The burden of coronavirus disease 2019 (COVID-19) is expected to diminish as the coverage of efficient messenger ribonucleic acid (mRNA) vaccines against severe acute respiratory coronavirus 2 (SARS-CoV-2) increases.
Numerous studies demonstrate a high degree of inter-individual variability in vaccine-induced antibody levels. Although several factors may contribute to the heterogeneity, it is critical to delineate modifiable factors affecting post-vaccine immunogenicity.
The epidemiological research on the relationship between smoking and alcohol consumption and post-vaccine antibody titers has been conflicting. There is no data associating post-vaccine antibody response to the use of heat-not-burn (HNB) tobacco, which has gained popularity in the global market.
 Study: Use of heat-not-burn tobacco products, moderate alcohol drinking, and anti-SARS-CoV-2 IgG antibody titers after BNT162b2 vaccination among Japanese healthcare workers. Image Credit: CGN089/Shutterstock
Study: Use of heat-not-burn tobacco products, moderate alcohol drinking, and anti-SARS-CoV-2 IgG antibody titers after BNT162b2 vaccination among Japanese healthcare workers. Image Credit: CGN089/Shutterstock

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
It is known that smoking and excessive alcohol impair the activation of innate and acquired immunity and consequently reduce vaccine-induced antibody formation. As with regular cigarettes, HNB tobacco products include nicotine, which can negatively affect the immune system. Relating to alcohol consumption, prior studies have not assessed the dose-response relationship with special emphasis on the effect of moderate drinking. The fact is particularly concerning for East Asians, specifically the Japanese – who have a high rate of mutations in an alcohol-metabolizing enzyme.
A preprint version of the study is available on the medRxiv* server, while the article undergoes peer review.
The study
The aim of this study was to determine the relationship between smoking (including the usage of HNB tobacco) and alcohol intake with immunoglobulin (Ig)G antibody titers against SARS-CoV-2 spike protein in personnel of Japanese national medical research facilities who received two doses of BNT162b2 vaccination.
In 2020, a joint study involving many centers and repeated serological surveys was initiated among employees of Japan's six National Centers for Advanced Medical and Research (6NC). The trial enrolled 5,718 workers, of whom 5,013 reported receiving two doses of the COVID-19 vaccination (BNT162b2, Pfizer-BioNTech). Participants were divided into five categories – never smokers; former smokers (quit smoking a cigarette and did not use HNB tobacco products); current smokers who used only HNB tobacco products; current smokers who smoke only conventional cigarettes; and current smokers who smoke both conventional cigarettes and HNB tobacco products (dual users). Additionally, the average daily alcohol consumption was assessed and subjects were divided into five groups – non-drinkers, occasional drinkers (one to three days per month), and weekly drinkers ingesting 1 go/day, 1–1.9 go/day, or 2 go/day (1 go of Japanese sake contains approximately 23 grams of ethanol).
The findings
The median age of the participants was 41 years (interquartile range [IQR]: 30–50 years); 72% were females with a 0.5% history of COVID-19.
Overall, 34% of the workforce were nurses; the others were allied health professionals (18%), administrative workers (15%), physicians (14%), and researchers (13%). Almost half of the 212 current smokers utilized HNB tobacco products. Meanwhile, 39% of the participants consumed alcohol at least once a week. Between the second vaccination and blood sample collection, the median delay was 64 days. Anti-SARS-CoV-2 spike IgG antibody titers were 144 SU/mL on an average, with 99.5% of subjects being seropositive.
Current smokers showed lower antibody titers compared to never-smokers. Exclusive users of HNB tobacco products and dual users had significantly lower estimated geometric means (GMTs); however, the differences were not statistically significant. The reduction achieved statistical significance in a post-hoc analysis combining the two categories of HNB tobacco smokers.
Daily cigarette smokers who consumed 11 or more cigarettes per day had a higher reduction in IgG titers than those who consumed less than 11 cigarettes per day.
