Current worldwide circulation and burden of Dengue and Zika
Arboviruses, in particular Dengue virus (DENV) and Zika virus (ZIKV), pose a significant and growing global health risk. Dengue fever, transmitted primarily by infected Aedes mosquitoes, is currently endemic in more than 100 countries. The virus puts about half of the world's population at risk, resulting in an estimated 400 million infections, 100 million symptomatic cases, and up to 40,000 deaths per year.1 The Zika virus, which caused a global public health disaster during the 2015-2016 outbreak, continues to spread widely (Figure 1).1
While generally asymptomatic, ZIKV poses a serious risk during pregnancy and can result in Congenital Zika Syndrome (CZS), which includes severe microcephaly, as well as neurological problems such as Guillain-Barré syndrome in adults.2
The geographic footprints of both viruses are spreading rapidly with urbanization, global travel, and climate change, highlighting the crucial and urgent need for safe, globally deployable vaccines.
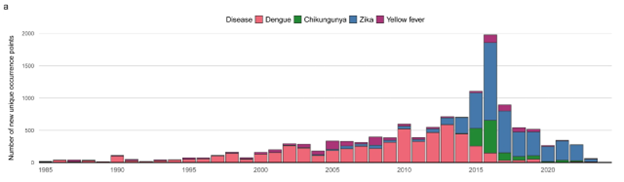
Figure 1. The global number of new unique occurrence points added each year for arboviruses. Image Credit: Lim, A., et al. (2025)
Clinical research status of arbovirus vaccines and the ADE challenge
The clinical development of vaccines against DENV and ZIKV has historically been hampered by a single, significant immunological barrier: antibody-dependent enhancement (ADE).
Dengue fever is caused by four separate, co-circulating serotypes (DENV1-4), and because DENV and ZIKV share significant structural resemblance, prior infections or poorly designed vaccines can produce cross-reactive, sub-neutralizing antibodies. These antibodies frequently target the viral Envelope (E) monomer's highly conserved, immunodominant "fusion loop".
As opposed to neutralizing the virus, these antibodies promote viral entrance into host immune cells, exacerbating its severity upon recurrent infection. As such, recent clinical research has evolved away from producing broad antibody responses and toward more targeted, structure-guided techniques.
To prevent ADE, next-generation subunit and mRNA vaccines rely quite heavily on stable Envelope (E) protein dimers.3 Researchers can effectively mask the harmful, ADE-prone fusion loop by locking the E protein in its native, dimeric state while exposing potent, dimer-dependent epitopes that induce robust, safe neutralizing immunity.
Although early-generation live-attenuated platforms now dominate the late-stage pipeline, the continued danger of ADE has fueled a varied array of advanced vaccination candidates using recombinant E dimers in active clinical trials.
Table 1. Examples of Recombinant E Protein Dimer-Based Arbovirus Vaccine. Source: Sino Biological Inc.
| Developer |
Vaccine Candidate |
Target Virus |
Platform Technology |
Latest Development Status |
Merck / Hawaii
Biotech |
V180 (DEN-80E) |
DENV |
Recombinant Antigen Subunit |
Phase 1 Completed |
| VBI Vaccines |
VBI-2501 |
ZIKV |
Enveloped VLP |
Phase 1/2a Completed |
Univ. of
Oxford / UNC |
Covalently locked
dimer (cvD) vaccines |
DENV/ZIKV |
Recombinant Antigen Subunit |
Preclinical / Near-Clinical: Complete |
Research applications
Arbovirus envelope (E) protein dimers have proven to be extremely efficient tools in vaccine research. They serve as vaccine antigens, scaffolds for safer vaccinations, antigen displays in particle systems, and antibody assay reagents.
Recent research shows that persistent E dimers increase antigen quality, focus immune responses to potent neutralizing epitopes, and limit targeting of ADE-prone regions, making them extremely useful for immune-response profiling in human samples.
