodies are produced at home and are available with customized services. These high-quality reagents have been validated using various methods to ensure consistent and reproducible results.
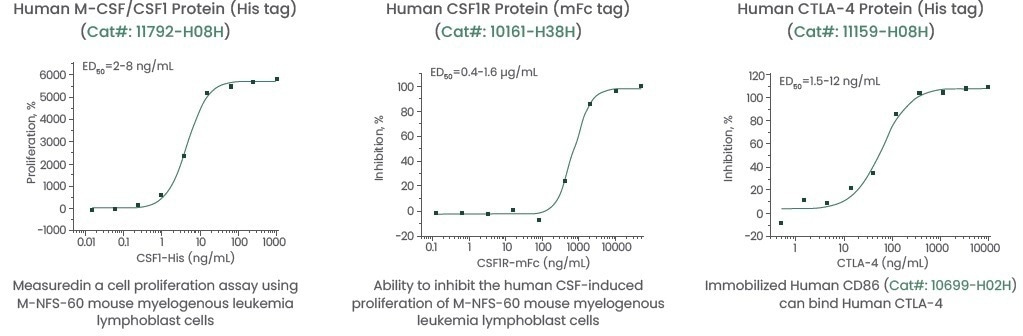
Figure 3. Featured products of high-quality CSF1, CSF1R, and CTLA-4. Image Credit: Sino Biological Inc.
Anticytokine therapy of MDSCs
Cytokines are low molecular proteins that mediate intercellular communication, which regulates cell activity, differentiation, immune cell activation, cell migration, and death.7
Immunosuppressive cytokines secreted by MDSCs are important factors in the tumor microenvironment which inhibit antitumor immune response and promote tumor progression.
Recent progress in anticytokine therapy in immunology has focused the attention of researchers, promoting interest in the application of this therapy in the research interest for its application in the field of oncology.
IL-1β promotes angiogenesis, tumor invasiveness, and carcinogenesis and affects immune function in various ways, including indirectly affecting immune function by inducing MDSC accumulation in the tumor microenvironment.
Anti-IL-1β monoclonal antibodies can reduce PMN–MDSCs in tumors and delay tumor growth.8,9 Additionally, recent studies have revealed that IL-6 and IL-10 from ascites lead to large amounts of M-MDSC accumulation in the peripheral blood and in patients with ascites of ovarian cancer.10
IL-6 and IL-8 play an important role in patients with melanoma. IL-6 potentially expands peripheral MDSCs, while IL-8 recruits these highly immunosuppressed cells into the tumor microenvironment.11
The growing evidence for the central role cytokines play has provided novel therapeutic directions for cancer patients. Targeting cytokines can improve antitumor efficacy by locally targeting MDSCs.
Sino Biological, therefore, has developed high-activity, high-purity, and low-endotoxin proteins from various species, including IL-1β and IL-6, to support this therapy research.
Moreover, the company has developed a comprehensive panel of antibodies targeting cytokines and receptors, which can be used in ELISA, WB, immunohistochemistry (IHC), and immunoprecipitation (IP).
All proteins and antibodies are produced at home and are available with customized services. These high-quality reagents have been validated using various methods to provide consistent and reproducible results.
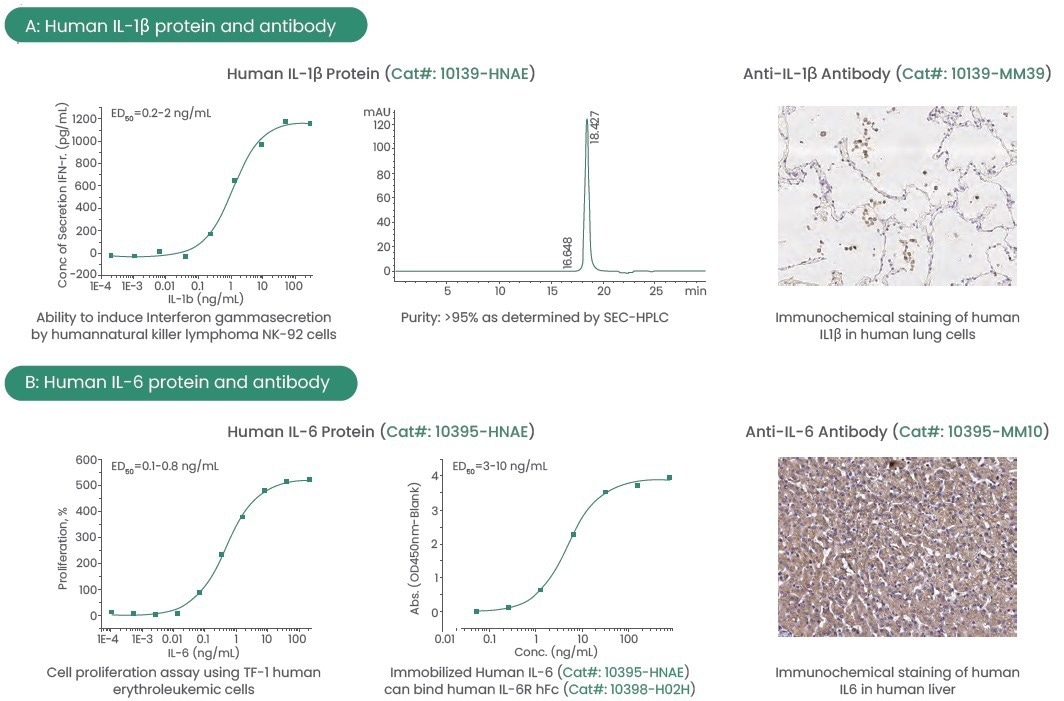
Figure 4. Examples of high-quality IL-1β and IL-6 protein and antibody. Image Credit: Sino Biological Inc.