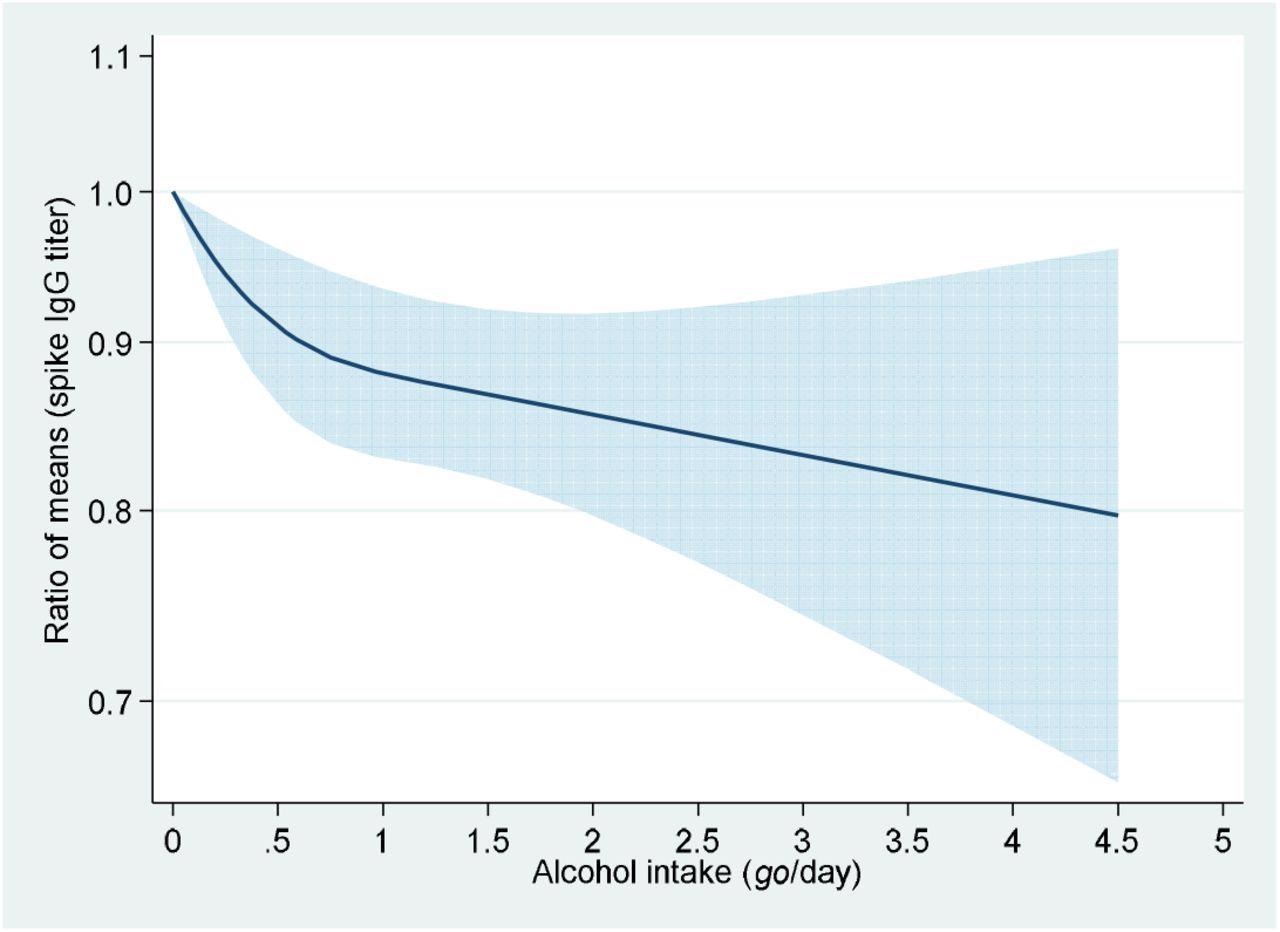 Dose-response association between alcohol consumption and anti-SARS-CoV-2 spike IgG titers The y-axis is the ratio of means with the shaded area representing 95% confidence intervals (linear trend, P<0.01; non-linear trend, P=0.02). The reference value is 0 go/day (non-drinkers). The model was adjusted for age (years, continuous), sex, job category (doctor, nurse, allied healthcare professional, administrative staff, researcher, and other), body mass index (BMI) (kg/m2, continuous), an interaction term of sex and BMI, the interval between the second vaccination and blood sampling (days, continuous), the squared term of the interval, the history of COVID-19, comorbid conditions (diabetes, hypertension, and cancer), leisure-time physical activity (0, 1 to 59, or ≥60 min/wk), sleeping duration (<6, 6 to 6.9, or ≥7 h), and smoking status. One go of Japanese sake contains approximately 23 g of ethanol.
