Current worldwide circulation and burden of RSV
Respiratory syncytial virus (RSV) is a highly contagious seasonal pathogen that is the major cause of acute lower respiratory infections (ALRIs) worldwide. RSV is responsible for an enormous health and economic burden worldwide, with an estimated 33 million new ALRI cases and over 3 million hospitalizations in children under the age of five each year.1
It causes around 118,000 pediatric deaths each year, the majority of which (> 99 %) occur in low- and middle-income countries (LMICs).2
Beyond the pediatric population, RSV is now commonly accepted as a leading cause of serious respiratory disease and mortality in older adults and immunocompromised people.
At the epidemiological level, global circulation is predominantly determined by distinct viral lineages: for the RSVA subtype, the A.D.3 lineage has progressively increased in prevalence since 2022, whereas the B.D.E.1 lineage has dominated RSVB circulation since 2023 (Figure 1).
This extensive transmission, shifting strain dynamics, and high disease burden highlight the critical need for effective preventive interventions, leading to significant global investment in RSV vaccine and monoclonal antibody research.
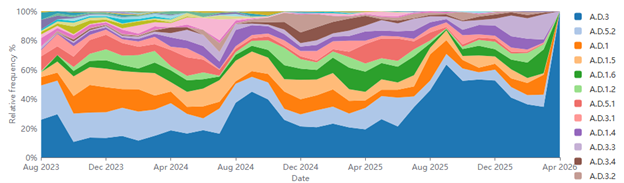
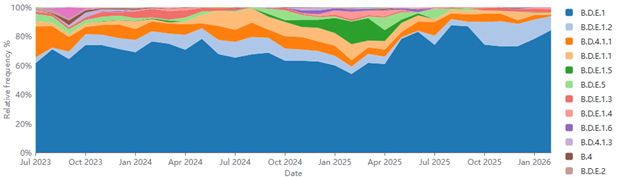
Figure 1. Lineage Progression of RSV Subtype A and B since 2023. Image Credit: Sino Biological Inc.
Clinical research status of RSV vaccines
The therapeutic landscape for RSV prevention has evolved rapidly in recent years, largely driven by advances in the structural biology of the RSV fusion (F) glycoprotein - specifically, the stabilization of the prefusion (preF) conformation.
The global RSV pipeline now comprises dozens of active vaccine candidates and monoclonal antibodies (mAbs) in clinical studies.
Vaccine development is heavily segregated into three key target populations: pediatrics (using mAbs or live-attenuated vaccines), mothers (to produce protective antibodies in babies), and older adults (using adjuvanted subunit, mRNA, and vector-based platforms).
With recent regulatory clearance of numerous first-generation RSV vaccines, clinical research has shifted to improving immunogenicity, assessing the durability of revaccination, and developing combination respiratory vaccines.
Examples of current vaccines under clinical trials
The clinical pipeline for next-generation RSV vaccines is extremely active, demonstrating a number of sophisticated platforms and combination strategies:
- Clover Biopharmaceuticals (SCB-1019 & Combination Vaccines): Clover Biopharmaceuticals is developing a recombinant RSV preF subunit vaccine (SCB-1019) as part of its combination vaccine program. Recent Phase 1 findings showed that it could be a highly effective revaccination booster for older persons who have already received other licensed RSV vaccines.3 In addition, Clover launched a Phase 2 trial to evaluate multivalent respiratory combination candidates SCB-1022 (RSV + hMPV) and SCB-1033 (RSV + hMPV + PIV3) using their protein-based trimer platform.4
- Moderna (mRNA-1365): Moderna is conducting Phase 1 clinical studies for mRNA-1365, a combination vaccination that targets both RSV and hMPV in a single dose, following the launch of its RSV mRNA vaccine (mRESVIA).5
- The PIPELINE-RSV Trial: The PIPELINE-RSV Trial is a large multinational adaptive platform assessing a combined preventative approach. The trial randomly assigns pregnant women and their infants to receive a maternal RSV vaccine, an infant monoclonal antibody, or both at the same time to see if dual administration improves infant protection.6
Real-world effectiveness of RSV vaccines in vulnerable populations
While phase 3 clinical trials have consistently shown moderate to high efficacy for RSV preF vaccines, determining their effects in real-world settings is essential.
Clinical studies frequently underrepresent people at the highest risk for severe RSV disease, such as those over 80 years old, those with substantial cardiac comorbidities, and immunocompromised patients. Recent observational research is closing the gap.