Metz et al. tested whether a stable Zika virus (ZIKV) E protein dimer outperformed an E monomer as a subunit vaccine antigen. In mouse immunization tests, the E dimer acted as both the immunogen and the binding target. They discovered that only the native-like E dimer was highly recognized by powerful neutralizing antibodies and successfully generated protective responses in mice, making it the superior subunit antigen (Figure 2).4
Slon-Campos et al. used a covalently stabilized ZIKV E dimer to create a safer subunit vaccine. Their goal was to keep protective dimer-dependent epitopes while masking the fusion loop, which is associated with dengue (DENV) cross-reactivity and ADE. The modified E dimer, which served as both an immunogen and a design scaffold, elicited specific antibodies that protected against ZIKV during pregnancy while preventing harmful DENV cross-reactivity, demonstrating its dual benefits for protection and safety (Figure 3).5
In another application, Phan et al. created a tetravalent dengue subunit vaccine using stabilized DENV1-4 soluble E dimers on CPQ liposomes. These stable dimers, which act as multivalent immunogens, elicit higher neutralizing reactions than wild-type soluble E proteins, and liposomes are even more immunogenic. Importantly, the tetravalent mixture produced type-specific neutralizing antibodies with minimal immunological interference, suggesting that E dimers can effectively facilitate advanced multivalent vaccine formulation (Figure 4).6
Lay et al. demonstrated that E dimers can be used as precise analytical tools in addition to being immunogens. To measure specific antibody groups in human plasma, they developed a multiplex test and antibody isolation technique that included recombinant DENV2 EDIII, soluble E, and sE-dimer proteins (Figure 5). The E dimer served as a reference and isolation reagent, successfully separating and enriching various antibody subsets. This showed that E dimers are not only effective vaccine antigens but also critical tools for immunological profiling and distinguishing between protective and possibly detrimental antibody responses.7