Dose-response association between alcohol consumption and anti-SARS-CoV-2 spike IgG titers The y-axis is the ratio of means with the shaded area representing 95% confidence intervals (linear trend, P<0.01; non-linear trend, P=0.02). The reference value is 0 go/day (non-drinkers). The model was adjusted for age (years, continuous), sex, job category (doctor, nurse, allied healthcare professional, administrative staff, researcher, and other), body mass index (BMI) (kg/m2, continuous), an interaction term of sex and BMI, the interval between the second vaccination and blood sampling (days, continuous), the squared term of the interval, the history of COVID-19, comorbid conditions (diabetes, hypertension, and cancer), leisure-time physical activity (0, 1 to 59, or ≥60 min/wk), sleeping duration (<6, 6 to 6.9, or ≥7 h), and smoking status. One go of Japanese sake contains approximately 23 g of ethanol.
On the other hand, weekly alcoholic drinkers exhibited considerably lower antibody titers than non-alcoholic beverage users. Moreover, the spike IgG antibody titers declined consistently as alcohol consumption increased.
Furthermore, antibody titers were substantially lower in moderate drinkers. Spline analysis revealed a clear dose-response association with a significant decrease in the ratio of means occurring until approximately 1 go/day of alcohol consumption – followed by a progressive decline with increasing alcohol consumption. However, the findings may not hold true in people with diverse genetic backgrounds.
In comparison to never-smokers, exclusive cigarettes smokers had considerably lower anti-SARS-CoV-2 spike IgG antibody titers. In addition, antibody titers demonstrated a clear decreasing trend with increasing alcohol use, with significant declines found even at low levels of alcohol.
The results supported a deleterious effect of HNB tobacco product usage on immunogenicity to the COVID-19 immunization.
Conclusion
In conclusion, smokers of cigarettes and, to a lesser extent, users of HNB tobacco products had lower IgG antibody titers against the SARS-CoV-2 spike protein, post-vaccination. While antibody titers declined consistently as alcohol consumption increased with a significant low found even with moderate alcohol intake.
Therefore, cigarette smoking, HNB tobacco products, and excessive alcohol consumption, and even alcohol in-moderation can impair the immunological response to the COVID-19 vaccination.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Yamamoto, S., Tanaka, A., Ohmagari, N., et al. (2021). Use of heat-not-burn tobacco products, moderate alcohol drinking, and anti-SARS-CoV-2 IgG antibody titers after BNT162b2 vaccination among Japanese healthcare workers. medRxiv preprint. doi: https://doi.org/10.1101/2021.11.29.21267032 https://www.medrxiv.org/content/10.1101/2021.11.29.21267032v1
- Peer reviewed and published scientific report.
Yamamoto, Shohei, Akihito Tanaka, Norio Ohmagari, Koushi Yamaguchi, Kazue Ishitsuka, Naho Morisaki, Masayo Kojima, et al. 2022. “Use of Heated Tobacco Products, Moderate Alcohol Drinking, and Anti-SARS-CoV-2 IgG Antibody Titers after BNT162b2 Vaccination among Japanese Healthcare Workers.” Preventive Medicine 161 (August): 107123. https://doi.org/10.1016/j.ypmed.2022.107123. https://www.sciencedirect.com/science/article/pii/S0091743522001724.