Symes and colleagues conducted a multicenter, test-negative case-control study at 14 hospitals in England to evaluate the bivalent RSV preF vaccination. The study focused on people admitted with acute respiratory infection (ARI) and found highly positive real-world protection rates:7
- Provides 78.8 % protection against hospital admission for exacerbations of chronic lung disease, heart disease, or frailty
- 86.7 % effective against severe RSV disease needing oxygen, ICU admission, or mechanical breathing
- Those with immunosuppression have an estimated vaccination efficacy of 72.8 %
- 82.3 % efficacy in preventing hospitalization for RSV-associated ARI
Research applications
Recombinant respiratory syncytial virus (RSV) fusion (F) proteins, particularly prefusion-stabilized pre-F antigens, are extensively applied in RSV vaccine development. In addition, these antigens serve as assay reagents for antigen detection and immune response characterization. Recent research indicates that recombinant pre-F proteins aid in RSV vaccine development, ranging from clinical vaccinations to nanoparticle and mRNA vaccine investigations.
In a phase 3 experiment, Papi et al. found that an AS01E-adjuvanted subunit vaccination containing recombinant prefusion RSV F protein (RSVPreF3 OA) effectively protected older individuals from RSV-related lower respiratory tract disease and acute infection (Figure 2). This indicates that recombinant pre-F acts as a highly effective clinical vaccination antigen.8
Marcandalli et al. used recombinant RSV F trimers (DS-Cav1) as the framework for a self-assembling nanoparticle vaccination (Figure 3). Multiple copies of recombinant preF on nanoparticles resulted in stronger neutralizing antibody reactions than soluble trimers alone, emphasizing its importance in structure-guided vaccine design.9