Sino Biological provides a wide range of high-quality recombinant Arbovirus envelope (E) proteins, including extremely stable E dimers, to help with this essential move toward structure-guided antigen design. Engineered to suit the stringent requirements of modern vaccination research, these E dimers accurately preserve the native-like dimeric structure, as shown by SEC-MALS. Whether you are creating safer subunit vaccines, multivalent nanoparticle displays, or multiplex immune-profiling assays to isolate specific antibody subsets, Sino Biological's recombinant DENV and ZIKV E dimers provide the dependable, highly pure antigens you need to succeed.
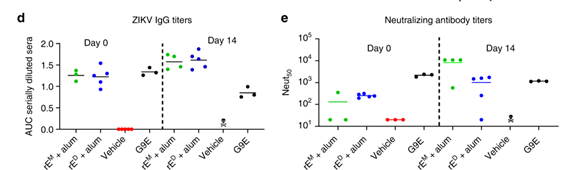
Figure 2. Only the recombinant ZIKV E dimer, not the monomer, induced protective antibodies in a ZIKV challenge model. Image Credit: DOI: 10.1038/s41467-019-12677-6
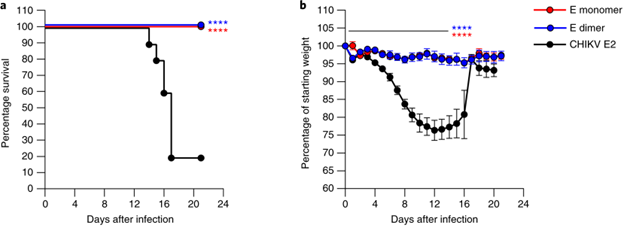
Figure 3. The ZIKV E-dimer vaccine protected pregnant mice and reduced viral burden in maternal and fetal tissues. Image Credit: DOI: 10.1038/s41590-019-0477-z
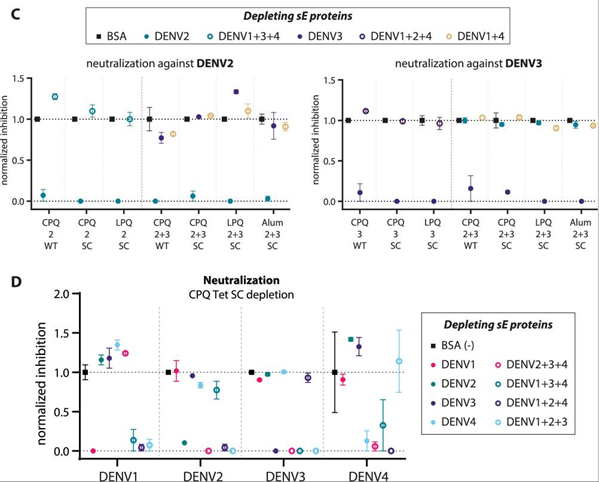
Figure 4. Tetravalent dengue E dimers produced type-specific neutralizing responses, and depletion studies showed little immune interference between serotypes. Image Credit: DOI: 10.1038/s41541-025-01179-w
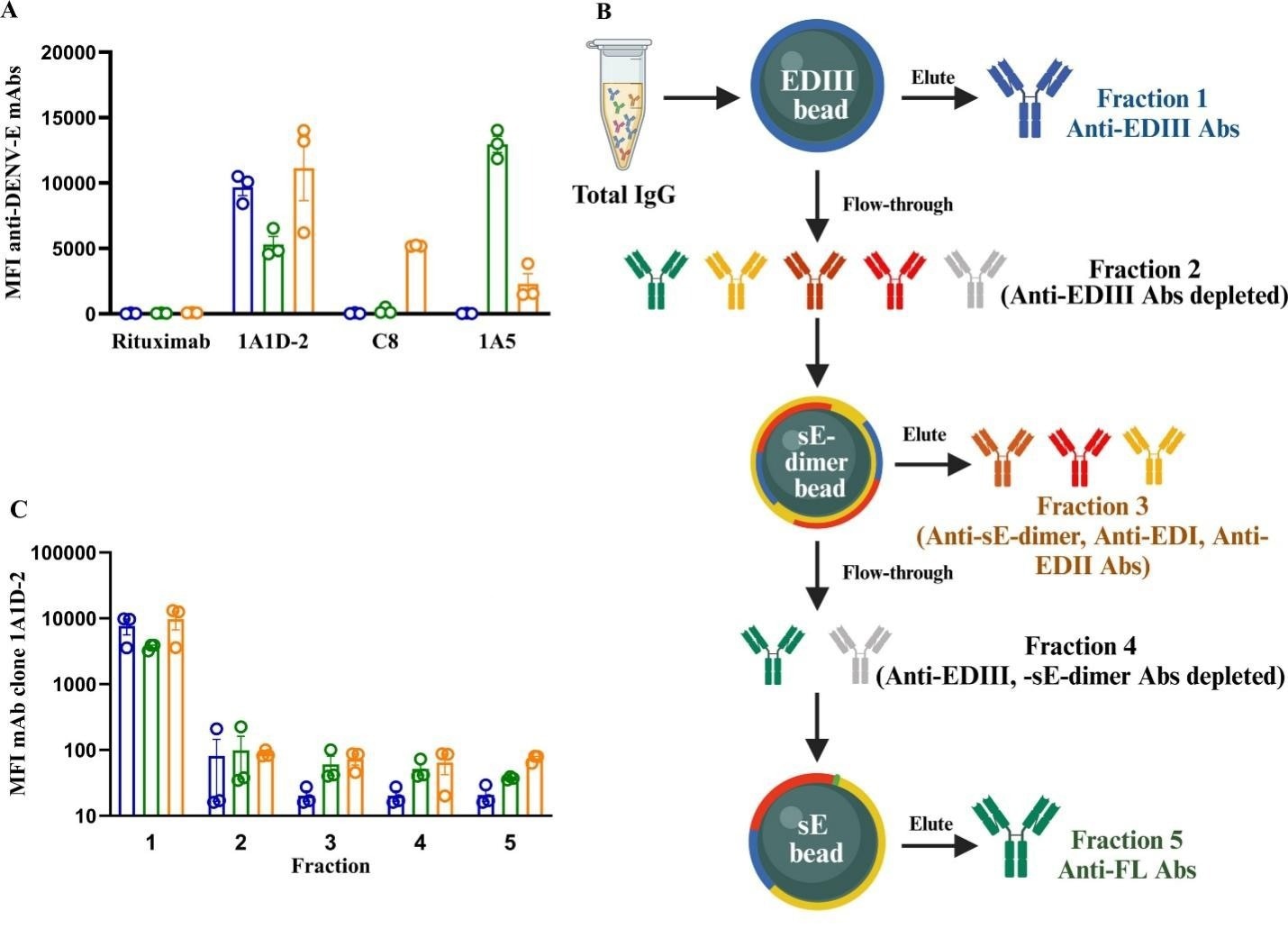
Figure 5. Recombinant DENV E proteins, including sE-dimer, were used to isolate and measure epitope-specific antibody subsets from human samples. Image Credit: DOI: 10.1128/msphere.00961-24
References
- Lim, A., et al. (2025). The overlapping global distribution of dengue, chikungunya, Zika and yellow fever. Nature Communications, (online) 16(1). DOI: 10.1038/s41467-025-58609-5. https://www.nature.com/articles/s41467-025-58609-5.
- Rabe, I.B., et al. (2025). A Review of the Recent Epidemiology of Zika Virus Infection. The American Journal of Tropical Medicine and Hygiene, (online) 112(5). DOI: 10.4269/ajtmh.24-0420. https://www.ajtmh.org/view/journals/tpmd/112/5/article-p1026.xml.
- Unali, G. and Douam, F. (2025). Orthoflavivirus Vaccine Platforms: Current Strategies and Challenges. Vaccines, 13(10), p.1015. DOI: 10.3390/vaccines13101015. https://www.mdpi.com/2076-393X/13/10/1015.
