Trace endotoxins (LPS) can have a negative impact on cancer research and therapy development. Their inclusion in preclinical investigations, including animal models for toxicity assessment or cytokine profiling, can jeopardize results and safety.
High-quality recombinant proteins are used in a variety of advanced techniques, including CAR-T and T cell treatments, monoclonal antibodies, ADCs, and bispecific antibodies. Therefore, endotoxin-free recombinant proteins are essential for credible research data and the successful development of next-generation cancer therapies.
Invisible interference in cancer therapy and cancer vaccine development
Endotoxin contamination can significantly impair antibody production in animal models. Even modest amounts can influence immunological responses, lowering antibody specificity, consistency, and overall quality.
Endotoxin-contaminated recombinant proteins can subtly yet significantly change cellular activity at the experimental level through immune stimulatory and cytotoxic effects. In animal research, endotoxin-induced systemic inflammation may compromise outcomes, potentially causing study delays or cancelation.
Endotoxin contamination can be a hidden disruptor in cell-based investigations, especially in LPS-sensitive assays. Immune cells such as dendritic cells, macrophages, monocytes, and T cells can react severely to even minimal quantities of endotoxin, resulting in cytokine production, altered proliferation, or unanticipated activation. These effects can easily lead to inaccurate or non-reproducible outcomes.
The need for endotoxin-free reagents is considerably greater in the development of cancer vaccines. Because these therapies rely on precise immune system modulation, LPS contamination can cause unwanted immunological activation, concealing the vaccine candidate's real efficacy and posing safety hazards.
Endotoxin-free proteins are therefore required to appropriately evaluate immunogenicity and support safe clinical translation.
Sino Biological’s ProPure™ solution to minimize endotoxin risk
Pharmacopeial guidelines, such as USP <85>, provide general limits for endotoxins, although cutting-edge immunology and translational oncology investigations often demand much stricter control.
Sino Biological's ProPure™ endotoxin-free recombinant proteins eliminate this variability at its source, ensuring consistent findings from early discovery to IND-enabling investigations.
ProPure™ reagents, manufactured at the cutting-edge Center for Bioprocessing (C4B) in Texas, are rigorously controlled to achieve levels as low as 0.05 EU/mg, with select items attaining an unprecedented 0.01 EU/mg, over 10 times lower than conventional industry standards.
By incorporating endotoxin-free proteins, researchers in cancer therapy and cancer vaccine development can acquire consistent and reliable results in essential applications, such as:
- Animal immunization for antibody generation, promoting high-quality antibodies and predictable immunological responses
- Preclinical toxicology and pharmacokinetics (PK), to minimize immune activation in animal models
- In vitro cell proliferation and differentiation experiments, reducing false positives from LPS-sensitive cells such dendritic cells, macrophages, and T cells
- Accurate cytokine detection and quantification, supporting valid immunological and biomarker analysis
How ProPure™ achieves ultra-low endotoxin levels
End-stage cleanup is not sufficient to reach ProPure™ quality. C4B implements an integrated Prevention-Isolation-Detection method throughout the production process.
- Prevention at the source: To prevent LPS introduction, it uses endotoxin-free plasmids and buffers, low-endotoxin-binding polymers, and strict CIP techniques throughout the cloning and purifying processes.
- Environmental isolation: A production process free of E. coli eliminates a key source of endotoxin exposure.
- Dual detection: Each batch undergoes dual detection using LAL and/or rFc assays to provide sensitive, redundant detection and traceability of results.
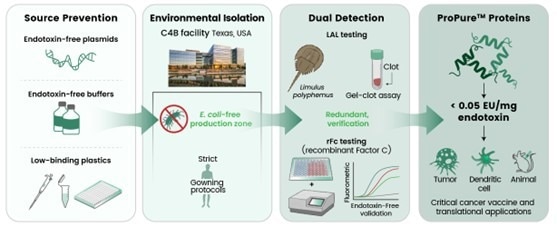
Figure 1. ProPure™ Triple-Control Strategy for Ultra-Low Endotoxin. Image Credit: Sino Biological Inc.
This end-to-end design ensures that when ProPure™ proteins arrive, they are ready for use in the most demanding oncology and immunology applications, without requiring additional purification processes that can harm protein quality or delay timeframes.
Summary
Sino Biological provides endotoxin-free proteins that meet the requirements of very sensitive research and translational applications through innovative technology and stringent quality control.
ProPure™ proteins aid in reducing variability, improving reproducibility, and accelerating the development of next-generation cancer medicines.
About Sino Biological Inc.
Founded in 2007, Sino Biological is a global biotechnology company specializing in high-quality recombinant proteins, antibodies, and CRO services. Serving researchers in over 90 countries, Sino Biological supports basic research, drug discovery, vaccine development, and diagnostics through its comprehensive product portfolio, proprietary quality systems, and innovative research platforms.
Sino Biological's core business
Sino Biological is a leading global biotechnology supplier dedicated to high-quality research reagents and comprehensive CRO services. Its extensive product portfolio covers over 9,800 recombinant proteins, 15,000 antibodies, and 50,000 genes, all produced in-house. As a trusted CRO partner, Sino Biological specializes in customized recombinant protein and antibody production. It stands out with its robust high-throughput antibody production system including HEK293/CHO and cell‑free platforms. This system enables rapid turnaround (as fast as 5 days), high-throughput production (>10,000 Abs/month) and cost-effective solutions for global academic and pharmaceutical clients.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.