As cell therapy moves closer to clinical application, the landscape of regenerative medicine and oncology is being reshaped by iPSC-derived cellular products, in vivo reprogramming techniques, and scalable reagent platforms. In this evolution, essential tools like endotoxin-free proteins, transmembrane proteins, and site-specific fluorescent labeled proteins, are becoming critical enablers of reproducibility, mechanistic insight, and clinical-scale success.
iPSC-derived products: Guiding cell fate with defined biomolecules
Induced pluripotent stem cells (iPSCs) are derived from adult cells, such as skin or blood cells, which have been “reprogrammed” back to a stem-cell-like state. This transformation grants them the potential to differentiate into virtually any cell type in the body.
A typical process involves expanding patient-derived iPSCs in culture and then guiding them through specific differentiation paths to generate desired cell types, such as heart cells, neurons, pancreatic beta cells, or immune cells. These differentiated cells can then be used directly as therapies or assembled into complex tissue-like structures known as organoids for disease modeling, drug discovery, or eventual transplantation.
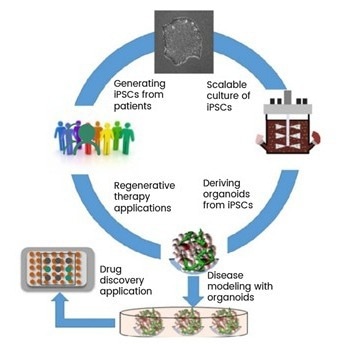
Figure 1. Workflow of patient-derived iPSCs. Image Credit: https://doi.org/10.1016/j.copbio.2019.05.011
The success of each stage depends on precise molecular cues. Recombinant growth factors and cytokines provide the crucial signals for cell fate determination, while extracellular matrix proteins like laminins and vitronectin establish a supportive environment for cell growth and maturation. Antibodies are used to verify cell identity and purity to guarantee that only properly differentiated cells progress toward therapeutic applications. Simultaneously, endotoxin-free proteins are vital to prevent triggering unwanted immune responses, particularly when cells are intended for clinical use.
Increasingly, researchers are using site-specifically labeled proteins to track receptor interactions and signaling events during differentiation, aiding protocol optimization to and improving overall reproducibility. Collectively, these molecular tools form the molecular foundation transforming patient-derived iPSCs into safe, effective therapeutic products.
In Vivo reprogramming: Precision tools for cellular conversion in the body
In vivo reprogramming represents a promising frontier in regenerative medicine, aiming to convert one cell type into another directly within the patient’s body, eliminating the need for cell extraction, culture, and reinfusion. By capitalizing on the intrinsic plasticity of cells, researchers can guide resident tissue cells toward therapeutic identities, potentially repairing damaged organs or restoring lost functions. For instance, preclinical studies in cardiac fibroblasts, and in the brain, have been converted into functional cardiomyocytes in the heart and astrocytes in the brain, highlighting the potential of this approach to regenerate tissues in situ without transplantation.
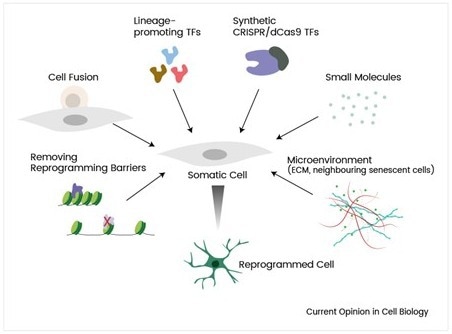
Figure 2. Overview of in vivo reprogramming strategies. Image Credit: https://doi.org/10.1016/j.ceb.2019.06.002
Another significant development is in vivo CAR-T therapy, where lipid nanoparticles (LNPs) or viral vectors deliver genetic instructions directly to a patient's T cells, reprogramming them to target and destroy tumors without ex vivo manipulation. While regulatory approvals for in vivo CAR-T therapies remain to be approved, the field has rapidly progressed from preclinical concepts to early-stage clinical trials. Promising early data in hematologic malignancies have already indicated preliminary efficacy, with investigations poised to expand into autoimmune diseases and solid tumors.
This paradigm shift marks a fundamental change in manufacturing by transitioning from complex, patient-specific 'living drug' production to scalable, off-the-shelf therapeutic administration. However, this transition imposes even greater demands on precise molecular tools. Recombinant proteins and antibodies are used to modulate pathways and validate cell identity, while endotoxin-free reagents minimize immune activation in animal models. Site-specifically labeled proteins and membrane targets enable researchers to track cellular conversion and monitor receptor interactions, offering critical insights into mechanisms and efficacy. Together, these tools facilitate controlled, reproducible exploration of in vivo reprogramming strategies, advancing their potential clinical applications.
Sino Biological’s contribution
The demands on molecular reagents increase dramatically as iPSC-derived cellular products and in vivo reprogramming strategies progress toward clinical development. Tools that perform well in research settings, including standard recombinant proteins, may not meet the requirements of scaled-up processes or regulatory expectations. Factors like batch-to-batch consistency, defined quality attributes, and ultra-low endotoxin levels become paramount for reproducible cell culture, precise differentiation, and safe administration in preclinical and clinical studies.
Sino Biological provides manufacturing-ready reagents that address these challenges. Their recombinant cytokines, growth factors, and extracellular matrix proteins are produced under controlled processes to maintain high purity and consistent bioactivity, supporting reliable cell expansion and lineage-specific differentiation. ProPure™ endotoxin-free proteins help minimize immune activation in animal studies and clinical-grade applications, while well-validated antibodies facilitate identity confirmation, potency testing, and quality control. Moreover, their platform includes transmembrane proteins for functional assays and site-specifically labeled biomolecules for real-time tracking of cellular responses and receptor interactions.
By offering high-quality, application-ready tools, Sino Biological allows research teams to bridge the gap between discovery and clinical translation. From supporting iPSC expansion and organoid formation to validating in vivo reprogramming strategies, Sino Biological reagents help ensure that promising cell therapies can progress with confidence from the bench to bedside. This integrated approach strengthens reproducibility and process robustness but also accelerates the journey of innovative cell therapies toward safe and effective patient applications.
About Sino Biological Inc.
Sino Biological is an international reagent supplier and service provider. The company specializes in recombinant protein production and antibody development. All of Sino Biological's products are independently developed and produced, including recombinant proteins, antibodies and cDNA clones. Sino Biological is the researchers' one-stop technical services shop for the advanced technology platforms they need to make advancements. In addition, Sino Biological offer pharmaceutical companies and biotechnology firms pre-clinical production technology services for hundreds of monoclonal antibody drug candidates.
Sino Biological's core business
Sino Biological is committed to providing high-quality recombinant protein and antibody reagents and to being a one-stop technical services shop for life science researchers around the world. All of our products are independently developed and produced. In addition, we offer pharmaceutical companies and biotechnology firms pre-clinical production technology services for hundreds of monoclonal antibody drug candidates. Our product quality control indicators meet rigorous requirements for clinical use samples. It takes only a few weeks for us to produce 1 to 30 grams of purified monoclonal antibody from gene sequencing.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.