Conclusion
Cancer, a major disease, remains a major threat to human health. This article from Sino Biological aimed to help offer new directions for the treatment of patients with cancer from the perspective of targeting MDSCs in the tumor microenvironment.
The immunotherapy strategies include depleting the populations and infiltration, inhibiting the recruitment and transport, and anticytokine therapy. These efforts will bring about huge opportunities for existing cancer treatments.
References and further reading
- Li, L et al. (2020) Effects of immune cells and cytokines on inflammation and immunosuppression in the tumor microenvironment Int Immunopharmacol. 88: 106939 [online] Available at: https://doi.org/10.1016/j.intimp.2020.106939
- Gao, X et al. (2020) Immunotherapy Targeting Myeloid-Derived Suppressor Cells (MDSCs) in Tumor Microenvironment Front. Immunol. 11: 585214 [online] Available at: https://doi.org/10.3389/fimmu.2020.585214
- Tamura, R et al. (2020) The role of vascular endothelial growth factor in the hypoxic and immunosuppressive tumor microenvironment: perspectives for therapeutic implications Medical Oncology 37: 2 [online] Available at: https://doi.org/10.1007/s12032-019-1329-2
- Law, A.M/K et al. (2020) Myeloid-Derived Suppressor Cells as a Therapeutic Target for Cancer Cells 27:9(3): 561 [online] Available at: https://www.mdpi.com/2073-4409/9/3/561
- Horikawa, N et al. (2017) Expression of Vascular Endothelial Growth Factor in Ovarian Cancer Inhibits Tumor Immunity through the Accumulation of Myeloid-Derived Suppressor Cells Clin Cancer Res. 23(2) pp. 587-99 [online] Available at: https://pubmed.ncbi.nlm.nih.gov/27401249/
- Holmgaard, R.B et al. (2016) Timing of CSF-1/CSF-1R signaling blockade is critical to improving responses to CTLA-4 based immunotherapy OncoImmunology 5(7) [online] Available at: https://doi.org/10.1080/2162402X.2016.1151595
- Landskron, G et al. (2014) Chronic inflammation and cytokines in the tumor microenvironment J Immunol Res. [online] Available at: https://pubmed.ncbi.nlm.nih.gov/24901008/
- Liu, Y et al. (2018) Targeting myeloid-derived suppressor cells for cancer immunotherapy Cancer Immunol Immunother. 67(8) pp. 1181-1995 [online] Available at: https://pubmed.ncbi.nlm.nih.gov/29855694/
- Elkabets, M et al. (2010) IL-1β regulates a novel myeloid-derived suppressor cell subset that impairs NK cell development and function European Journal of Immunology 40(12) pp. 3347-3357 [online] Available at: https://doi.org/10.1002/eji.201041037
- Wu, L et al. (2017) Ascites-derived IL-6 and IL-10 synergistically expand CD14+HLA-DR-/low myeloid-derived suppressor cells in ovarian cancer patients Oncotarget 8(44) pp. 76843-76856 [online] Available at: https://pubmed.ncbi.nlm.nih.gov/29100353/
- Tobin, R.P et al. (2019) IL-6 and IL-8 Are Linked With Myeloid-Derived Suppressor Cell Accumulation and Correlate With Poor Clinical Outcomes in Melanoma Patients Frontiers in Oncology 9 [online] Available at: https://www.frontiersin.org/articles/10.3389/fonc.2019.01223
About Sino Biological Inc.

Sino Biological is an international reagent supplier and service provider. The company specializes in recombinant protein production and antibody development. All of Sino Biological's products are independently developed and produced, including recombinant proteins, antibodies and cDNA clones. Sino Biological is the researchers' one-stop technical services shop for the advanced technology platforms they need to make advancements. In addition, Sino Biological offers pharmaceutical companies and biotechnology firms pre-clinical production technology services for hundreds of monoclonal antibody drug candidates.
Sino Biological's core business
Sino Biological is committed to providing high-quality recombinant protein and antibody reagents and to being a one-stop technical services shop for life science researchers around the world. All of our products are independently developed and produced. In addition, we offer pharmaceutical companies and biotechnology firms pre-clinical production technology services for hundreds of monoclonal antibody drug candidates. Our product quality control indicators meet rigorous requirements for clinical use samples. It takes only a few weeks for us to produce 1 to 30 grams of purified monoclonal antibody from gene sequencing.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.