Ma et al. used recombinant RSV proteins for testing and immune response analysis when developing an AAV5-based RSV vaccine. They used RSV-F antibodies (11049-R302, Sino Biological) for Western blot expression confirmation, as well as recombinant RSV-F (11049-V08B, Sino Biological) and RSV-G (40041-V08H, Sino Biological) proteins as ELISA coated antigens to track serum antibody responses after vaccination (Figure 4).10
Sun et al. (2025) created a Pre-F-EABR eVLP mRNA vaccination using the pre-F construct as an immunogen scaffold (Figure 5). They used Sino Biological recombinant RSV Pre-F (11049-VNAS, Sino Biological) and Post-F (11049-V08H5, Sino Biological) as ELISA coated antigens to successfully distinguish and validate robust, pre-F-specific protective antibodies.11
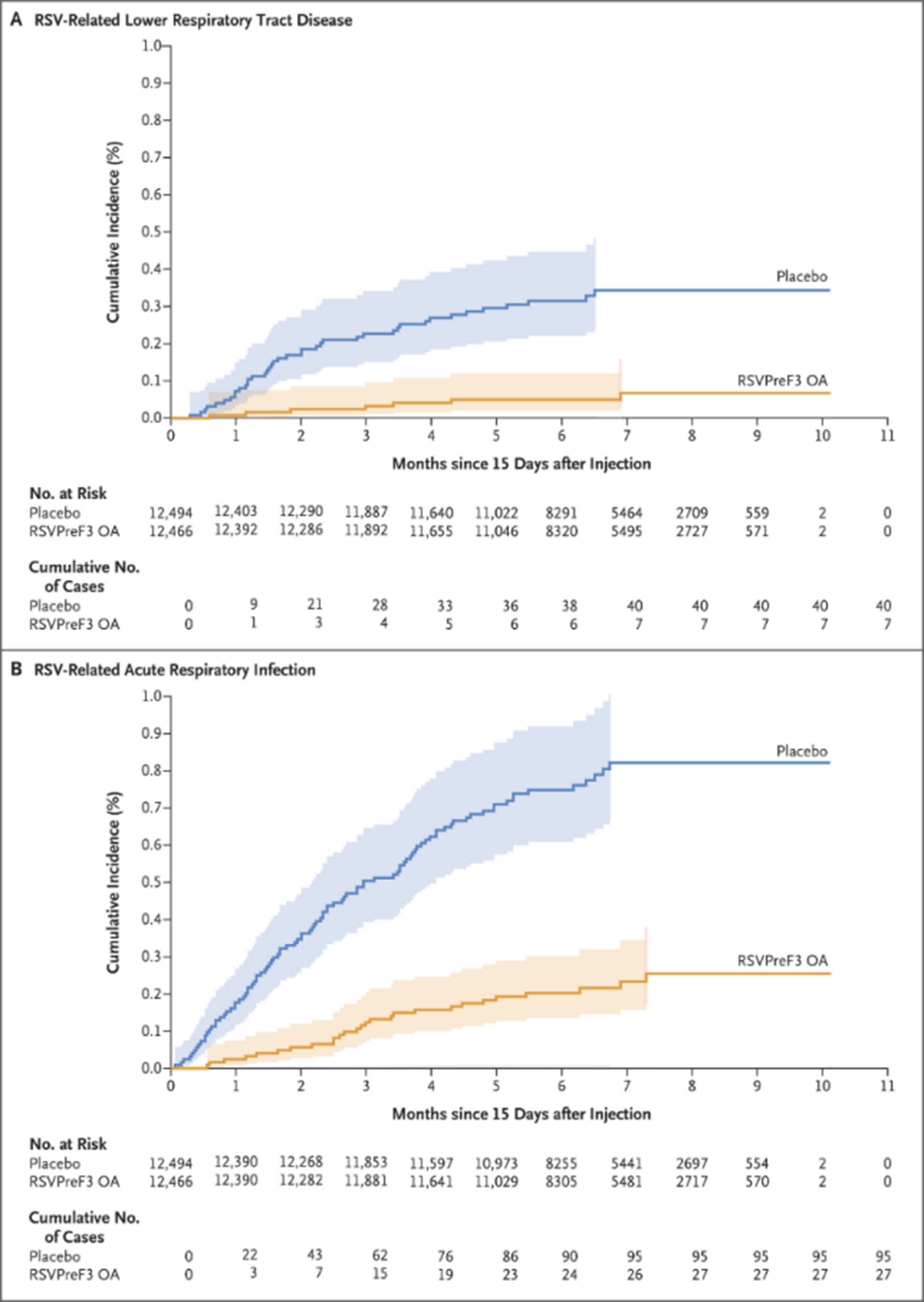
Figure 2. Cumulative incidence of RSV-related lower respiratory tract disease and RSV-related acute respiratory infection in older adults receiving the RSVPreF3 OA vaccine or placebo. Image Credit: DOI: 10.1016/j.cell.2019.01.046
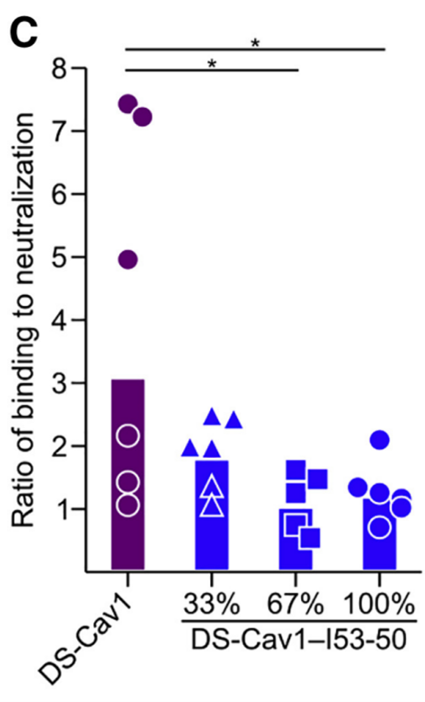
Figure 3. Structure-based design of a self-assembling nanoparticle displaying 20 copies of prefusion RSV F and its stronger induction of neutralizing antibody responses. Image Credit: DOI: 10.1016/j.cell.2019.01.046
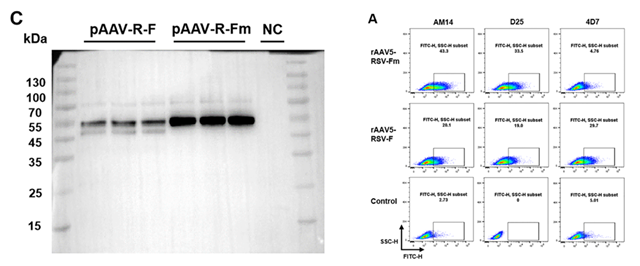
Figure 4. Sino Biological RSV reagents supported antigen expression confirmation and conformational characterization of RSV F in an AAV5-based vaccine study. Image Credit: DOI: 10.3389/fimmu.2024.1451433
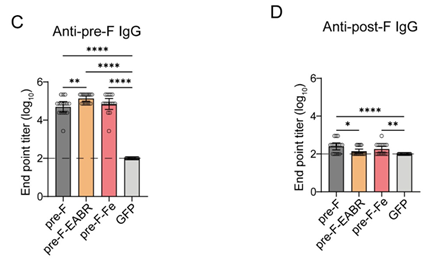
Figure 5. Sino Biological Recombinant Pre-F and Post-F proteins were used as ELISA coating antigens to distinguish preF-specific and postF-specific antibody responses elicited by RSV mRNA vaccine candidates. Image Credit: DOI: 10.1128/jvi.01209-25
References
- Li, X., et al. (2020). Health and economic burden of respiratory syncytial virus (RSV) disease and the cost-effectiveness of potential interventions against RSV among children under 5 years in 72 Gavi-eligible countries. BMC Medicine, 18(1). DOI: 10.1186/s12916-020-01537-6. https://link.springer.com/article/10.1186/s12916-020-01537-6.