- Metz, S.W., et al. (2019). Oligomeric state of the ZIKV E protein defines protective immune responses. Nature communications, 10(1). DOI: 10.1038/s41467-019-12677-6. https://www.nature.com/articles/s41467-019-12677-6.
- Slon-Campos, J.L., et al. (2019). A protective Zika virus E-dimer-based subunit vaccine engineered to abrogate antibody-dependent enhancement of dengue infection. Nature Immunology, 20(10), pp.1291–1298. DOI: 10.1038/s41590-019-0477-z. https://www.nature.com/articles/s41590-019-0477-z.
- Phan, T.T.N., et al. (2025). Multivalent administration of dengue E dimers on liposomes elicits type-specific neutralizing responses without immune interference. npj Vaccines, (online) 10(1). DOI: 10.1038/s41541-025-01179-w. https://www.nature.com/articles/s41541-025-01179-w.
- Lay, S., et al. (2025). Toward a deeper understanding of dengue: novel method for quantification and isolation of envelope protein epitope-specific antibodies. mSphere. DOI: 10.1128/msphere.00961-24. https://journals.asm.org/doi/full/10.1128/msphere.00961-24.
About Sino Biological Inc.
Founded in 2007, Sino Biological is a global biotechnology company specializing in high-quality recombinant proteins, antibodies, and CRO services. Serving researchers in over 90 countries, Sino Biological supports basic research, drug discovery, vaccine development, and diagnostics through its comprehensive product portfolio, proprietary quality systems, and innovative research platforms.
Sino Biological's core business
Sino Biological is a leading global biotechnology supplier dedicated to high-quality research reagents and comprehensive CRO services. Its extensive product portfolio covers over 9,800 recombinant proteins, 15,000 antibodies, and 50,000 genes, all produced in-house. As a trusted CRO partner, Sino Biological specializes in customized recombinant protein and antibody production. It stands out with its robust high-throughput antibody production system including HEK293/CHO and cell‑free platforms. This system enables rapid turnaround (as fast as 5 days), high-throughput production (>10,000 Abs/month) and cost-effective solutions for global academic and pharmaceutical clients.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.