- Mazur, N.I., et al. (2022). Respiratory syncytial virus prevention within reach: the vaccine and monoclonal antibody landscape. The Lancet Infectious Diseases. DOI: 10.1016/s1473-3099(22)00291-2. https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(22)00291-2/fulltext.
- Cloverbiopharma. (2026). Clover Biopharmaceuticals | Home. (online) Available at: https://www.cloverbiopharma.com/media/191.
- Cloverbiopharma. (2026). Clover Biopharmaceuticals | Home. (online) Available at: https://www.cloverbiopharma.com/media/189.
- Moderna. mRNA medicines we are currently developing. (online) Moderna. Available at: https://www.modernatx.com/en-US/research/product-pipeline.
- Clinicaltrials. (2026). Available at: https://clinicaltrials.gov/study/NCT07041190.
- Symes, R., et al. (2025). Vaccine effectiveness of a bivalent respiratory syncytial virus (RSV) pre-F vaccine against RSV-associated hospital admission among adults aged 75–79 years in England: a multicentre, test-negative, case–control study. The Lancet Infectious Diseases. DOI: 10.1016/s1473-3099(25)00546-8. https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(25)00546-8/fulltext.
- Papi, A., et al. (2023). Respiratory Syncytial Virus Prefusion F Protein Vaccine in Older Adults. New England Journal of Medicine, 388(7), pp.595–608. DOI: 10.1056/nejmoa2209604. https://www.nejm.org/doi/full/10.1056/NEJMoa2209604.
- Marcandalli, J., et al. (2019). Induction of Potent Neutralizing Antibody Responses by a Designed Protein Nanoparticle Vaccine for Respiratory Syncytial Virus. Cell, (online) 176(6), pp.1420-1431.e17. DOI: 10.1016/j.cell.2019.01.046. https://www.cell.com/cell/fulltext/S0092-8674(19)30109-6?sf214631395=1.
- Ma, G., et al. (2024). Induction of neutralizing antibody responses by AAV5-based vaccine for respiratory syncytial virus in mice. Frontiers in Immunology, 15. DOI: 10.3389/fimmu.2024.1451433. https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2024.1451433/full.
- Sun, L., et al. (2025). Developing an eVLP mRNA vaccine for respiratory syncytial virus with enhanced pre-fusion targeting humoral responses. Journal of Virology, 99(10). DOI: 10.1128/jvi.01209-25. https://journals.asm.org/doi/full/10.1128/jvi.01209-25.
About Sino Biological Inc.
Founded in 2007, Sino Biological is a global biotechnology company specializing in high-quality recombinant proteins, antibodies, and CRO services. Serving researchers in over 90 countries, Sino Biological supports basic research, drug discovery, vaccine development, and diagnostics through its comprehensive product portfolio, proprietary quality systems, and innovative research platforms.
Sino Biological's core business
Sino Biological is a leading global biotechnology supplier dedicated to high-quality research reagents and comprehensive CRO services. Its extensive product portfolio covers over 9,800 recombinant proteins, 15,000 antibodies, and 50,000 genes, all produced in-house. As a trusted CRO partner, Sino Biological specializes in customized recombinant protein and antibody production. It stands out with its robust high-throughput antibody production system including HEK293/CHO and cell‑free platforms. This system enables rapid turnaround (as fast as 5 days), high-throughput production (>10,000 Abs/month) and cost-effective solutions for global academic and pharmaceutical clients.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.